Bone Lesions
Editors: Tornetta, Paul; Einhorn, Thomas A.; Damron, Timothy A.
Title: Oncology and Basic Science, 7th Edition
Copyright ©2008 Lippincott Williams & Wilkins
> Table of Contents > Section
II – Specific Bone Neoplasms and Simulators > 5 – Benign Bone Tumors
> 5.1 – Bone Lesions
II – Specific Bone Neoplasms and Simulators > 5 – Benign Bone Tumors
> 5.1 – Bone Lesions
5.1
Bone Lesions
Felasfa M. Wodajo
Osteoma
Osteoma of bone is a densely mineralized endosteal
lesion almost always seen in the head. The most common locations are in
the ethmoid and frontal sinuses. The lesions are uniformly benign and
usually discovered incidentally. In unusual cases, very large osteomas
of the paranasal sinuses can cause obstruction of the nasal air ducts
or other cranial symptoms. The radiographic hallmark is dense,
ivory-like mineralization with smooth margins. Unless there are
obstructive symptoms, observation is appropriate.
lesion almost always seen in the head. The most common locations are in
the ethmoid and frontal sinuses. The lