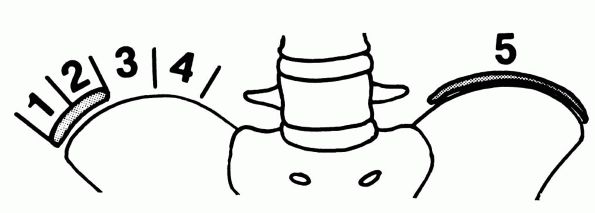
The Thoracolumbar Spine
or sagittal plane. All deformities of the spine are classified
according to the magnitude and the direction of the curvature, the
location of its apex, and the origin. Curvature of the spine may be
associated with many conditions (Table 16-1). The language used to describe the various aspects of spinal deformity is often confusing (Table 16-2).
incumbent on the examining physician to try to ascertain the origin
because this has bearing on the natural history of the condition and
implications for treatment. The cause may be determined on the basis of
a careful history, complete physical examination, and appropriate
imaging studies. The patient and family should be asked how the curve
was detected (e.g., school screening, observation by a friend or health
care worker, apparent body asymmetry) and their impression about
whether the curve is static or progressive. Idiopathic scoliosis in
children is not a painful condition. If the patient gives a history of
back pain associated with the deformity, other sources should be
considered; Scheuermann kyphosis, spondylolysis, spondylolisthesis, and
spinal or spinal cord tumor must be ruled out. Symptoms of shortness of
breath, physical limitations, and psychosocial effects possibly caused
by the curvature should be noted. The timing of reaching developmental
milestones should be recorded. A careful family history must be
obtained seeking any history of neurologic or congenital conditions
that may be associated with the curvature.
|
TABLE 16-1. SRS Classification of Spine Deformity
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
TABLE 16-2. Glossary of Scoliosis-Related Terms
|
|||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
progression depend on the patient’s growth potential, so it is
important to assess maturity historically. The age at onset of pubic
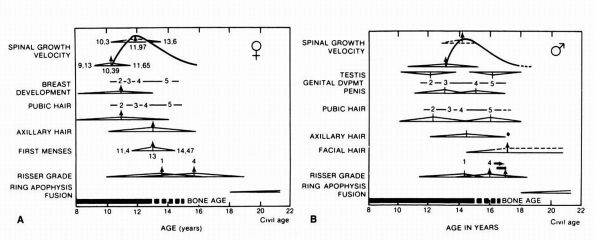
hair, axillary hair, breast budding, and menarche should be noted (Figure 16-1). Finally, details of any previous treatment for the condition should be noted.
paying particular attention to the patient’s body habitus and any
evidence of congenital abnormalities in the face, palate, ears, upper
extremities, and heart. The skin should be inspected for neurofibromas
or café-au-lait spots indicative of neurofibromatosis. The skin over
the sacral area should be examined for evidence of hair patches,
dimpling, pigmentation changes, nevi, and lipomas, which may be
associated with spinal dysraphism. Secondary sex characteristics,
including breast development and the presence of pubic and axillary
hair (graded according to the Tanner scale), must be noted. Limb
lengths should be measured to rule out limb length inequality. Sitting
and standing height should also be measured. A complete neurologic
examination including gag and abdominal reflexes must also be
performed. Any abnormality indicative of an associated neurologic
condition should be further investigated by a neurologist, and possible
further diagnostic studies—such as a spinal cord magnetic resonance
imaging (MRI), electromyography (EMG), and nerve conduction
velocities—should be considered. Abnormalities detected that suggest a
syndrome may require genetic consultation.
 |
|
FIGURE 16-1. (A) The relation of spinal growth velocity to maturity landmarks and the events of puberty in girls. (B)
The relation of spinal growth velocity to maturity landmarks and the events of puberty in boys. (Modified from Trever S, Kleinman R, Bleck EE. Growth landmarks and the evolution of scoliosis: a review of pertinent studies on their usefulness. Dev Med Child Neurol 1980;22: 675-684) |
The patient should be examined from both the front and the back,
looking for asymmetry in shoulder height, waistline, chest, scapular
height, and prominence. The relation of the thorax to the pelvis should
be noted. Spinal compensation can be measured by dropping a plumb line
from the spinous process of C7 and measuring the distance it falls from
the midgluteal cleft.
forward bend test. The test is performed by having the patient stand
with the feet together and the knees straight. The patient bends
forward at the waist with the arms dependent and the hands held with
palms opposed (Figure 16-3). The rotational
asymmetry is best assessed by viewing the patient from in front. Any
leg length inequality should be compensated for by placing an
appropriate-sized block underneath the short leg. The patient should be
assessed in three positions to observe the thoracic, thoracolumbar, and
lumbar spine (Figure 16-4). The patient should
also be viewed from the side to assess any abnormal increases in
thoracic or thoracolumbar kyphosis and any evidence of failure to
reverse the normal lumbar lordosis.
structural scoliosis, a standing posteroanterior (PA) unshielded
upright radiograph of the spine is ordered. The entire spine, as well
as iliac crests, must be included on the radiograph so that no curves
are missed and skeletal maturity can be assessed. If the patient has
complaints of pain or signs of a sagittal spinal deformity, a standing,
full-length lateral radiograph of the spine is also ordered.
 |
|
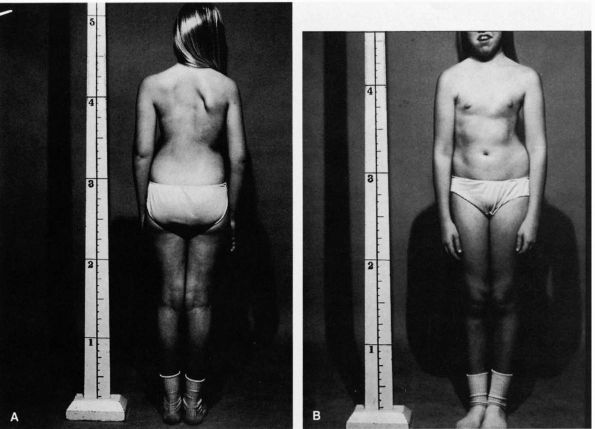
FIGURE 16-2. (A)
A patient with a typical right thoracic curve as viewed from the back. The left shoulder is lower, and the right scapula is more prominent. The thorax is shifted to the right with a decreased distance between the right arm and the thorax. Because of the shift in the thorax to the right, the waistline is altered with the left iliac crest appearing higher. This crest asymmetry is apparent, not real. (B) A patient with a typical right thoracic curve as viewed from the front. The left shoulder appears lower. The thorax is shifted to the right with a decreased distance between the right arm and the thorax. The left hip appears more prominent secondary to the rightward shift of the thorax. |
 |
|
FIGURE 16-3.
A patient with atypical right thoracic curve as viewed from the front on the forward bend test. Note the right thoracic prominence. |
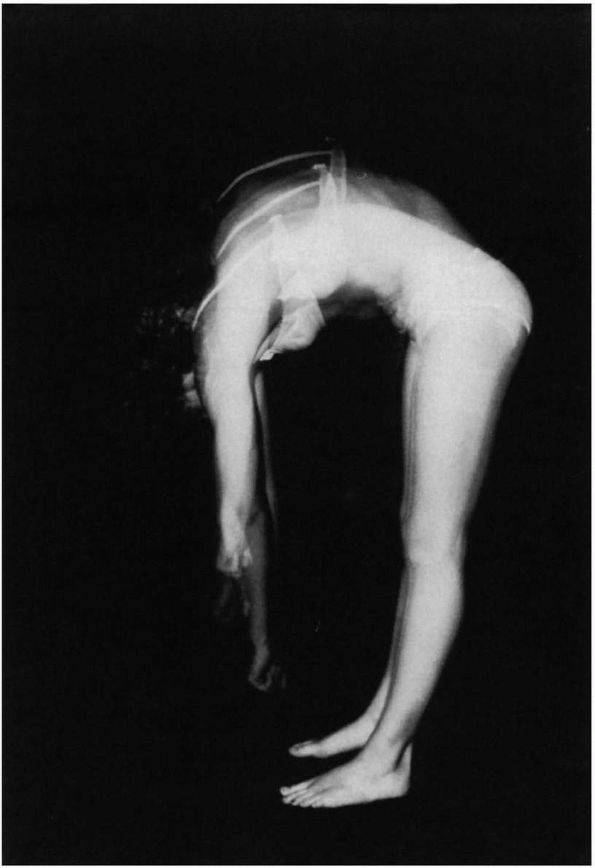 |
|
FIGURE 16-4. Forward bend test: three positions are required to observe the thoracic, thoracolumbar, and lumbar levels of the spine.
|
abnormalities in the vertebral bodies, ribs, and pelvis. All curves are
assessed for magnitude (Cobb measurements), direction (direction of the
curve convexity), location (e.g., thoracic lumbar, thoracolumbar,
double major, double thoracic), and pedicle rotation (Figure 16-5). Maturity is assessed by the Risser sign (Figure 16-6). Additional diagnostic and radiographic studies may be indicated if other than idiopathic scoliosis is suspected.
the probabilities of curve progression. These probabilities are based
on the natural history of the specific curve, which depends on its
origin, pattern, magnitude, and associated sagittal plane deformity.
This natural history must be considered in relation to the patient’s
growth potential as determined by history, physical assessment of
skeletal maturity, plotting of a growth chart and radiographic
assessment of maturity (i.e., Risser sign, ossification of vertebral
apophyses, wrist film for bone age assessment, or a combination of
these tests).
term that refers to a lateral curvature of the spine. The scoliosis may
be structural or nonstructural. A nonstructural scoliosis corrects or
overcorrects on supine side-bending radiographs or traction films. A
structural scoliosis is a fixed lateral curvature with rotation. On a
radiograph, the spinous processes in a structural curve rotate to the
curve concavity. On a supine side-bending radiograph or a traction
radiograph, a structural curve lacks normal flexibility. Many
conditions are associated with structural scoliosis (see Table 16-1). The most common structural curvature has no known cause and is referred to as idiopathic scoliosis.
Examples of nonstructural curvatures include scoliosis secondary to
limb length inequality or scoliosis secondary to a herniated nucleus
pulposus with nerve root irritation causing a list. If the primary
problem is corrected (e.g., the limb length inequality), the scoliosis
resolves.
natural histories. These varying natural histories profoundly influence
the effect of the curvature on the patient’s life and the indications
for treatment.
predisposition, and although many etiologic theories have been proposed, the cause remains unknown.
 |
|
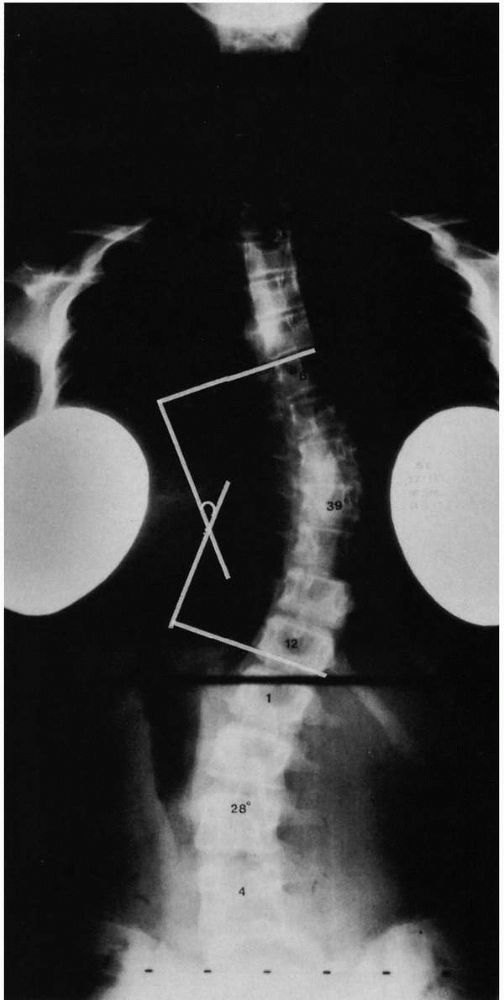
FIGURE 16-5.
Curve measurements (Cobb method). (1) Apparent perpendicular is erected from the endplate of the most caudal vertebrae, whose inferior endplate tilts maximally to the concavity of the curve (inferior end vertebrae). (2) A perpendicular is erected from the end-plate of the most cephalad vertebrae, whose superior end-plate tilts maximally to the concavity of the curve (superior end vertebrae). The curve value is the number of degrees formed by the angle of intersection of these perpendiculars, in this case 39°. |
 |
|
FIGURE 16-6.
Ossification of the epiphysis usually starts at the anterosuperior iliac spine and progresses posteriorly. The iliac crest is divided into four quarters, and the excursion or stage of maturity is designated as the amount of progression. |
by age at onset of the conditions: infantile (0 to 3 years of age),
juvenile (3 to 10 years of age), and adolescent (older than 10 years of
age but before maturity). Although these subtypes may represent a
continuum of the same condition, their natural histories differ.
Therefore, these three subtypes of idiopathic scoliosis are considered
separately. An alternative classification puts patients into two main
categories: early onset (under 10 years of age) and late onset (over 10
years of age). This classification is more reflective of outcome; with
early onset cases having more severe effects on outcome measurements
(pulmonary function compromise; deformity; mortality) than those with
the late onset variety.
deformity detected during the first 3 years of life. It accounts for
less than 1% of all cases of idiopathic scoliosis in the United States.
It is more commonly seen in Europe, especially Great Britain. Most of
these curves develop within the first 6 months of life, with the left
lumbar curve pattern being the most common. Epidemiologic and
associated problems include older maternal age, increased incidence of
inguinal hernias among relatives, and association with congenital heart
disease (2.5%), congenital hip dysplasia (3.5%), and developmental
problems, particularly mental retardation (13%). Intrauterine molding
is thought to be a cause because 83% of patients have plagiocephaly,
and more than half have evidence of rib-molding deformities. Natural
history studies indicate that 85% of the curves regress spontaneously,
particularly if the curve onset was before 12 months of age. Fifteen
percent of the curves may progress, often leading to severe
deformities. Compensatory curves are generally not seen in patients
with infantile idiopathic scoliosis. Because of the early onset of the
spinal curvature and its effect on pulmonary parenchyma development,
patients with progressive untreated infantile, early onset scoliosis
may develop severe restrictive pulmonary disease, cor pulmonale, and
early death.
 |
|
FIGURE 16-7. (A)
The rib-vertebral angle difference is calculated by subtracting the convex value from the concave value at the thoracic curve apical vertebra. (B) Phase changes at apical vertebra. Phase 2 appearance denotes probable progression. |
years) includes congenital scoliosis and scoliosis of a neuromuscular
origin or intraspinal pathology (e.g., Chiari malformations;
syringomyelia). Careful neurological examination is imperative.
Radiographs rule out congenital spinal abnormalities. In this age
group, MRI of the brain and spinal cord is warranted because of the
increased association with intraspinal pathology (Chiari malformation
and syrinx) with early onset scoliosis.
radiographs should be obtained. The Cobb angle and the rib-vertebral
angle difference should be measured (Figure 16-7).
If the rib-vertebral angle difference is greater than 20°, the
curvature is likely progressive. With measurements of less than 20°,
the curve is likely to resolve. All patients must be followed by serial
radiographic examination, calculating the Cobb angle and the
rib-vertebral angle difference at each visit. In progressive curves,
the convex side rib head is overlapped by the shadow of the vertebral
body. This radiographic sign indicates a progressive curve. Curves that
maintain a Cobb angle of less than 35° have a high likelihood of
resolution.
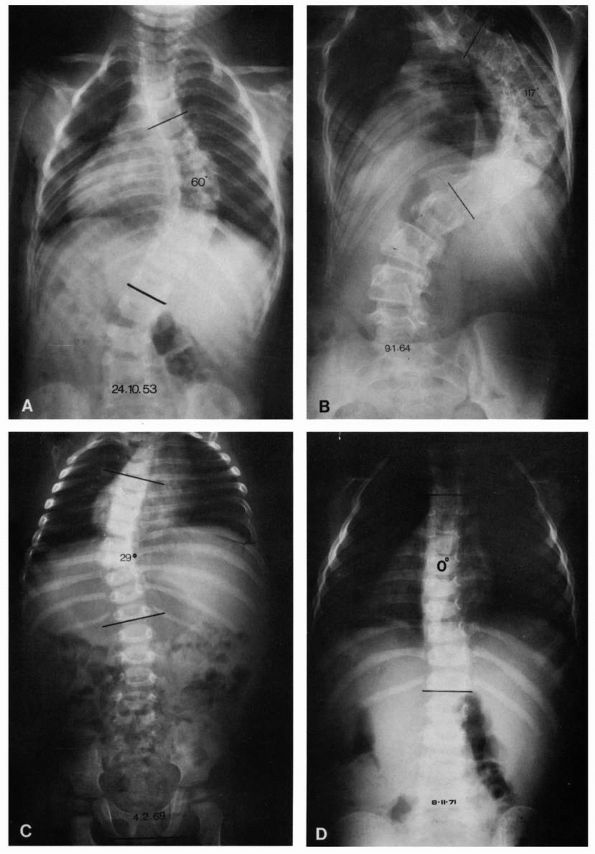
Compensatory curves are not common in patients with infantile
idiopathic scoliosis. The development of a compensatory curve is a bad
prognostic sign that indicates a probable curve progression (Figure 16-8).
Cobb angle and less than a 20° rib-vertebral angle difference. The
patient should be reevaluated in 4 to 6 months with a repeat standing
radiograph of the spine. With resolution of the curvature, the patient
can be followed at 1- to 2-year intervals. In curves with a 25 to 35°
Cobb angle and with a rib-vertebral angle difference of 25°, repeat
clinical and radiographic evaluation at 4- to 6-month intervals is
warranted. If the Cobb angle increases by 5 to 10° with or without
changes in the rib-vertebral angle difference, treatment is indicated.
serial casting to correct the deformity followed by the use of a
Milwaukee brace (cervical-thoracic-lumbar-sacral orthosis) or another
spinal orthotic to maintain correction. In the infantile patient, the
corrective cast usually needs to be applied with sedation or under
general anesthesia. The casts are worn for 6 to 12 weeks and are
serially changed until maximal correction is obtained. An orthotic is
then fabricated and worn full time (22 to 23 hours per day) for 2 to 3
years to maintain the correction obtained by casting. If correction is
maintained, the patient may be gradually weaned from the brace. If
progression occurs, full-time orthotic use must be reinstituted.
orthotic, subcutaneous “growing rods”—spinal instrumentation without
fusion—followed by bracing is indicated. The rod can then be lengthened
periodically to allow for growth with a formal posterior spinal fusion
and instrumentation at maturity. Recent studies have questioned the
success of these “growing rod” techniques, and thus in certain cases,
even at a very young age (over 5 years of age), definitive surgical
intervention may need to be considered.
the spine that presents after 3 years of age but before the adolescent
growth spurt. Patients classified as having juvenile idiopathic
scoliosis may actually have late-onset infantile idiopathic scoliosis
or early-onset adolescent idiopathic scoliosis. This group of patients
accounts for about 20% of all idiopathic scoliosis patients.
girls, with right thoracic curves accounting for about two-thirds of
all curve patterns. Double major curves (right thoracic and left
lumbar) and thoracolumbar curves follow in frequency.
and cause severe deformity. Some may progress relentlessly from onset,
but others may be stable or progress slowly until the adolescent growth
spurt when rapid progression ensues. Unlike some infantile curves,
juvenile idiopathic scoliosis curvatures do not resolve spontaneously.
This group of patients should also have routine brain and spine MRI
studies to rule out intraspinal pathology.
25° or more. The rib-vertebral angle difference has not been shown to
be prognostic in these patients. Curves rarely progress more than 1°
per month. Therefore, if the patient has a curve of less than 20°,
follow-up evaluation in 6 to 8 months is appropriate. Treatment is
indicated for progression of at least 10°.
follow-up evaluation clinically and radiographically should be obtained
in 5 to 6 months, with treatment indicated for a greater than 5°
increase in curve. For curves greater than 25°, because of the high
probability of progression, treatment should begin immediately.
evaluation and in some cases by supine side-bending radiographs,
orthotic treatment is indicated in an attempt to prevent further
progression. If the curve is rigid, serial cast correction, much like
that recommended for infantile idiopathic scoliosis, is warranted
before fitting the patient with a spinal orthotic. The orthosis is worn
on a full-time basis (although protocols for brace wear may vary) for
several years until curve correction is achieved. Then, weaning may
begin and continue as long as curve correction is maintained. The brace
is then worn at night only until the patient reaches skeletal maturity
(i.e., Risser grade 4 or 5, or no spinal growth for the previous 18
months).
After surgery, the patient continues in an orthosis. The distraction
device is lengthened with skeletal growth until the patient reaches
puberty, when posterior spinal fusion and instrumentation are
performed. In this age group, however, the worry about loss of growth
potential associated with definitive surgical fusion of the spine is
less concerning than in the very early onset group (infantile) and
hence definitive fusion and instrumentation may need to be considered
in this group if progression occurs despite bracing (Figure 16-9).
 |
|
FIGURE 16-8. (A) Progressive type of infantile idiopathic scoliosis, early radiograph. Curve measures 60°. (B) Late radiograph showing marked increase of primary curve and developing secondary curves. (C) Resolving type, early radiograph. Curve measures 29°. No secondary curve. (D) Resolving type, later radiograph. Curve reduced to zero. (Courtesy of Dr. J. I. P. James)
|
of the spine presenting at or about the onset of puberty and before
maturity. Adolescent idiopathic scoliosis accounts for about 80% of
cases of idiopathic scoliosis. Its origin is unknown. The prevalence
(i.e., occurrence in the at-risk population, children 10 to 16 years of
age) of adolescent idiopathic scoliosis is about 2 to 3%. Although
there is an overall female predominance for the condition (3.6:1); the
prevalence in males and females is equal in small-magnitude curves
(10°). With increasing curve magnitude, there is an overwhelming female
predominance (curves greater than 30°; female predominance 10:1).
 |
|
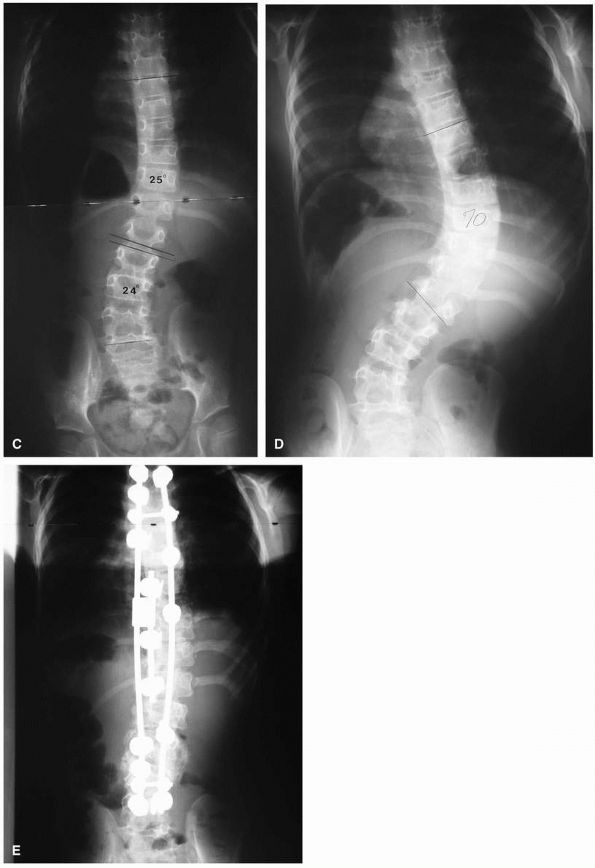
FIGURE 16-9. (A) Juvenile idiopathic scoliosis in a 7½-year-old boy with an 80° thoracic curve that progressed despite bracing. (B)
Same patient at age 8 years and 11 months. Curve is maintained at 41° with distraction instrumentation and fusion at hook sites. Patient is wearing TLSO external support. Definite surgery is planned at puberty. (C) A 7½-year-old girl with juvenile onset scoliosis; she was initially treated in a brace but her curve progressed (D), necessitating anterior and posterior fusion (E). |
Because most thoracic curve patterns are convex to the right, a child
presenting with a left convex thoracic curve should be examined
carefully for a neurologic deficit. In this situation, neurologic
consultation and MRI scanning should be considered because of the high
association of intraspinal pathology with this curve pattern. Also, a
history of rapid progression of an adolescent curvature should alert
the physician to consider similar diagnostic evaluations.
radiographs of the entire spine taken in the standing position. At
follow-up visits, only PA radiographs are usually necessary. It is
important to minimize the radiation that the patient receives over
time. Radiographs should be taken only when necessary for treatment
decisions. Appropriate technique should be used to avoid the need for
repeat films. Other radiation protection measures include beam
collimation, antiscatter grids, beam filtration, high-speed film,
intensifying screens, gonadal and breast shields, and PA as opposed to
anteroposterior (AP) projections. The use of the PA projection avoids
radiation to the developing breast tissue, which is radiosensitive.
Special radiographic views, such as side-bending radiographs, are
rarely indicated unless the patient is being considered for surgical
management. Each radiograph is measured for the Cobb angle, rotation,
and ossification of the iliac apophysis (Risser sign).
 |
|
FIGURE 16-9. (Continued)
|
 |
|
FIGURE 16-10. (A)
Thoracic curve. Ninety percent right convexity involving an average of six vertebrae: apex—T8, T9; upper end vertebrae—T5, T6; lower end vertebrae—T11, T12. (B) Lumbar curve. Seventy percent left convexity involving an average of five vertebrae: apex—L1, L2; upper end vertebrae—T11, T12; lower end vertebrae—L3, L4. (C) Thoracolumbar curve. Eighty percent right convexity involving an average of six to eight vertebrae: apex—T11, T12; upper end vertebrae—T6, T7; lower end vertebrae—L1, L2. (D) Double curve. Ninety percent right thoracic convexity and left lumbar convexity. Thoracic component, average five vertebrae: apex—T7; upper end vertebrae—T5, T6; lower end vertebrae—T10. Lumbar component, average five vertebrae: apex—T2; upper end vertebra—T11; lower end vertebra—L4. |
natural history of that condition. It is important to understand the
natural history of untreated adolescent idiopathic scoliosis with
regard to curve progression, effect on pulmonary function, back pain,
mortality, psychosocial problems, and effect of on pregnancy.
or the probability of curve progression. Most information available on
curve progression is from studies of girls, particularly those with
thoracic curves. The factors that influence the probability of curve
progression in the immature patient include growth potential factors,
such as age, gender, and maturity, and curve factors, such as type and
magnitude. Double-curve patterns have a greater tendency for
progression than single-curve patterns. Curves detected before menarche
have a much greater chance of progression than those detected after
menarche. With increasing age at detection, there is a decreasing risk
of curve progression. The larger the curve magnitude at detection, the
greater the chance of progression; the lower the Risser grade at curve
detection, the greater the risk of progression. The risk of progression
for boys is about one-tenth that of girls with comparable curves.
skeletal maturity. Large-magnitude curves, however, may continue to
progress after maturity (Table 16-3). Many
curves continue to progress throughout the patient’s life. In general,
curves less than 30° at maturity tend not to progress regardless of the
curve pattern. Many curves greater than 30°, and particularly thoracic
curves greater than 50°, continue to progress.
general population is about 60 to 80%, although the incidence varies
considerably. The incidence of back pain in patients with scoliosis is
comparable to that in the general population. Scoliosis patients,
however, often have an increased incidence of frequent or daily
backache compared with the general population. Patients with lumbar and
thoracolumbar curves, particularly those with lateral listhesis or
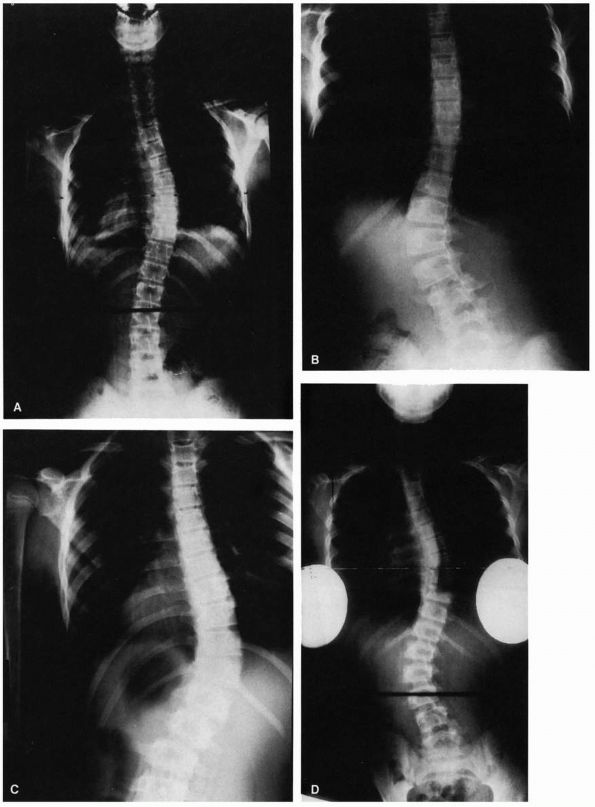
translatory shifts (Figure 16-11) at the lower
end of their curves, tend to have a slightly greater incidence of
backache than patients with other curve patterns. Back pain in adult
patients with scoliosis is not always related to the curvature; it may
emanate from the counter-curve below, or it may be discogenic,
neurogenic, or facet joint related.
|
TABLE 16-3. Probabilities of Progression Based on Curve Magnitude and Age
|
|||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||
curves is there a direct correlation between decreasing vital capacity
and FEV1 with increasing curve severity. In all other curve patterns in
idiopathic scoliosis, there is no direct correlation between curve
magnitude and limitation in pulmonary function. Most patients with
adolescent idiopathic scoliosis have loss of the normal thoracic
kyphosis. This loss of thoracic kyphosis (hypokyphosis) further
diminishes pulmonary function associated with increasing curve severity.
scoliosis. The only patients at risk are those with high-angled
(greater than 100°) thoracic curvatures. In these patients, mortality
rates are significantly increased because of secondary cor pulmonale
and right ventricular failure.
with psychosocial concerns. There is, however, no correlation between
the location or degree of the curvature and the extent of the
psychosocial effects. Some adults with moderate to severe deformity may
have severe psychosocial problems.
are the same as those of nonscoliotic women. Whether pregnancy causes
curve progression is unknown, with evidence being present on both sides
of the issue.
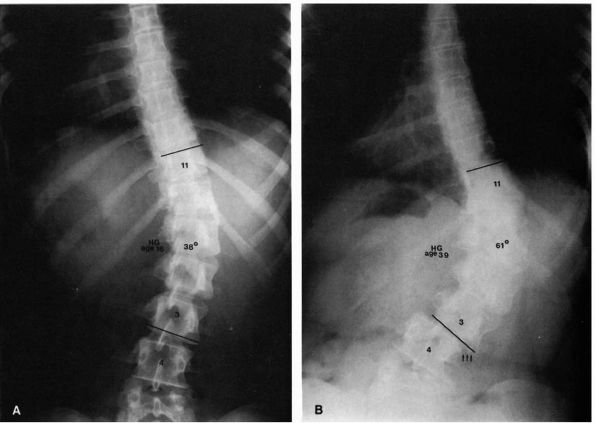 |
|
FIGURE 16-11. (A)
Sixteen-year-old girl with a 38° right lumbar curve from T11 to L3. Her skeletal maturity is assessed as grade 5 on the Risser scale. (B) At 39 years of age, her right lumbar curve has increased to 61°. Note the translatory shift of L3 on L4 (arrows). (Weinstein SL. The natural history of scoliosis in the skeletally mature patient. In: Dickson JH, ed. Spinal deformities, vol 1. Philadelphia: Hanley & Belfus, 1987:199) |
require active treatment (less than 10%). It is important to
individualize all treatment decisions, taking into consideration the
probabilities of progression based on the curve magnitude, skeletal and
sexual maturity of the patient, and age of the patient. The general
indications for treatment are a progressive curve of 25° or more in a
skeletally immature patient. In a skeletally immature patient with a
curve of less than 19°, curve progression of 10° should be documented
before instituting treatment. If the curve is between 20 and 29°,
progression of least 5° should be documented before instituting
treatment. If a curve on initial evaluation is over 30°, because of the
high probability (over 90%) of progression, no documentation of
progression is necessary, and treatment should be initiated
immediately. Because curves rarely progress at more than 1° per month,
follow-up appointments can be scheduled accordingly (i.e., for a 15°
curve in a skeletally immature girl, follow-up reevaluation is in 10
months).
adolescent idiopathic scoliosis has been the Milwaukee brace. Despite
widespread use, few long-term studies have evaluated the results of
treatment with the Milwaukee brace (Figure 16-12).
In most of the studies performed, curve progression was not documented;
thus, it is uncertain whether those patients braced would have had
continued progression had they not been braced.
for scoliosis has been done to date. Reviews of the few studies that
are available demonstrate conflicting results. The general feeling
among physicians treating scoliosis is that bracing appears to alter
the natural history of curve progression. It is generally accepted that
the curve progression can be arrested in 85 to 90% of at-risk patients.
The most common response to bracing is a moderate amount of correction
while the brace is worn, with slow, steady progression of the curve
back to the original magnitude after weaning from the brace.
Occasionally, maintenance of correction obtained in the brace occurs in
some patients who achieve at least a 50% reduction in their curvature
during the course of treatment. The brace is worn 22 to 23 hours a day
and
is removed only for bathing or sporting activities. When the patient
reaches skeletal maturity (i.e., Risser grade 4, or no spinal growth
over an 18-month period), the patient is gradually weaned out of the
brace. The brace is used on a part-time basis followed by nighttime use
only so long as no increases in curvature are noted. Bracing is
reported to be ineffective in curves greater than 40°.
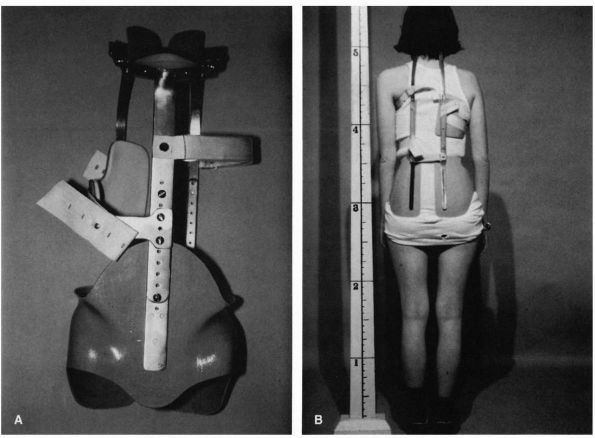 |
|
FIGURE 16-12. Milwaukee brace (CTLSO). The brace was developed in the late 1940s (A, frontal view of bone). Despite its widespread use, few long-term studies are available that evaluate the results of treatment (B,
view from behind with patient wearing brace). In addition, most studies fail to document curve progression; thus, it is uncertain whether those patients braced would have had continued progression had they not been braced. This type of brace was the “gold standard” for bracing but is rarely used today. It has been replaced by various models of underarm low profile braces such as seen in Figure 16-13. There are no published prospective controlled studies on bracing. |
physicians are choosing part-time bracing programs and using underarm
orthoses (Figure 16-13) because of compliance
problems. An underarm brace (thoracic-lumbar-sacral orthoses, TLSO) is
generally acceptable for use in curves with an apex of T8 or below.
Electrical stimulation was used in the past but has been shown to be
ineffective. Although bracing for scoliosis remains in common usage, as
mentioned earlier, final determination as to the success of bracing in
adolescent (late onset) idiopathic scoliosis must await a randomized,
prospective, controlled clinical trial.
evidence of curve progression despite bracing or has a curve magnitude
that would be unsuccessfully treated by a brace (i.e., greater than 45
to 50° and skeletally immature). In the adult patient with adolescent
idiopathic scoliosis, indications for surgical treatment include pain
unresponsive to nonsurgical treatment and documented curve progression.
involves a posterior spinal fusion in combination with one of the
various forms of spinal instrumentation (Figure 16-14).
The purpose of the procedure is to obtain a spinal fusion.
Instrumentation is used to correct the deformity and prevent bending of
the fusion mass (Figure 16-15). Over the past
few years, many implant devices have been introduced for the correction
of spinal curvatures. These devices allow for correction of the
sagittal in addition to the coronal plane deformity (see Figure 16-14).
Some of these devices allow for decreasing the number of segments of
the spine that need to be fused by doing the procedure from the front
of the spine.
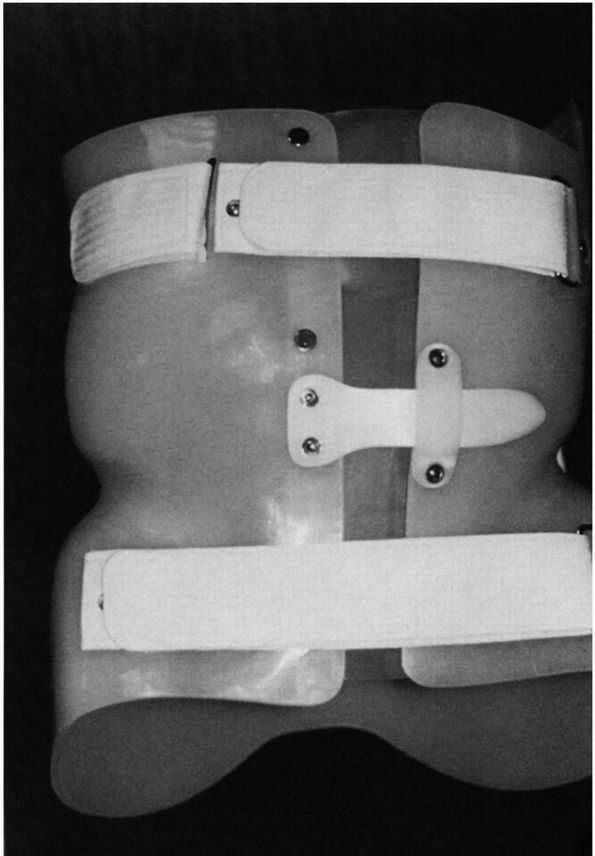 |
|
FIGURE 16-13. Underarm brace (TLSO): frontal view of one model.
|
 |
|
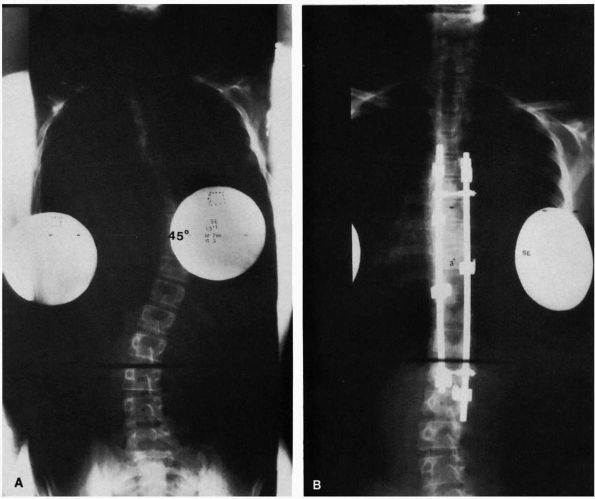
FIGURE 16-14. (A) Thirteenyear, 1-month-old girl with a curve that progressed from 23 to 45° despite bracing. (B) After spinal fusion and Cotrel-Dubousset instrumentation, the same patient’s curve measures 3°.
|
of vertebral development. These abnormalities may result in scoliosis,
kyphosis, lordosis, or combinations of these. Deformities may be of
three structural types: failure of formation (e.g., hemivertebrae);
failure of segmentation (e.g., unilateral unsegmented bar); or
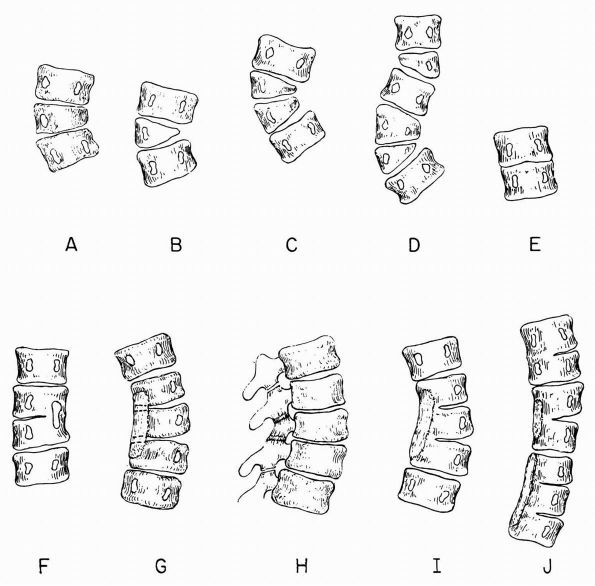
combinations of defects of segmentation and formation (Figures 16-16 and 16-17).
The resultant deformity is related to the location and type of the
congenital anomaly and to the growth potential of the unaffected
segments; for example, a lateral segmentation defect causes a pure
scoliosis, a posterolateral segmentation defect causes a
lordoscoliosis, and an anterior failure of segmentation causes a
kyphosis. With defects of formation, any portion of the vertebrae may
be hypoplastic or absent. Absence of a vertebral body causes a pure
kyphosis, and presence of the posterolateral portion of the vertebrae
causes a kyphoscoliosis. Failure of formation of various portions of
the posterior elements results in spina bifida.
thought to have genetic implications. Patients with congenital
scoliosis may, however, have other associated congenital abnormalities.
The most frequently affected systems are the genitourinary, cardiac,
and spinal cord. Some syndromes (e.g., VATER syndrome) are also
associated with congenital vertebral abnormalities. Many congenital
spine deformities are discovered only incidentally on radiographs taken
for other reasons, and some are associated with severe deformities
noted at birth.
 |
|
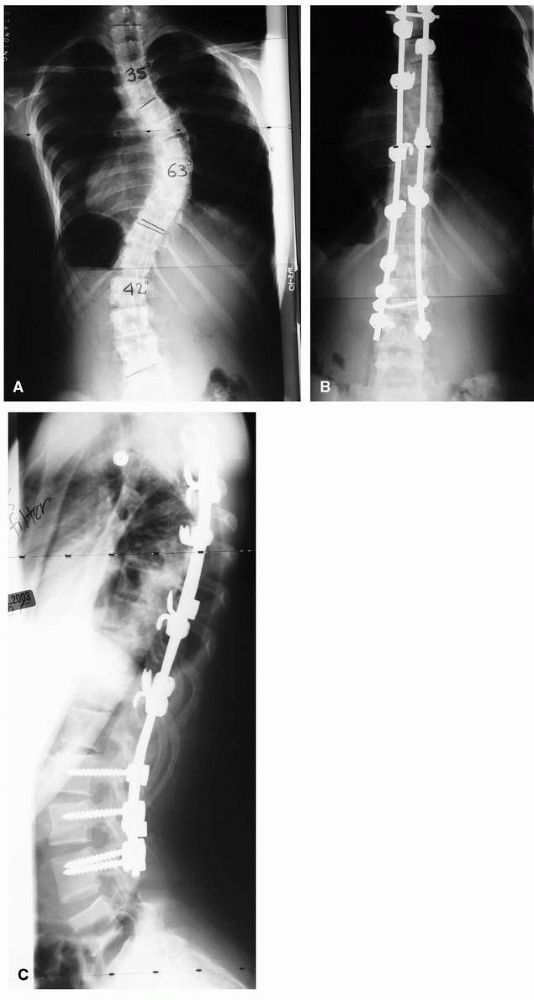
FIGURE 16-15. Spinal fusion for progressive idiopathic scoliosis using a hybrid system of rods, hooks, and pedicle screws. (A) preoperative PA view; (B) postoperative PA; and (C) lateral view.
|
 |
|
FIGURE 16-16. Scoliosis anomalies. (A) Unilateral failure of vertebral formation, partial (wedged vertebra). (B) Unilateral failure of vertebral formation, complete (hemivertebra). (C) Double hemivertebrae, unbalanced. (D) Double hemivertebrae, balanced. (E) Symmetric failure of segmentation (congenital fusion). (F) Asymmetric failure of segmentation (unsegmented bar). (G) Asymmetric failure of segmentation (unsegmented bar involving posterior elements only, anteroposterior view). (H)
Asymmetric failure of segmentation, oblique view showing intact disc space and lack of segmentation confined to the posterior elements (surgically easy to divide). (I) Unsegmented bar involving both the disc area and posterior elements (a very difficult surgical problem to divide this). (J) Multiple unsegmented bars, unbalanced. (Winter RB, Moe JH, Eilers VE. Congenital scoliosis: a study of 234 patients treated and untreated. J Bone Joint Surg 1968;50A:1) |
 |
|
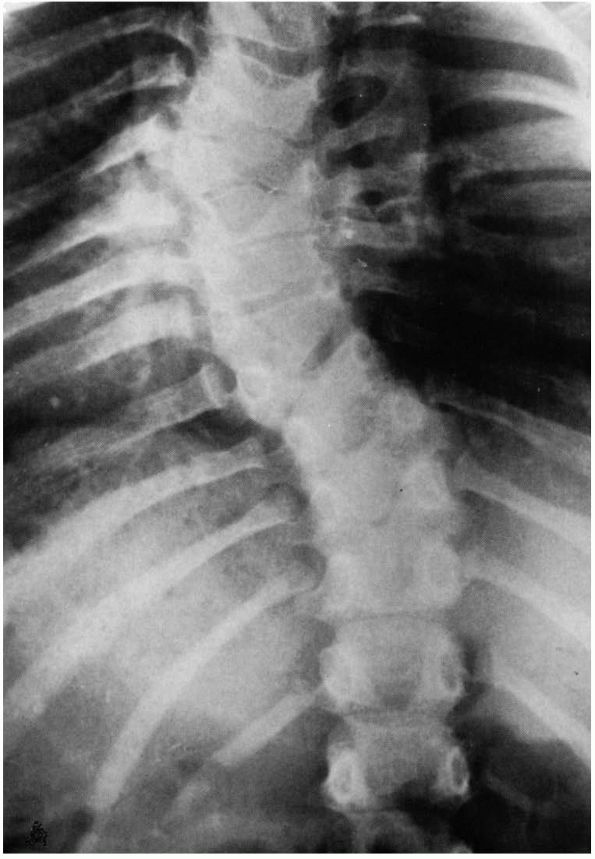
FIGURE 16-17. Congenital scoliosis. The ribs on the concave side are misshapen and fused. The seventh thoracic vertebra is wedged.
|
produce severe functional and cosmetic deformities. If the deformity is
associated with kyphosis, spinal cord compression and paralysis may
occur. Early detection and careful follow-up of these patients is
imperative.
a careful and detailed neurologic examination. Five to 20% of patients
have associated spinal dysraphism (e.g., tethered cord,
diastematomyelia, dural lipoma). Subtle physical findings, such as limb
atrophy or mild foot abnormalities, may be the only evidence of spinal
dysraphism. The skin should be inspected over the spine, looking for
hair patches, dimpling, cyst formation, and hemangiomas, which are
often associated with a spinal dysrhaphic condition. The chest wall
should be examined for any evidence of defects or asymmetry.
associated cardiac abnormalities. A cardiologist should evaluate any
cardiac abnormality detected.
have a urologic evaluation. Twenty to 40% of patients with congenital
spinal abnormalities have an associated abnormality in the
genitourinary tract. Six percent of these genitourinary tract
abnormalities are potentially life threatening. Renal ultrasound is
generally sufficient to provide a screening test for genitourinary
tract abnormalities. Evaluation of the lower tracts, however, may
require excretory or retrograde urograms. The kidneys are also seen
well on the MRI.
supine AP and lateral radiographs. These films provide the best detail
of the congenital abnormalities (Figure 16-18).
It is important to pay attention to the sagittal plane deformity
because many patients with congenital scoliosis may have accompanying
kyphosis or lordosis. A baseline lateral radiograph should be obtained
to assess any associated lordosis or kyphosis. Sagittal plane deformity
may progress without progression of the scoliosis as the patient
matures. Follow-up radiographs can be taken in the standing position
(when possible) and measured by the Cobb angle to follow curve
progression.
 |
|
FIGURE 16-18.
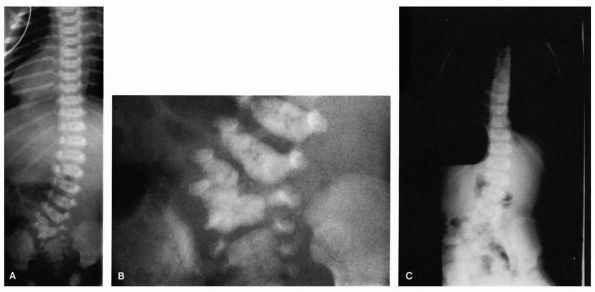
Eight-day-old boy with lumbosacral hemivertebrae. Radiographs of newborns generally provide the best detail of congenital spine abnormalities. (A) Spine film. (B) Coned-down view of lumbosacral junction. (C) Standing film at 2 years of age; because of progression of curve and pelvic obliquity, patient underwent excision of hemivertebra. |
abnormalities, such as hemivertebrae or nonsegmented vertebrae. The
ribs should be examined for congenital abnormalities, and all pedicles
should be counted, disc spaces examined, and growth potential assessed.
The prognosis for congenital deformities depends on the presence of
asymmetric growth. Pedicular widening may be a sign of
diastematomyelia, especially in patients with cutaneous or clinical
manifestations of spinal dysraphism. CT scanning may be helpful in
defining some congenital lesions, particularly in older patients. MRI
is indicated if the patient has any evidence of spinal dysraphism or
prior to any corrective surgery.
relates specifically to the location of the abnormality (e.g.,
thoracic, thoracolumbar, lumbar spine), the type of abnormality (e.g.,
unilateral unsegmented bar, hemivertebrae), and the patient’s spinal
growth potential. In general, about half of all congenital spine
anomalies have significant enough progression to require treatment. By
counting the number of growth centers, as represented by the pedicles
on the concave and convex sides of the curve, as well as examining the
quality of the disc spaces between vertebrae, the physician can
estimate the probability of curve progression. If there is greater
growth potential in the convexity of the curve than the concavity,
progression is certain. The
worst
prognosis for progression of congenital anomalies is the unilateral
unsegmented bar opposite to a convex hemivertebrae, followed by the
unilateral unsegmented bar and the double-convex hemivertebrae (see Figure 16-16).
hemivertebrae is difficult to predict in that there are three types of
hemivertebrae: fully segmented (worst prognosis), semisegmented, and
nonsegmented (most benign). The younger the patient at the age of
detection, the more likely the patient is to have a progressive
deformity. If a congenital anomaly is detected at an older age or only
incidentally, it rarely causes significant problems.
until skeletal maturity. Patients with congenital spinal deformity
should be followed with radiographs every 6 months during the first 3
years of life. If the curve remains stable, follow-up can be on a
yearly basis until the adolescent growth spurt, when repeat evaluations
every 6 months may be warranted.
therefore, bracing is generally not a treatment option. Bracing,
however, can be used in certain situations, particularly in long,
flexible curves or compensatory curves above or below congenital
abnormalities.
is an indication for surgical stabilization. In certain instances in
which the natural history is well known (e.g., unilateral unsegmented
bar opposite a convex hemivertebrae), surgical stabilization is
indicated without documentation of progression. The standard method of
surgical stabilization is posterior spinal fusion without
instrumentation. The deformity is then corrected by a plaster cast.
This type of treatment is at times (particularly in the young patient)
associated with bending of the fusion mass and continued rotational
curve progression (often referred to as the “crankshaft phenomenon”).
Promising results have been reported with combination anterior
hemiepiphysiodesis and posterior hemiarthrodesis, even demonstrating
correction for some curves by growth on the unfused concave side in
very young patients with significant growth potential and with certain
types of congenital deformities.
cases. This mode of treatment, however, is associated with an increased
risk of neurologic deficit. If correction of the congenital spinal
deformity is contemplated, thorough investigation for intraspinal
pathology (MRI) must be done before operation. The presence of a
diastematomyelia without neurologic deficit does not require treatment.
If correction of the spinal deformity is contemplated, however, then
the diastematomyelia must be addressed surgically. Hemivertebrae
excision may be considered in cases of a lumbosacral hemivertebrae with
spinal decompensation (see Figure 16-18).
surgical. Patients with failure of formation causing either pure
kyphosis or kyphoscoliosis are at high risk for spinal cord
compression. Posterior hemivertebrae or failure of formation of a
vertebral body should be treated by immediate surgical stabilization.
These patients generally benefit from a combined anterior and posterior
surgical spinal fusion (Figure 16-19).
may ensue, although this rarely leads to neurologic deficit. A short,
posterior fusion arrests spinal growth and halts progression of the
deformity.
If
the deformity exceeds 50°, anterior fusion must be done in conjunction
with the posterior spinal fusion; otherwise progression may occur by
bending of the fusion mass. Any neurologic deficit associated with
congenital kyphosis or congenital kyphoscoliosis must be treated by
spinal cord decompression and stabilization.
 |
|
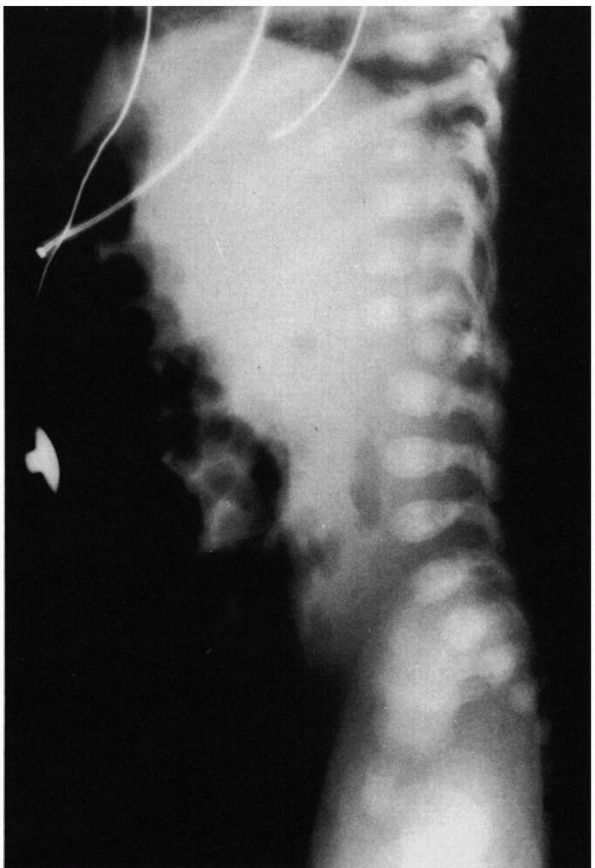
FIGURE 16-19.
Six-week-old boy with hypoplasia of L1 with kyphosis. At 14 months of age, the patient underwent AP spinal fusion to prevent progression. |
warrants careful evaluation because, although back pain is a common
complaint in adults, back pain in children is uncommon. The younger the
child is the more worrisome the complaint.
pain after strenuous physical activity, prolonged sitting or standing,
or repetitive heavy lifting. Back pain may also accompany a viral
illness. Repetitive or chronic backache symptoms should be carefully
investigated. There are many causes of backache in children (Table 16-4), and the cause can be established in about 85% of cases.
and characterize the pain. The pain should be characterized as to
nature, intensity, location, and time of occurrence. Was the onset of
the pain acute or insidious? Is it associated with any other illnesses?
Is it activity related? Is it relieved by rest? Does it occur in the
morning, the afternoon, or with repeated stress? Are there any
associated bowel or bladder symptoms? What makes the pain better? What
makes it worse? Is there associated night pain? Does aspirin or NSAIDs
relieve the pain? Are there associated radicular symptoms, leg pains,
or paresthesia? Does the child have any complaints of a systemic
illness, such as fever, weight loss, or general malaise? Is there a
history of injury? Is the pain associated with repetitive trauma, such
as gymnastic activities or other sports?
|
TABLE 16-4. Causes of Back Pain in Children
|
||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||
able to narrow down the possible sources of the back pain. For example,
if the patient is complaining of back pain usually at night with
symptoms relieved by aspirin, an osteoid osteoma or osteoblastoma
should be suspected. Pain during athletic events, such as gymnastics or
blocking in a football lineman, is suggestive of spondylolysis or
spondylolisthesis. Leg pain with or without associated back pain may be
radicular pain associated with a herniated nucleus pulposus or may be
secondary to hamstring tightness because of cauda equina irritation
from a spondylolysis or spondylolisthesis. Back pain in children with
or without neurologic deficits may also be a manifestation of
intraspinal pathology.
chief complaint of back pain should consist of a general overall
evaluation, including examination of the head and neck, the upper and
lower extremities, gait, and spine. A careful, detailed neurologic
examination, including muscle, reflex, and sensory testing, is
mandatory. The presence of normal or abnormal contours of the spine in
either the coronal or sagittal plane should be noted. Skin lesions,
such as nevi, sinuses, hair patches, or abnormal pigmentation over the
lumbosacral area may indicate spinal dysraphism. The Adams forward-bend
test should be performed, looking for evidence of scoliosis or an
exaggerated kyphosis. Spinal motion should be assessed to be certain
that the patient reverses the normal lumbar lordosis. Any loss of
normal spinal motion or failure to reverse lumbar lordosis on forward
flexion is suggestive of a pathologic condition and should be
investigated.
over the spine or paraspinous region. The flank should be percussed,
looking for areas of tenderness that may indicate a visceral
abnormality. The abdomen should be palpated for masses. Limb lengths
should be measured. Thighs and calves should be measured for any
evidence of atrophy. Straight-leg raising tests are performed looking
for signs of nerve root irritation or excessive hamstring tightness.
The sacroiliac region, particularly the sacroiliac joints, should be
examined for any signs of joint pathology. Signs of meningeal
irritation should be sought.
pain, a supine AP and lateral radiograph of the involved area of the
spine is taken to assess bony detail. Standing PA and lateral
radiographs can be ordered if the patient is being assessed for
scoliosis or kyphosis associated with the pain. If spondylolisthesis or
spondylolysis is suspected, a cone-down lateral view of the L3 to S1
region should be ordered. Oblique views may also be helpful.
warranted. Bone scanning is useful in the face of a negative radiograph
in diagnosing stress fractures, bone tumors, or infections of the
spine. CT scanning may be helpful in documenting the anatomic details
of lesions seen on plain films. For soft tissue lesions around the
spine or for evaluating the spinal cord, MRI is the diagnostic
procedure of choice.
examination, and radiographic findings, certain laboratory studies may
be helpful. A complete blood count and an erythrocyte sedimentation
rate or C-reactive protein, although nonspecific, may be abnormal in
infections, tumors, or rheumatologic conditions. If a collagen disease
is suspected, HLA-B27 and rheumatoid factors may help in the diagnosis.
covered in the remainder of this section. Other causes, such as
vertebral osteomyelitis, diskitis, and neoplastic lesions, are
discussed elsewhere in the book.
deformity in the thoracic or the thoracolumbar spine. Patients have an
increased kyphosis in the thoracic or thoracolumbar spine with
associated diagnostic radiographic changes. Normal thoracic kyphosis is
generally accepted to be between 20 and 45°. The degree of kyphosis in
the thoracic spine increases with age. Kyphosis should never be present
at the thoracolumbar junction. Any kyphotic deformity present at this
level is considered abnormal.
between 0.4 and 8%, with a slight male predominance. The diagnosis is
usually made during the adolescent growth spurt and is rarely made in
patients younger than 10 years of age. An increased incidence of
spondylolysis and spondylolisthesis is reported in patients with
Scheuermann kyphosis (this fact has been refuted by a more recent
study) as well as a 20 to 30% incidence of an associated scoliosis in
the region of the kyphosis.
theories have been advanced, including mechanical, metabolic, and
endocrinologic. There is a definite hereditary component, but no mode
of inheritance is known. Patients with Scheuermann kyphosis are
generally taller than comparably aged patients, and their skeletal age
is advanced over their chronologic age.
endplate cartilage is abnormal, with a decreased collagen-proteoglycan
ratio on electron microscopic examination. Enchondral ossification is
profoundly altered in affected segments, and proteoglycan levels are
increased. The matrix of the endplates is abnormal, thus interfering
with normal vertebral growth.
kyphosis with the apex at the T7-T9 level and kyphosis with the apex in
the lower thoracic spine at the thoracolumbar junction (T11-T12). There
is generally an associated secondary increased lumbar lordosis. The
so-called lumbar Scheuermann kyphosis has the apex at L1-L2. This
condition is generally more common in boys and in young athletes. It is
thought to have a traumatic origin.
kyphosis present with a history of deformity. The child is often
brought in by the parent because of poor posture or is referred from a
school screening program. The incidence of pain in the adolescent is
low, although about 20% of patients present with a history of
discomfort in the region of the kyphosis. In patients with lumbar
Scheuermann kyphosis, the chief complaint is generally that of pain
(80%). The pain is usually intermittent in nature. It is characterized
as dull and aching and is generally activity related and relieved by
rest.
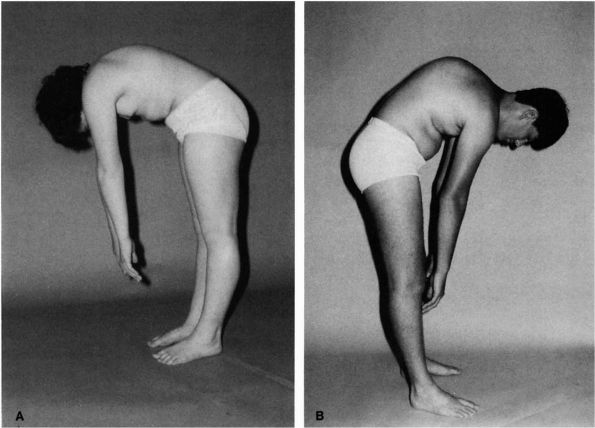
Scheuermann disease present with a kyphotic deformity. This is best
demonstrated in the forward flexed position (Figure 16-20).
The flexibility of the kyphosis can be demonstrated by having the
patient either hyperextend from a prone position or sit on a chair with
the hands held behind the head and hyperextend. Lack of flexibility
indicates the structural nature of the kyphotic deformity in contrast
to patients with flexible postural kyphosis. These patients also have a
hyperlordosis in the lumbar spine. In lower thoracic Scheuermann
disease, the kyphosis is at the thoracolumbar junction. There
may also be hypokyphosis above the thoracolumbar junction and hypolordosis in the lumbar spine.
 |
|
FIGURE 16-20. (A) Thirteen-year-old patient with normal sagittal plane spine contours on forward flexion. (B) Thirteen-year-old patient with Scheuermann kyphosis. Note the sharp angular thoracic spine kyphosis on forward flexion.
|
Because of the high association of scoliosis with Scheuermann kyphosis,
scoliosis too must be assessed. The thoracic Scheuermann patient may
have tenderness to palpation above or below the apex of the kyphosis.
In the lumbar variety, tenderness to palpation is generally in the
region of the curve apex.
examination. Although rare, with extreme degrees of kyphosis,
neurologic deficit can ensue. In addition, there is an association of
epidural cyst, causing spastic paraparesis in patients with Scheuermann
kyphosis.
Scheuermann kyphosis. Many authors think that, in the thoracic variety,
if pain is present, it subsides with growth and that there are few
adverse long-term sequelae of the condition. Others postulate that the
incidence of pain with Scheuermann kyphosis increases throughout life,
as may the deformity. The pain in adults with Scheuermann disease is
generally described as the feeling of tiredness in the back. These
patients may have pain in the hyperlordotic lumbar spine or at the apex
of the kyphosis because of ankylosis.
standing lateral radiograph of the spine. Standard radiographic
technique is important. The radiograph should be taken with the arms
parallel to the floor and resting on a support (Figure 16-21).
An alternative method is to curl the fingers and to put the pip finger
joints in the supraclavicular notch on each side. It is important to
see the entire spine to measure the thoracic kyphosis, lumbar lordosis,
and any secondary cervicothoracic curves that may accompany the
kyphosis. The kyphosis is measured by determining the angle between the
maximally tilted end vertebrae (similar to the Cobb method for
measuring scoliosis). A PA scoliosis film should be obtained to detect
the presence and magnitude of any associated scoliosis.
made by the presence of irregularities of the vertebral endplates,
anterior vertebral body wedging, Schmorl nodes, and decreased
intervertebral disc space height. In older patients, degenerative
changes may be evident. The endplate irregularity, Schmorl nodes, and
disc space narrowing are often but not always seen. There is some
discrepancy in the
literature
regarding the number of consecutive vertebrae that need to be wedged to
make the diagnosis of Scheuermann kyphosis. By one criterion (Sorenson
criterion), there should be wedging in three or more adjacent vertebrae
of more than 5°. In other studies, the diagnosis is made by the
presence of only one wedged vertebrae of more than 5°. Discrepancies
compound the problem of determining the natural history.
widening should be noted because of the association of epidural cysts
with Scheuermann kyphosis. Any scoliosis present should be assessed;
curves rarely exceed 20 to 25°. The flexibility of the kyphosis is best
demonstrated in a supine hyperextension lateral view with a “bump”
under the apex of the kyphosis.
vertebral endplates are usually present, as are Schmorl nodes. The
intervertebral disc spaces are normal, and there is no evidence of
vertebral wedging.
differential diagnosis includes postural round back. In postural round
back, there is a slight increase in the thoracic kyphosis. The
kyphosis, however, is flexible, as demonstrated on the prone or sitting
hyperextension tests. On the standing lateral radiograph, there are no
structural changes as noted for Scheuermann kyphosis. The kyphosis in
postural kyphosis patients is usually in the range of 45 to 60°. On the
supine hyperextension lateral view, the deformity is totally flexible.
The question remains whether a postural kyphosis left untreated may
progress and get secondary bony changes resembling Scheuermann disease.
Postural kyphosis, if flexible, should be treated by exercising.
various types of skeletal dysplasia, such as spondyloepiphyseal
dysplasia congenita and Morquio disease. These conditions can usually
be diagnosed by the clinical examination and other radiographic
features. Ankylosing spondylitis may present a similar picture, but 97%
of these patients are HLA-B27-positive. Kyphosis may also be present in
patients who had a laminectomy before skeletal maturity and in patients
who had radiation to the spine for a regional tumor, such as Wilms,
tumor or neuroblastoma. Kyphosis may also been seen with eosinophilic
granuloma. Type II congenital kyphosis (failure of segmentation) may be
confused with Scheuermann disease. It may be necessary to use CT
scanning to identify the anterior failure of segmentation seen in this
condition to differentiate it from Scheuermann kyphosis.
Lumbar Scheuermann disease generally responds well to nonoperative
measures, such as nonsteroidal anti-inflammatory agents and temporary
activity restriction. There are no adverse long-term sequelae from
lumbar Scheuermann disease.
Scheuermann kyphosis is benign and therefore needs no treatment. Others
report increasing pain with progression of the deformity. It is
uncertain whether treatment prevents any of the consequences that may
occur without treatment. Treatment of Scheuermann kyphosis in the
skeletally immature patient is recommended in the hope of preventing
excessive deformity that may cause pain and cosmetic concerns.
Exercises alone are not beneficial. Hyperextension body casts changed
at monthly intervals to correct the curvature may be used in the
skeletally immature patient with a rigid Scheuermann kyphosis (i.e.,
less than 10 or 15° of correction on hyperextension lateral radiograph)
followed by bracing; this technique is rarely used today. In those
patients with a somewhat flexible Scheuermann kyphosis, the Milwaukee
brace is prescribed, although some orthopaedists prefer to use a
hyperextension underarm orthosis to hopefully improve brace wear
compliance. In some centers, particularly in Europe, casting alone is
used as a treatment for this condition. Treatment is generally
continued until the patient reaches skeletal maturity. In immature
patients, some of the anterior wedging associated with Scheuermann
kyphosis may be corrected by treatment. Follow-up studies of patients
treated for Scheuermann kyphosis demonstrate increase of the kyphosis
over time even after brace treatment.
kyphosis. In patients with curves greater than 75° and with pain
unresponsive to nonoperative measures, spinal fusion can be considered.
Treatment of kyphosis of this magnitude requires anterior and posterior
spinal fusion throughout the length of the kyphosis. Cord decompression
is indicated for the rare patient who has neurologic deficits secondary
to epidural cysts or increased kyphotic angulation.
descriptive term referring to a defect in the pars interarticularis.
The defect may be unilateral or bilateral and may be associated with
spondylolisthesis. Spondylolisthesis refers to the anterior displacement (translation) of a vertebra with
respect to the vertebra caudal to it. This translation may also be
accompanied by an angular deformity (kyphosis). These two topics are
considered together in that the most common cause of spondylolisthesis
in children is spondylolysis.
 |
|
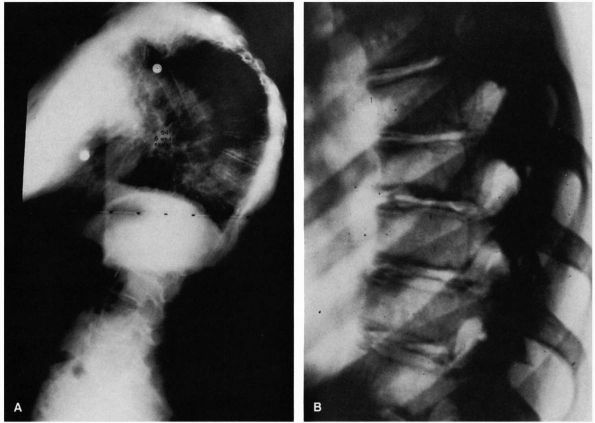
FIGURE 16-21. (A) Standing lateral radiograph of patient with Scheuermann disease (94°). Note the marked vertebral wedging at the curve apex. (B)
Coned-down views of spine in another patient with Scheuermann disease. Note the vertebral wedging, endplate irregularity, and disc space narrowing. |
and less frequently at the L4-L5 region. Spondylolytic lesions may
occur at other lumbar levels or at multiple levels. Spondylolytic
lesions are found in about 5% of the general population. Spondylolysis
is an acquired condition. It has not been reported in infants and is
rarely present before 5 years of age. There is an increased incidence
of spondylolysis and spondylolisthesis up to the age of 20 years, after
which the incidence remains stable.
|
TABLE 16-5. Classification of Spondylolisthesis
|
|||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||
It is further classified by the degree of angular and translational
displacement. The diagnosis and treatment of the condition depends on
the type.
secondary to a stress fracture at the pars interarticularis.
Experimental studies showed that extension movements of the spine,
particularly in combination with lateral flexion, increase the shear
stress at the pars interarticularis. Clinical evidence for this theory
includes the high association (four times more than normal) in female
gymnasts, football linemen, and soldiers carrying backpacks. This
etiologic theory is also supported by a reported higher association in
patients with Scheuermann kyphosis with secondary excessive lumbar
lordosis. In contrast, spondylolysis has never been seen in patients
who have never walked.
spondylolisthesis may be inherited conditions. There is a high
association of the condition in family members of affected patients.
There are racial and gender differences, with the lowest incidence in
black females and the highest incidence in white males. Most patients
with type I spondylolisthesis have abnormalities at the lumbosacral
junction with poor development of the superior aspect of the sacrum and
superior sacral facets and with associated sacral spina bifida. Similar
congenital changes have also been reported in about one-third of
patients with type II spondylolisthesis. Thus, these conditions may be
genetic, acquired, or both.
and spondylolisthesis are determined primarily by the age of the
patient and, in spondylolisthesis, by the type. Although pain is the
most common presenting complaint in the adult, it is relatively
uncommon in children or the symptomatology is usually mild. Children
most commonly present with gait abnormalities, postural deformity, and
hamstring tightness. Back pain is usually localized to the lower-back
region, with occasional radiation to the buttocks and the thighs.
Occasional L5 radiculopathies are present, although this is not common
in children.
spondylolisthesis generally is older than 40 years of age, and women
are more commonly affected than men. Pain in degenerative
spondylolisthesis is often similar to the pain patterns in patients
with a herniated nucleus pulposus (i.e., the patient has pain radiating
down the leg and complaints of sciatica). Patients may complain of pain
similar to spinal stenosis and have claudication-type symptoms (i.e.,
pain and cramping in the calves and back brought on by walking and
relieved by sitting in a flexed spinal posture). In most cases of
spondylolysis and spondylolisthesis, pain is precipitated by activity,
especially flexion and extension on a repetitive basis, and relieved by
rest or lowered activity levels.
including detailed neurologic examination. About 80% of children with
spondylolysis and accompanying spondylolisthesis have evidence of
hamstring tightness. The cause for this is unknown but is thought to be
instability in the area of the spondylolysis and spondylolisthesis
resulting in cauda equina irritation. Hamstring tightness is
responsible for the postural abnormalities often seen as the presenting
complaint of the patients with spondylolisthesis. Restrictive flexion
secondary to the hamstring tightness and the pelvic tilt gives the
patient a stiff-legged gait with a short stride length. The pelvis
rotates as the child takes a step, and often the child walks on tiptoes
with the knees slightly flexed. The hamstring tightness may be so
severe in some children that, in performing a straight-leg raising test
in a supine position, the leg can only be lifted several inches off the
table.
the type and degree of the slip. Patients may present with mild
tenderness to palpation in the area of the spondylolysis or
spondylolisthesis. In severe grades of slip, a “step-off” may be
palpated. There may be an apparent increase in lumbar lordosis with a
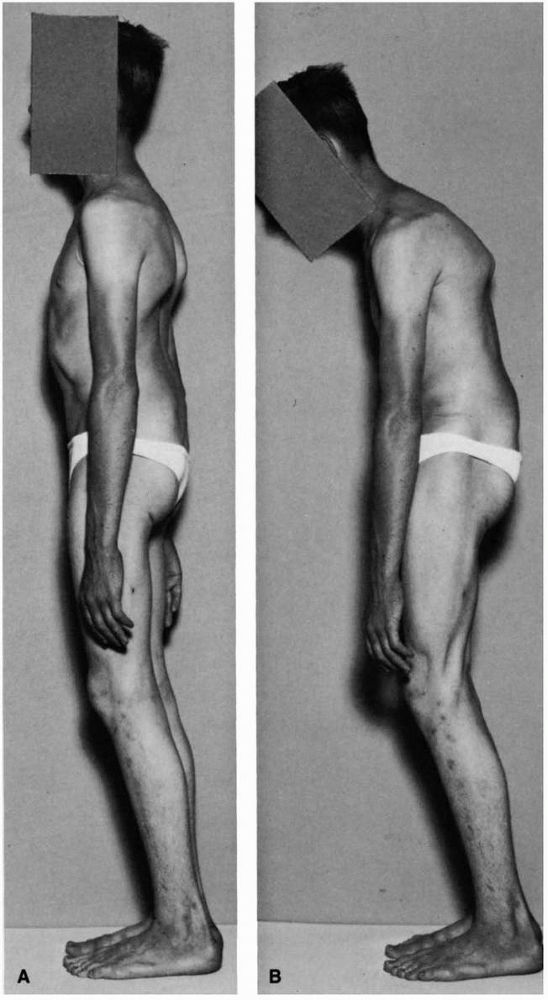
backward tilting of the pelvis (Figure 16-22).
The patient may present with protrusion of the lower abdomen, and in
severe cases of spondylolisthesis, a deep transverse abdominal crease
may be noted. A detailed neurologic examination, including deep tendon
reflexes, sensory examination, and motor strength, should be performed
on each patient with particular attention to any dysesthesia near the
sacrum and rectum. A history of bowel or bladder dysfunction may be
indicative of cauda equina syndrome.
spondylolisthesis have evidence of scoliosis. The scoliosis most
commonly seen in association with symptomatic spondylolisthesis is
generally not structural. It is more commonly seen in patients with
high-degree slips. The curve is usually in the lumbar region and
resolves with the resolution of the symptoms of the spondylolisthesis.
Some patients have a characteristic idiopathic scoliosis that is
unaffected by the spondylolisthesis or its treatment.
may occur any time after the pars fractures. Most slippage occurs
during the adolescent growth spurt. Rarely are significant increases
in the degree of spondylolisthesis seen after skeletal maturity.
 |
|
FIGURE 16-22. High grade spondylolisthesis, physical findings. (A)
Note the flattening of the buttock, anterior protrusion of the pelvis, visible lumbar slip-off, and apparent shortening of the trunk. (B) Displaying characteristic hamstring tightness, limiting his ability to touch his toes without flexing the knees. (Turner RH, Bianco AJ. Spondylolysis and spondylolisthesis in children and teenagers. J Bone Joint Surg 1971;53A:1298) |
suspected, standing PA and standing lateral radiographs of the spine
with a cone-down lateral view of L3 to the sacrum are indicated. In
most cases, the pars interarticular defect can be seen on the spot
lateral views. The defect is usually at the L5 to S1 level. Defects at
the L4 level are more common in patients who have complete or partial
sacralization of the L5 vertebra. If the defect is not visualized on
the lateral film and the condition is suspected, an oblique view may be
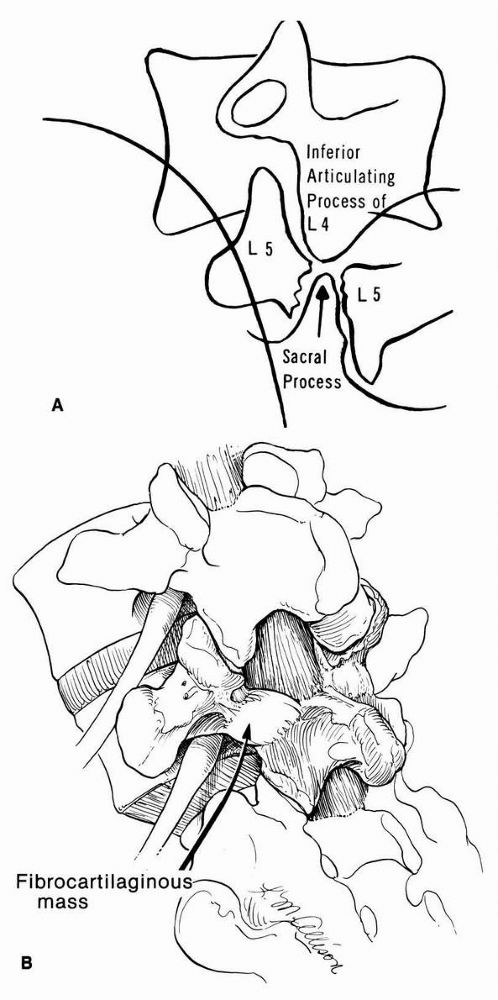
helpful. On this view, one can see what has been described as a Scotty
dog with a broken neck or wearing a collar (Figure 16-23).
In about 20% of patients, the lytic defect is unilateral and may be
accompanied by reactive sclerosis in the opposite pedicle, lamina, or
both. This situation often presents a difficult diagnostic dilemma in
that the sclerotic region can be confused with lesions
such
as osteoid osteoma and osteoblastoma. If the lesion is not visualized
on plain radiograph, technetium bone scanning, Spect scan, or CT scan
may be helpful in identifying the lesion. In an acute injury, a “hot”
bone scan may allow for early detection. Bone scanning is also used to
assess whether an established lesion has the potential to heal. If the
lesion is “cold,” there is an established nonunion, and hence
immobilization would probably not result in healing of the stress
fracture.
 |
|
FIGURE 16-23. (A)
Diagram of oblique radiograph of lumbosacral junction, showing cleft in isthmus of the fifth lumbar vertebra. The articular process of the sacrum projects upward and penetrates the cleft, meeting the inferior articular process of the fourth lumbar vertebra. (B) Pathology of spondylolisthesis showing the relation of the nerve root as it courses through the intervertebral foramen. The continuity of the pars interarticularis is bridged at the defect by a fibrous or fibrocartilaginous mass that rarely may encroach on the nerve root of L5. |
 |
|
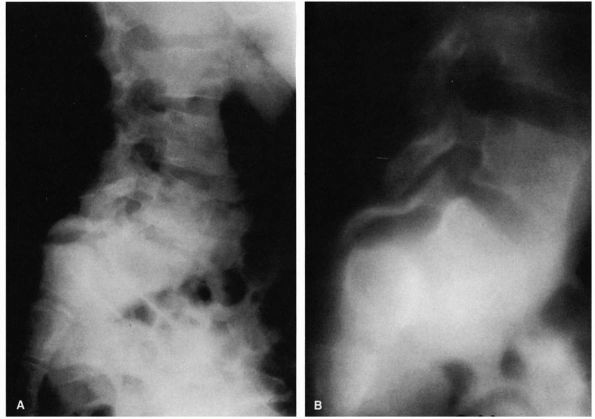
FIGURE 16-24. (A)
Nineyear-old girl with type I spondylolisthesis. Note how pars interarticularis has become attenuated, allowing for severe slippage (translation and angulation). The entire posterior arch has slipped forward. (B) Polytome demonstrating the elongation of pars interarticularis. |
There is dysplasia of the superior articular facets of the sacrum and
inferior articular facets of L5. Type I slips are generally limited to
25 to 30% slippage unless the pars becomes attenuated or fractures,
allowing for severe degrees of slippage to occur.
In the adult disc space, narrowing and degenerative changes at the
intervertebral disc and posterior elements should be noted. Patients
with a more rounded S1 and a more wedged-shaped L5 have a greater risk
of progression.
depending on the percentage of anterior translation of L5 on S1, with
grade 1 being a 25% slip; grade 2, a 50% slip; grade 3, a 75% slip; and
grade 4, a complete slip (Figures 16-28 and 16-29). The term spondyloptosis
is used to describe complete displacement of L5 in front of S1. It is
important in assessing spondylolisthetic patients to have standing
lateral radiographs of the lumbosacral junction because instability is
not uncommon, particularly in childhood (Figure 16-28).
Several standard methods of measurements are used to quantitate
spondylolisthesis; these include the percentage of translation,
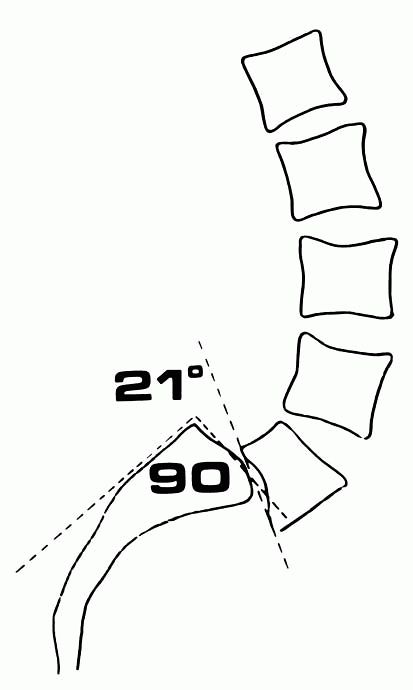
sagittal roll, and slip angle (Figures 16-30 and 16-31). These measurements of angulation or lumbosacral kyphosis are important prognostic indicators.
especially in the adult patient with degenerative spondylolisthesis.
These studies include flexion-extension lateral views to detect
instability and CT scanning with or without myelographic enhancement to
assess the integrity of the disc and to look for other potential
sources of the discomfort. They are also useful to ascertain the
specific pathology in spondylolisthesis associated with or causing
spinal stenosis and to rule out intraspinal pathology in patients who
do not have resolution of symptoms by nonoperative measures.
Myelography is rarely indicated in the child or adolescent with
spondylolisthesis; MRI may be used if the patient has signs or symptoms
of nerve root compression or cauda equina syndrome. In the adult
patient, other diagnostic tests, such as EMG, motor nerve condition
studies, and psychological testing, may be considered.
 |
|
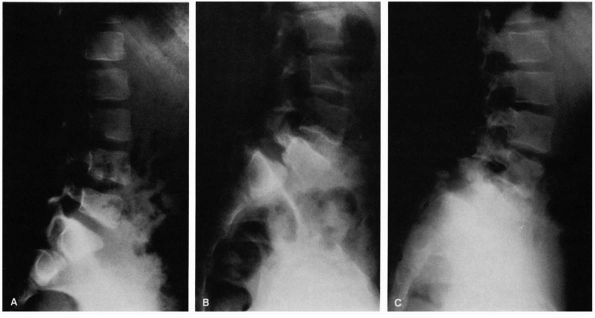
FIGURE 16-25. (A) Six-year-old female gymnast who complained of mild backache. Narrowing of pars interarticularis noted in radiograph. (B)
At age 11, she had increasing pain. Radiograph demonstrates a lytic defect (type IIA) of the pars interarticularis and significant translation; surgery was recommended but refused. (C) Because of increasing pain, surgery was performed at age 12. The preoperative radiograph demonstrates increasing anterior translation and lumbosacral angulation (kyphosis). |
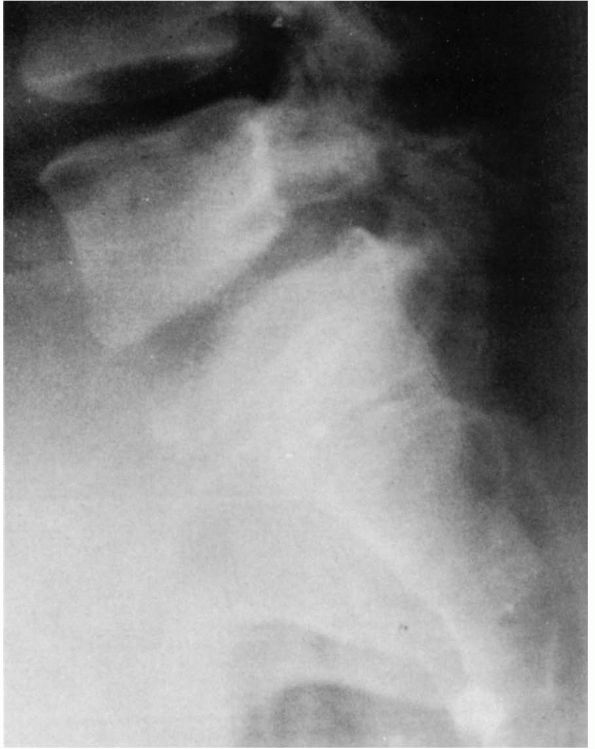 |
|
FIGURE 16-26. Spondylolisthesis. Note the attempt at formation of a supporting ledge at the anterior edge of the sacrum.
|
 |
|
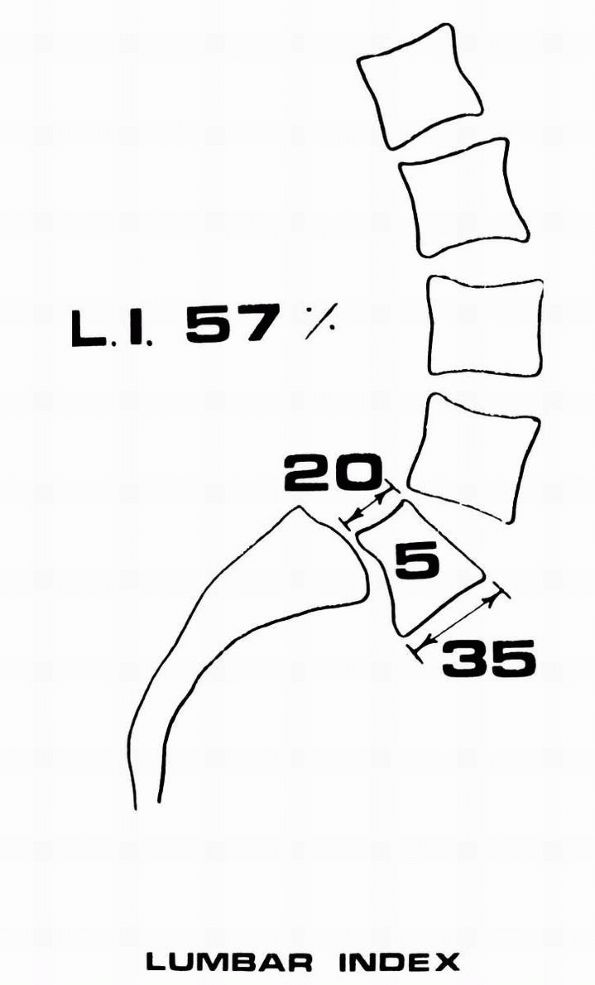
FIGURE 16-27.
The lumbar index represents the degree of trapezoidal deformation of the fifth lumbar vertebral body. Although a decreased lumbar index is secondary to increased slipping, when considered in conjunction with other factors, such as the adolescent growth spurt, a domeshaped first sacral vertebra, and female gender, it indicates that the patient is at risk for progression of slipping. (Boxall D, Bradford DS, Winter RB et al. Management of severe spondylolisthesis in children and adolescents. J Bone Joint Surg 1979;61A:479) |
 |
|
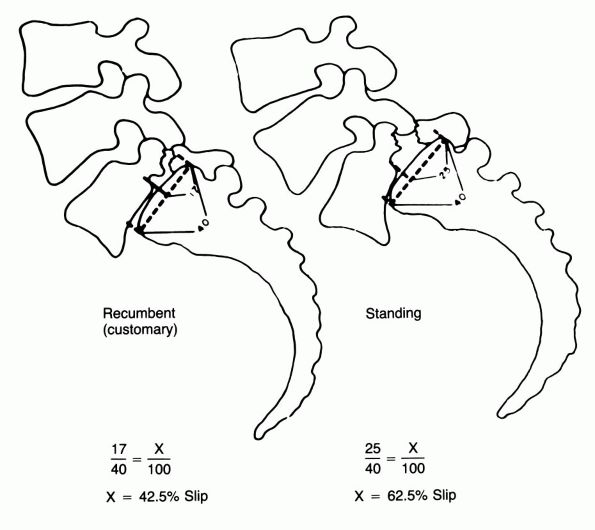
FIGURE 16-28.
Spondylolisthesis, demonstrating the accuracy of the standing radiograph. The displacement is measured as a percentage of the width of the adjacent vertebral body. The standing and recumbent views are compared. (Lowe RW, Hayes TD, Kaye J, et al. Standing roentgenograms in spondylolisthesis. Clin Orthop 1976;117:80) |
 |
|
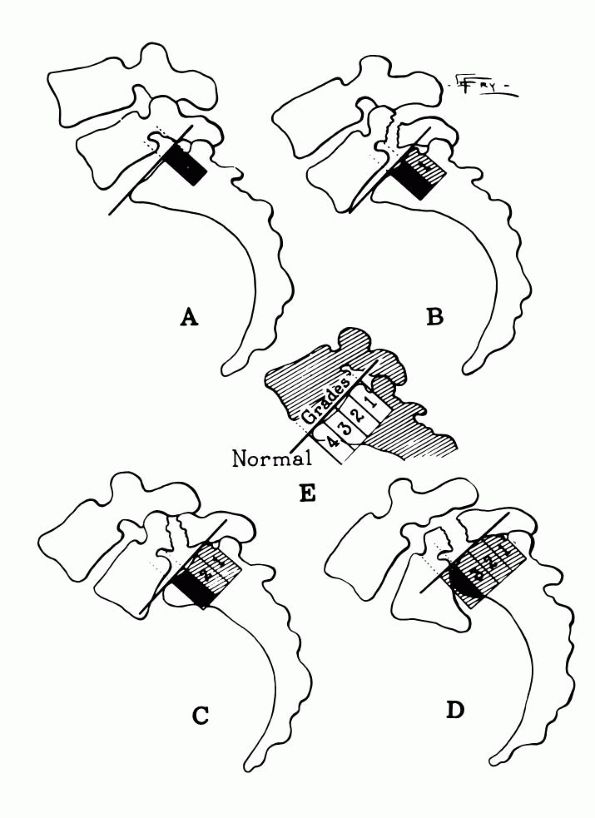
FIGURE 16-29.
Meyerding grading system for spondylolisthesis, demonstrating the degrees of slipping of the fifth lumbar vertebra on the sacrum. (Meyerding HW. Spondylolisthesis. Surg Gynecol Obstet 1932;54:374) |
 |
|
FIGURE 16-30.
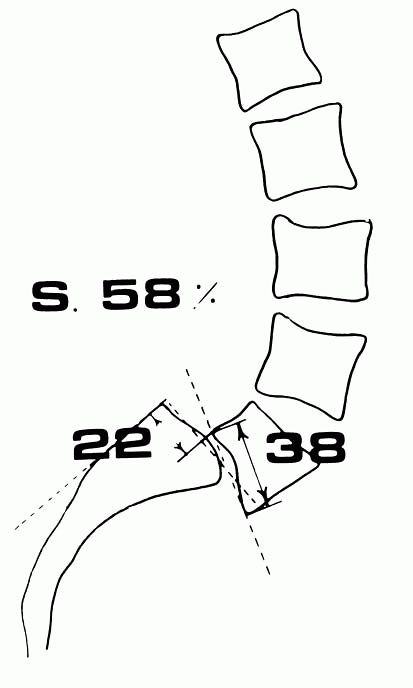
Percentage of slipping. A line is extended upward from the posterior surface of the first sacral vertebral body, and a second line is drawn downward from the posterior surface of the fifth lumbar vertebral body. The extent of slip is the distance between these two lines. This measurement is expressed as a percentage of the AP dimension of the fifth lumbar vertebral body. (Boxall D, Bradford DS, Winter RB et al. Management of severe spondylolisthesis in children and adolescents. J Bone Joint Surg 1979;61A:479) |
successfully without surgery. If the diagnosis is made as an incidental
finding, no activity restrictions are necessary. The patient should,
however, be followed through skeletal maturity with standing spot
lateral radiographs of the lumbosacral spine every 6 to 8 months to
watch for the development of spondylolisthesis.
symptoms, bone scanning is helpful to assess the lesion and to follow
healing. In such patients, particularly athletes, some evidence
suggests that immobilization in a cast or brace allows for healing of
the lesion. There are some differences of opinion about whether the
cast should extend from the nipple line to include one or both legs or
if the same results may be achieved in a TLSO. If this treatment is
attempted, the patient can be followed by serial radiographs and bone
scans to assess healing.
treated by activity restriction and exercises, including hamstring
stretching. If symptoms are more severe, a short period of bed rest may
be tried before immobilization in a TLSO. Exercises should be
prescribed. Nonsteroidal anti-inflammatory medications are a useful
adjuvant to treatment.
 |
|
FIGURE 16-31.
The slip angle measures the degree of forward tilting of the fifth lumbar vertebral body over the first sacral vertebral body, or the kyphosis at the level of slipping. The angle is formed by a line drawn perpendicular to the posterior aspect of the first sacral body and one drawn parallel to the inferior aspect of the fifth lumbar vertebral body. It represents the degree of instability and potential for progression, particularly when the slip angle is high and a significant increase is noted in the standing roentgenogram as compared with the supine roentgenogram. (Boxall D, Bradford DS, Winter RB et al. Management of severe spondylolisthesis in children and adolescents. J Bone Joint Surg 1979;61A:479) |
management, other conditions, including neurologic conditions and
tumors of the spine and spinal cord, must be ruled out before
instituting surgical treatment. In patients whose symptoms do not
respond to nonoperative treatment, lumbosacral intertransverse process
fusion of L5 to S1 has a 90% chance of obtaining a solid fusion with
relief of symptomatology, including resolution of hamstring tightness.
In patients with isolated or multiple defects in the L1 to L4 region,
surgical repair of the defect by one of many available techniques is
often recommended to allow for sparing of lumbar motion segments.
less than a 25% slip, no treatment is indicated. Natural history
indicates that the likelihood of having future problems is essentially
the same as
that
of the general population. With slips greater than 25%, there is an
increased likelihood of the patient having lower-back symptoms compared
with the general population. Certain clinical and radiographic risk
factors have been determined to be associated with future pain,
progressive deformity and increasing degree of spondylolisthesis (Table 16-6).
slip, nonoperative measures should be tried in attempt to control
symptomatology. These measures are similar to those described
previously for the treatment of spondylolysis. About two-thirds of
these patients have resolution of symptomatology with nonoperative
treatment.
patient are failure of relief of symptoms by nonoperative measures or a
slip of greater than 50%. L4-sacrum intertransverse process fusion is
recommended in those patients with a greater than 50% slip, and
L5-sacrum fusion is recommended for those patients with a less than 50%
slip. By this method, 90% of children can expect to have a solid fusion
within 1 year of surgery and gain resolution of any mild neurologic
symptoms, including hamstring tightness, over the ensuing 12 to 18
months. Nonunions and curve progression despite a solid fusion,
however, have been reported in children undergoing in situ fusion.
Because of these problems, particularly with high grade slips, many
authors advocate fusion and closed reduction, followed by pantaloon
casting for about 3 months to decrease the sagittal rotation and
improve the cosmetic deformity. Although sagittal role or slip angle
can be changed by closed methods, vertebral translation is generally
unaltered or is changed little by these techniques. The use of
instrumentation accompanying fusion with or without reduction is being
done more commonly to increase the rates of union and decrease the
prolonged immobilization necessary with other techniques. Decompression
is rarely necessary in childhood spondylolisthesis and must be
accompanied by a fusion to prevent further progression.
|
TABLE 16-6. Risk Factors in Spondylolisthesis for Pain, Progression, and Deformity
|
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
spondylolisthesis, careful assessment must be done to rule out other
associated conditions (e.g., herniated nucleus pulposus) and to
evaluate disc degeneration and nerve compression. In the adult, the
symptomatology is generally confined to back pain. In situ fusion may
be all that is necessary. If the patient has leg pain, however,
decompression and fusion may be warranted. In the adult patient with
spondylolisthesis with or without radiculopathy, it is incumbent on the
surgeon to obtain a solid fusion. Many surgeons advocate internal
fixation along with a spinal fusion. Internal fixation is most commonly
attained by means of pedicle screws, plates, or rods. In the adult,
because the risk of further displacement is minimal, pantaloon cast
immobilization as advocated by some in children as unnecessary.
III), the source of the pain must be sought before recommending
surgical treatment. These patients rarely develop spondylolisthesis
greater than 25%. Radiographs show evidence of degenerative disc
disease as well as degeneration of the facet joints. Some patients
develop retrolisthesis, and others may develop intraspinal synovial
cysts. A complete diagnostic evaluation must be done in these patients,
including diskography to ascertain the source of the pain. Adult
patients require a much more extensive workup, including the
possibility of EMG, motor nerve conduction studies,
psychological
testing, diskography, epidural steroid injections, or nerve root
blocks, to ascertain the cause of pain and prognosticate the
effectiveness of treatment. Most patients with degenerative
spondylolisthesis can also be treated nonoperatively. In those who fail
conservative treatment, exhaustive diagnostic measures must be
undertaken to determine the source of pain.
less than 3% occur in children. An estimated 30 to 60% of cases
reported have an associated history of trauma. There is a male
predominance. Herniated nucleus pulposus in children is associated with
additional vertebral disease, including sacralized L5, lumbarized S1
(either complete or incomplete), asymmetric articular facets, and spina
bifida.
may be minimal or they may be characteristic of the adult condition. In
children, however, there is occasionally an associated scoliosis
secondary to muscle spasm. The neurologic symptoms in children are less
common and less severe. Herniated nucleus pulposus may also occur in
association with a slipped vertebral apophysis or fracture of the
vertebral apophysis. The most common levels involved in children are L4
to L5 and L5 to S1. Treatment recommendations are the same as those in
the adult.
costly medical problems. About 80 to 90% of the adult population
suffers from back pain during their life. Most cases of back pain
resolve spontaneously. Half of patients who complain of back pain are
generally asymptomatic within 2 weeks, and 90% are asymptomatic after 3
months. It is estimated that 1 year after symptom onset, only 2% of all
adults with back pain have persistent pain.
patients under 45 years of age. Although back pain is a common
complaint, in 80 to 90% of cases, a pathologic cause cannot be
determined. Epidemiologic studies determined that risk factors related
to the development of back pain include job dissatisfaction, repetitive
lifting, low-frequency vibration, low educational level, smoking, and
social problems.
history, careful physical examination of the musculoskeletal system,
and complete neurologic examination generally allow the physician to
make the diagnosis. Diagnostic tests are only used to confirm the
suspected diagnosis. Most causes of back pain are self-limiting. Over
half of patients with back pain recover within 7 days. Patients who
have back pain-associated sciatica (pain down the distribution of the
nerve roots contributing to the sciatic nerve) generally recover within
4 weeks.
assessed in terms of whether it is acute or chronic and whether it had
an insidious onset or whether it can be related to a traumatic event.
Risk factors should be sought. Pain should be characterized regarding
location and whether it is confined to the back or the leg. It is
important to determine the pattern of the pain. In patients with
sciatica, the pain (and sometimes numbness or tingling)
characteristically radiates down the distribution of one of the nerve
roots that contributes to the sciatic nerve, characteristically L4, L5,
or S1. If the pain does not radiate below the knee but is localized
primarily to the back, buttock, hip, and distal thigh, it is likely to
be referred pain rather than pain caused by compressive irritation of
one of the nerve roots of the sciatic nerve.
and activity on the pain. The patient should also be queried about
muscle weakness or numbness and its location. Medications taken by the
patient and their effect on pain should be noted. The effect of
previous treatments should also be assessed. A necessary line of
questions includes whether or not there is workman’s compensation or
litigation involved. Job description and satisfaction should also be
assessed. The patient must also be questioned about the symptoms impact
activities of daily living. Swelling, erythema, and pain in other
joints should be noted because they may be indicative of a generalized
condition, such as metabolic bone disease or rheumatoid spondylitis.
incontinence or retention or change in pattern of stream, which may
indicate bowel or bladder dysfunction. Complaints of nonanatomic
sensory loss (e.g., stocking-glove anesthesia or nonspecific motor loss
throughout the entire lower extremities) should alert the physician to
the possibility of a neuropathy or a nonorganic cause of the symptoms.
Psychosocial factors can contribute to back pain or response to
treatment in patients with chronic back
pain.
It is important to use additional studies, including psychological
inventories such as the Minnesota Multiphasic Personality Inventory, to
assess these patients.
patient’s symptoms can be ruled in or out by the history alone. Careful
physical examination, however, must be done. The patient should be
evaluated in a standing position to assess the presence or absence of
the normal sagittal and coronal plane contours. The spine should be
assessed in flexion, extension, and lateral bending. Restriction of
motion or abnormality in any of these motions should be noted. The
effect of various motions on the patient’s back or leg pain should be
noted. The spine should be examined in the prone position. The entire
spine should be palpated, assessing both the soft tissues and bone
elements for tenderness. The effect of palpation on the patient’s
symptomatology should also be noted. The sacroiliac joint should be
examined, as should the hips. Pathology in these areas may be confused
with pain of spinal origin. Each dermatomic region must be assessed for
all sensory modalities (Figure 16-32). Motor function in each muscle group must be graded. Reflexes should be tested and any asymmetry noted (Table 16-7).
A positive Babinski sign may indicate an upper motor neuron lesion.
Assessment of rectal tone and perianal sensation is imperative in any
patient complaining of back pain. The straight-leg raising test should
be performed both in the supine and prone (femoral stretch) positions.
In the supine position, the test is performed by passively elevating
the patient’s leg with the knee extended and pelvis stable. The test is
positive if this maneuver reproduces sciatic pain (radiating below the
knee). If the patient only complains of back pain or tightness of the
hamstring muscles behind the thigh, the test is considered negative.
 |
|
FIGURE 16-32. (A) The usual relation of the protruded disc at the fourth lumbar interspace. The fifth lumbar nerve root is compressed. (B) An uncommon relation of the protruded disc at the fourth lumbar interspace. The fourth lumbar nerve root is compressed.
|
|
TABLE 16-7. Reflexes
|
|||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
tension signs indicative of neural irritability. This specific test
should be done on both legs. If the patient’s asymptomatic straight-leg
raising test elicits sciatic pain down the contralateral leg, this is
called a positive contralateral straight-leg raising test and indicates
nerve root irritation.
straight-leg raise performed with the knee flexed. With the leg held in
this position, the popliteal fossa is palpated. A positive bow-string
test occurs
if,
during the compression of the tibial nerve in the popliteal fossa, the
patient complains of radiating pain both proximally and distally or of
paraesthesia in the distribution of any particular branch of the
sciatic nerve.
test) is performed with the patient in the prone position. The leg is
extended from the hip, stretching the femoral nerve. Any pain along the
distribution of the femoral nerve is considered a positive stretch test.
Patients with spinal stenosis have symptoms of claudication. The
presence of palpable pulses in the extremities is helpful in ruling out
a vascular cause of the claudication symptoms.
for inconsistencies. The Wadell tests for nonorganic causes of
lower-back pain are useful in evaluating patients with chronic backache
(Table 16-8).
further diagnostic workup depends on whether the patient has a history
or physical examination compatible with lumbar radiculopathy, keeping
in mind the natural history. The remainder of the diagnostic tests and
treatment alternatives depend on whether the patient has acute or
chronic back pain and whether there is an associated radiculopathy.
|
TABLE 16-8. Nonorganic Physical Signs in Lower-Back Pain
|
|||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||
patients younger than 40 years of age is a herniated nucleus pulposus.
The nucleus pulposus may bulge into the canal. Tears of the fibers of
the annulus fibrosis may allow the disc to extrude through the annulus,
or the disc may sequester through the annulus and lie free in the
spinal canal or neural foramina. Nerve root compression may cause
secondary inflammation of the nerve root, giving the patient subjective
symptoms of pain, numbness, or tingling along the distribution of the
particular nerve root. The most commonly affected nerve roots are L5
and S1. In the lumbar spine, the exiting nerve root is named for the
vertebra about which it exits. Therefore, the L5 nerve root exits
before the L5 to S1 disc space below the L5 pedicle. Thus, L5 to S1
disc herniation usually causes irritation of the S1 nerve root (Figure 16-33).
Herniations occur less frequently at higher levels. Pain associated
with a herniated nucleus pulposus varies from mild pain along the
distribution of the nerve to severe incapacitating pain.
 |
|
FIGURE 16-33.
Distribution of spinal dermatomes. Considerable overlap occurs; consequently, involvement of a single spinal segment may not be evident. |
nucleus pulposus generally reveals restricted range of motion with
forward flexion increasing the pain. The patient may list to one side.
Sensory and motor examination may show evidence of nerve root
compression with decrease in sensation, muscle weakness, or both and
diminished reflexes in the region of the affected nerve (see Table 16-7 and Figure 16-32).
Circumference of extremities should be measured to detect any evidence
of atrophy. Acute nerve root irritation is evidenced by positive
tension signs and straight-leg raising, contralateral straight-leg
raising, and bow-string stretch tests. A contralateral straight-leg
raising test is the most specific sign of herniated nucleus pulposus.
about half resolve within 4 weeks. Diagnostic studies thus are not
indicated initially. The patient should be treated with a short period
of bed rest (1 to 2 days), limitation or modification of activities,
and in some cases, anti-inflammatory drugs (aspirin or nonsteroidal
anti-inflammatory agents). Epidural steroid injections may provide
short-term relief for patients with a herniated nucleus pulposus.
Treatment modalities advocated in the past (traction, spinal
manipulation, corsets or braces, and physical therapy) have little
scientific validity. As the adult symptoms resolve, it is important to
get the patient on a rehabilitation program to prevent recurrent
episodes of back pain and disability. Patients should be encouraged to
increase their activity level and begin a conditioning and physical
fitness program.
pulposus to cause bilateral sciatica, the presence of bilateral
lower-extremity neurologic signs and symptoms should alert the
physician to the possibility of a central disc herniation or, rarely,
intraspinal
pathology other than disc herniation. The presence of back pain,
sciatica, and bowel or bladder dysfunction associated with motor
weakness is referred to as cauda equina syndrome
and is generally caused by extrinsic compression of the thecal sac in
the area of the cauda equina. Cauda equina syndrome often requires
immediate surgical intervention. Only patients with acute cauda equina
syndrome should undergo immediate diagnostic evaluation. These patients
should have immediate MRI, myelography, or both to determine the cause
of the cauda equina syndrome before surgical intervention.
nonoperative treatment or progressive neurologic deficit, further
diagnostic evaluation is often necessary. Plain lumbosacral radiographs
rarely provide diagnostic information. With aging, normal degenerative
changes occur in the lumbar spine that often confuse the diagnostic
picture. Changes of spine degeneration are seen in as many of 70% of
radiographs. These radiographic changes have little effect on
management outcomes. Plain radiographs, however, may be taken to rule
out other pathologic conditions, such as infection or tumor.
nucleus pulposus is MRI. MRI is both sensitive and specific. CT
scanning can act as an alternative if MRI is not available. Patients
who have CT scans should have a myelogram to assess the thecal sac both
proximal and distal to the suspected level so that abnormalities
simulating radiculopathies are not missed. If radiographic studies
correlate with the patient history and physical findings, treatment
outcomes are generally favorable. If the patient’s symptomatology
persists despite nonoperative measures, then operative intervention can
be considered. Discrepancies between the radiographic findings and the
patient’s clinical picture require further investigation.
evidence to suggest that surgical intervention offers any improvement
in weakness or sensory deficit over continued nonoperative treatment.
In the short term, however, most surgically treated patients with acute
lumbar radiculopathy are pain free in a relatively shorter time.
Limited surgical disc excision has a success rate of 90% in patients
who have good correlation between history, physical examination, and
diagnostic studies. Laminectomy is often the procedure of choice when
surgery is indicated. If half or more of both or all of one facet joint
is injured causing secondary instability, fusion across the segment may
be necessary. In the absence of facet joint injury, fusion in
conjunction with diskectomy is unnecessary.
diskectomy, and microsurgical diskectomy have been advocated to
decrease hospitalization, minimize perineural scarring associated with
laminectomy, and speed rehabilitation. Chemonucleolysis has the
potential complications of anaphylactic shock and neurologic deficit
secondary to acute transverse myelitis. It is contraindicated in
patients who previously underwent operative treatment or have evidence
of spinal stenosis. Transverse myelitis has been reported in patients
undergoing chemonucleolysis using chymopapain and having concomitant
diskography. Percutaneous diskectomy may not be used in patients with
sequestered fragments or in the presence of spinal stenosis. With
microdiskectomy, sequestered fragments can be missed, and the lateral
recess may not be adequately decompressed.
with or without radicular pain is much more difficult than that of the
patient with acute pain. Chronic back pain is usually defined as back
pain present for at least 6 months and not responsive to nonoperative
interventions. This is the most difficult group of patients facing the
clinician. As with the patient with acute onset of back pain, the
chronic pain must be characterized regarding onset location, radiation,
pattern, effect of positions, associated weakness, bowel or bladder
symptomatology, and effect of medications. Most important, however, a
psychosocial history needs to be taken. How the pain affects the
patient’s lifestyle is important; any litigation or workman’s
compensation involved must be noted. Previous treatment modalities and
their effects must be carefully documented because many patients with
chronic lower-back pain previously had either surgical or nonoperative
treatment. Each patient must have a complete and thorough physical
examination, including neurologic examination. It is important to test
the patient for nonorganic physical signs. Many patients have been seen
by multiple physicians. Psychosocial evaluation of these patients is
often necessary.
assessment and treatment of patients with chronic back disorders use
the multidisciplinary
approach.
These clinics employ physicians, surgeons, psychologists, social
workers, occupational therapists, vocational rehabilitation counselors,
and others to evaluate these difficult and often complex patients.
plain films are not often helpful. Degenerative changes may be seen on
plain radiographs in a high percentage of normal patients. This is
secondary to the normal aging process. Spina bifida occulta, Schmorl
nodes, vacuum discs, mild scoliosis, transitional vertebrae
(sacralization of L5 or lumbarization of S1, partially or completely)
occur as frequently in asymptomatic patients as in patients with
lowerback pain.
metabolic bone disease, tumor, fracture, or traumatic injury of the
spine is suspected. As mentioned in the section on spondylolisthesis,
degenerative spondylolisthesis may be evident on plain radiographs
secondary to facet joint degeneration with subsequent subluxation.
Subluxation may occur in a posterior direction (retrolisthesis). These
conditions in some cases cause nerve root compression and radicular
symptoms. Inflammatory disease may also be detected on plain
radiographs. If the patient has any other joint complaints in
association with spinal pain, radiographs of the sacroiliac joint may
help to make the diagnosis of ankylosing spondylitis.
determining the presence of recurrent disc herniation. MRI is
especially helpful in patients who have had previous lumbar spine
surgery and continue to have chronic back pain to differentiate
recurrent disc herniation from dural scarring. Gadolinium-enhanced MRI
increases diagnostic accuracy for identifying recurrent disc
herniation. MRI has the ability to reveal early degenerative changes in
the disc. The MRI is, however, overly sensitive in that about 40% of
asymptomatic people older than 50 years of age have an abnormal signal
on MRI. It is also difficult to visualize the lateral recesses with MRI.
the evaluation of a patient with back pain, has limited use because of
the increased diagnostic sensitivity and specificity of MRI and CT
scanning. Myelography complications include seizures, arachnoiditis,
and induction of nausea and vomiting. It is also inadequate for
evaluating pathology in the lateral recesses. Myelography with
water-soluble dye is most commonly used in conjunction with CT scanning
to evaluate the thecal sac proximally and distally to the suspected
level.
parameters of spinal stenosis and lateral recess stenosis as well as
foraminal stenosis. CT scanning is also useful in patients suspected of
having vertebral osteomyelitis, tumors of the spine, and unrecognized
trauma. In patients with chronic back pain and radiculopathy,
electromyography may be useful in documenting a radiculopathy and in
differentiating a neuropathy from a myelopathy. Motor nerve conduction
velocity and sensory nerve conduction velocity testing may also help in
differentiating neuropathies. Electrodiagnostic studies help
differentiate patients with peripheral neuropathy or generalized or
demyelinating disorders from patients with compressive neuropathy.
conjunction with the patient’s history and physical findings to help
the physician arrive at a diagnosis and appropriate plan of management.
Few patients with chronic back pain require surgical intervention. The
most common conditions requiring back surgery in the adult are
recurrent disc herniation, spinal stenosis, and segmental lumbar
instability. Patients with chronic back pain without evidence of any of
the above conditions should be treated nonoperatively. Patients who do
not improve with symptomatic treatment (i.e. limited periods of rest,
anti-inflammatory agents, exercise and muscle strengthening programs)
should be evaluated in a comprehensive back pain clinic. These patients
generally need the multidisciplinary approach to provide for lifestyle
modification and rehabilitation. Otherwise, this limited group of
patients continues to be a financial, social, and economic burden on
society.
term that refers to any narrowing of the spinal canal or intervertebral
foramen. Stenosis of the spinal canal occurs primarily, as in
congenital spinal stenosis, or it can be a developmental condition,
such as in achondroplasia. Most often, it occurs secondary to
degenerative changes in the lumbar disc and facet joints, leading to
compression of the dural sac by the ligamentum flavum disc or bony
hypertrophy of the facet joints. Spinal stenosis can also occur after
an infectious process or a traumatic injury to the spine. Men and women
in the seventh and eighth decades of life are
the group primarily affected by degenerative spinal stenosis.
variable. The most common symptom scenario is that of either unilateral
or bilateral leg pain precipitated by walking and relieved by rest.
Other patients complain of pain or paresthesia in the buttocks, thighs,
or groin or in various distributions near the lower extremity.
absent. Neurologic deficits may or may not be present. Most patients,
however, have aggravations of symptomatology by extension of the lumbar
spine with relief of symptoms by forward flexion. Because of the
claudication-type symptoms, the main differential diagnostic disorder
is vascular claudication. All peripheral pulses should be checked;
vascular consultation may be needed. Plain radiographs generally reveal
degenerative changes of the spine consistent with aging changes.
Patients may show evidence of degenerative spondylolisthesis or
retrolisthesis. The diagnosis of spinal stenosis can best be made by
myelography followed by CT scanning.
anti-inflammatory agents and epidural steroid injections, but these
methods are generally unsuccessful. The treatment of choice for spinal
stenosis is often surgical decompression of the stenosed area. All bone
and soft tissues compressing the thecal sac or roots should be removed,
with care taken when possible to preserve the facet joints to avoid
creating segmental instability and the need for a fusion. About 70 to
85% of patients have good results from this procedure.
review article on early onset types of scoliosis outlines fundamental
aspects of diagnosis and management of these entities.
LG, Bridwell KH, Blanke K et al. Radiographic results of arthrodesis
with Cotrel-Dubousset instrumentation for the treatment of adolescent
idiopathic scoliosis: a five- to ten-year follow-up study. J Bone Joint
Surg 1998;80A:807. The radiographic results of
posterior spinal arthrodesis using Cotrel-Dubousset instrumentation in
76 patients with adolescent idiopathic scoliosis were evaluated. At an
average of 6 years postoperatively, the fusion appeared to be solid in
all patients.
authors studied the risk of progression in 727 patients with idiopathic
scoliosis. The risk of progression versus skeletal maturity, as well as
other risk factors in progression, are discussed.
is an in-depth analysis of the issues related to school screening for
scoliosis, the pertinent aspects of screening programs, and the
controversy surrounding them.
AL, Peterson LE. Effectiveness of treatment with a brace in girls who
have adolescent idiopathic scoliosis: a prospective controlled study
based on data from the brace study of the Scoliosis Research Society. J
Bone Joint Surg 1995;77A:815. In a prospective
study, 286 girls with adolescent idiopathic scoliosis, a
thoracic/thoracolumbar curve of 25 to 35°, and a mean age of 12.6 years
were followed. Brace treatment was successful in 74%; observation only
in 34%; and electrical stimulation in 33%.
KJ, Weinstein SL, Jacobson WC et al. Use of the Milwaukee brace for
progressive idiopathic scoliosis. J Bone Joint Surg 1996;78A:557. Immature
patients with idiopathic scoliosis who were treated with a Milwaukee
brace were evaluated. This study raises questions about whether the
natural history of progressive idiopathic scoliosis is truly altered by
use of the Milwaukee brace.
VT, Gillespie R. The characteristics of juvenile idiopathic scoliosis
and results of its treatment. J Bone Joint Surg 1978;60B:181. This
is a review article of 59 patients with juvenile idiopathic scoliosis.
The prognostic value of the rib-vertebral angle is discussed.
June 2004. Agency for Healthcare Research and Quality, Rockville, MD.
http://www.ahrq.gov/clinic/3rduspstf/scoliosis/scoliors.htm
reference textbook on all pediatric spine conditions; detailed
bibliography included and surgical techniques are discussed.
SL, Dolan LA, Spratt KF et al. Health and function of patients with
untreated idiopathic scoliosis: a 50-year natural history study. JAMA
2003;289:559. Fifty-one year long-term follow up
of 117 patients with untreated adolescent idiopathic scoliosis
evaluating outcomes related to health and function. They were compared
with 62 age- and sex-matched volunteers. The main outcome measures were
mortality, back pain, pulmonary symptoms, general function, depression,
and body image. The article concluded that untreated adults with
adolescent idiopathic scoliosis are productive and functional at a high
level at 50-year follow-up. Untreated adolescent idiopathic scoliosis
causes little physical impairment other than back pain and cosmetic
concerns.
authors discuss the factors in curve progression after skeletal
maturity in a group of 102 untreated patients followed for an average
of 40 years.
SL, Zavala DC, Ponseti IV. Idiopathic scoliosis: long-term follow-up
and prognosis in untreated patients. J Bone Joint Surg 1981;63A:702. The
authors followed 194 patients with untreated adolescent idiopathic
scoliosis for an average of 39.3 years. The authors studied the effects
on pulmonary function, psychological effects, mortality, morbidity, and
backache in this large, untreated population.
authors reviewed the natural history of 154 hemivertebrae in 104
patients. The authors determined the various risk factors, including
the type of hemivertebrae, location, age of the patient, and number of
hemivertebrae as well as their relation to each other.
MJ. Congenital scoliosis caused by unilateral failure of vertebral
segmentation with contralateral hemivertebrae. Spine 1998;23:998. This
article examines the behavior of congenital scoliosis caused by this
particular anomaly. The combination of growth on one side of the spine
and complete absence of longitudinal growth on the opposite side
inevitably results in severe deformity. The author also reports the
presence of intraspinal and neurologic anomalies, as well as anomalies
in other organ systems.
authors reviewed the natural history of 251 patient with congenital
scoliosis. Abnormalities are classified in regard to prognosis for each
pattern and curve location.
MJ, Singh H. Natural history of congenital kyphosis and kyphoscoliosis:
a study of one hundred and twelve patients. J Bone Joint Surg
1999;81A:1367. This study examines congenital
vertebral anomalies that result in deformity in the sagittal plane. The
authors expand the existing classification system and emphasize that
this pattern of congenital vertebral malformation has the highest risk
of neurologic defect, including paraplegia.
RB, Moe JH, Lonstein JE. The surgical treatment of congenital kyphosis:
a review of 94 patients 5 years or older, with 2 years or more
follow-up of 77 patients. Spine 1985;10:224. The
authors reported on a 7-year average follow-up of 94 patients with
congenital kyphosis. The results of posterior fusion alone versus
combined anterior and posterior fusion are presented.
RB, Moe JH, Lonstein JE. Posterior spinal arthrodesis for congenital
scoliosis: an analysis of the cases of 290 patients, 5 to 19 years old.
J Bone Joint Surg 1984;66A:1188. This article
reports on a 6-year average follow-up of 290 patients between 5 and 19
years of age who were treated by posterior spinal arthrodesis with or
without Harrington instrumentation. The authors report that the most
common problem was bending of the fusion mass.
BE, Baker D, McHolick WJ et al. The natural history of spondylolysis
and spondylolisthesis. J Bone Joint Surg 1984;66A:699. This
article describes a prospective study of 500 unselected firstgrade
children and their families, discussing the incidence, relation of
listhesis to lysis, and cause of disease.
BL III, Donati NL. Spinal arthrodesis for severe spondylolisthesis in
children and adolescents. J Bone Joint Surg 1989;71A:594. The
authors report on a 12-year follow-up of 12 patients with grade III or
IV (over 50%) spondylolisthesis. The article demonstrated that
posterior in situ arthrodesis is effective, reliable, and safe for
treatment of severe spondylolisthesis.
pain in children and adolescents usually has a recognizable organic
origin. The most common entities seen are spondylolysis,
spondylolisthesis, Scheuermann’s kyphosis, disc herniations,
infections, and tumors. Early recognition and treatment can provide
patients the best chance at relief of symptoms and eradication of the
underlying disease process.
IE, Weinstein SL. Long-term follow-up of patients with grade-III and IV
spondylolisthesis: treatment with and without posterior fusion. J Bone
Joint Surg 1987;69A:960. This article compares an
18-year follow-up of 11 patients with grade-III and IV
spondylolisthesis treated nonoperatively with a 24-year follow-up of 21
surgically treated patients. The surgical group was less symptomatic
and less restricted in their activities than the nonsurgical group. In
situ fusion gave good functional long-term results in grade-III and IV
spondylolisthesis.
RN. Current concepts review: spondylolysis and spondylolisthesis in
children and adolescents. J Bone Joint Surg 1989;71A:1098. This superb review article covers all aspects of the topic and has an extensive bibliography.
review article covers the history, physical examination, radiographic
examination, and differential diagnosis of the child presenting with
the chief complaint of back pain.
authors investigated 185 adolescents younger than age 19 years with
spondylolysis. The authors suggest that spondylolysis is caused by
repetitive microtrauma during growth and can be successfully treated
nonsurgically if treatment is started in the early stage.
M, Hahnel H, Robinson PN et al. Competitive sports and the progression
of spondylolisthesis. J Pediatr Orthop 1996;16:364. The
authors investigated the effects of several yeas of competitive sports
training on children and adolescents with spondylolisthesis. The
authors conclude that there is no justification for generally advising
children and adolescents with limited spondylolytic spondylolisthesis
not to take part in competitive sports.
PM, Weinstein SL, Spratt KF. Natural history and long term follow-up of
Scheuermann kyphosis. J Bone Joint Surg 1993;75A:236. The
authors report on a 31-year follow-up of 81 patients with Scheuermann’s
kyphosis. Pulmonary function, pain, work attendance, and disability are
evaluated.
RD, Wiltse L, Reynolds JB et al. In situ arthrodesis without
decompression for grade-III or IV isthmic spondylolisthesis in adults
who have severe sciatica. J Bone Joint Surg 1989; 71A:62. The
authors report on in situ fusions in 8 patients who had back pain and
sciatica caused by grade-III or IV isthmic spondylolisthesis of the
lumbar vertebrae and the sacrum. All patients achieved a solid fusion
with excellent relief of back pain and sciatica at 5.5 year average
follow-up.
PD, Hummer CD III. Nonoperative treatment for painful adolescent
spondylolysis or spondylolisthesis. J Pediatr Orthop 1989;9:538. The
authors demonstrate symptomatic relief of pain in two-thirds of
patients with spondylolysis and grade-I and II spondylolisthesis
treated nonoperatively. Adolescents with symptomatic grade-III and IV
spondylolisthesis are appropriately treated surgically.
N, Johnston CE, Browne RH. The prevalence of back pain in children who
have idiopathic scoliosis. J Bone Joint Surg 1997;79A:364. Although
back pain was noted in 32% of 2,442 patients with presumed idiopathic
scoliosis, only 9% of the patients with pain had an underlying
pathologic condition. If the neurologic examination was normal and
plain radiographs did not reveal lesions, MRI and bone scans were not
helpful.
B, Bradford D, Winter R et al. Scheuermann kyphosis: follow-up of
Milwaukee brace treatment. J Bone Joint Surg 1987;69A:50. This
article describes the long-term follow-up of 120 patients treated for
Scheuermann kyphosis with a Milwaukee brace, with a minimum follow-up
of 5 years after treatment. The authors demonstrate that the Milwaukee
brace is an effective method of treating patients with Scheuermann
kyphosis.
author reports on a long-term (mean, 29 years) clinical and
radiographic follow-up of 255 patients with spondylolisthesis and
spondylolysis. Half of these patients were treated for lower-back
symptoms.
is a superb monograph that reviews the history of lower-back pain and
sciatica for the past 3,500 years. The authors also discuss the problem
of chronic disability screening.
KC, Bridwell KH, Eisenberg BA et al. Minimum 5-year results of
degenerative spondylolisthesis treated with decompression and
instrumented posterior fusion. Spine 1999;24: 1721. This
study evaluated outcome and complication rate in 49 patients who had
undergone no prior surgery for degenerative spondylolisthesis.
Eight-three percent reported satisfaction with the procedure.
Radiographic transition syndromes were common (12 patients) with 5 of
12 patients being symptomatic. Major complications (2%), implant
failures (2%), and symptomatic pseudoarthrosis (0) were low.
patients come to the physician with uncomplicated low back pain.
Identifying the rare patient with significant pathology is challenging.
Assessment must be rapid with limited extensive investigation, a
rational pragmatic approach is outlined.
FJ, Currier B. Current concepts review: surgical management of lumbar
intervertebral-disc disease. J Bone Joint Surg 1989;71A:1266. This
is an excellent review article on management of the lumbar spine
intervertebral-disc disease. The authors discuss treatment by various
modalities and the status of imaging techniques.
JW, Newberg A, Pope MH et al. Spine radiographs in patients with
low-back pain: an epidemiologic study in men. J Bone Joint Surg
1984;66A:1048. The authors demonstrate that
degenerative changes of the lumbar spine increase with age. They report
that congenital and developmental changes and aging changes of the
spine occur in frequencies that do not support the use of radiographs
in back pain as a predictive tool for individual cases.
comprehensive, well-referenced review of the current state of knowledge
of the science of back pain and future directions of research is
presented.
prospective study evaluated predictors of outcome, including
sociodemographic factors, physical examination, radiographic,
psychological, social, and clinical history variables. The patients’
assessments of their own health and co-morbidity are the most important
outcome predictors of surgery for spinal stenosis.
article reports that back pain is the most common cause of disability
for people under 45 years of age. A herniated disc is the most common
problem in the age group of 30 to 39 years and is associated more with
sedentary occupations than active occupations. Degenerative changes
seen on spinal radiographs are more closely linked to the natural aging
process.
long-term result of standard lumbar diskectomy is not very satisfying.
More than one-third of the patients had unsatisfactory results and more
than one-fourth complained of significant residual pain. Heavy manual
work, particularly agricultural work, and low educational level were
negative predictors of a good outcome.
this study, between 85 and 90% of surgically treated and nonsurgically
treated patients with disc hernias were asymptomatic after 4 years.
Only 2% of subjects in both groups were symptomatic after 10 years.
After 1 year, the surgical group had less pain, but after 4 years there
was no statistically significant difference in relief of symptoms
between the two groups.
textbook, from the International Society for the Study of the Lumbar
Spine, covers all aspects of adult lumbar spine disease in great detail
and has a superb bibliography.
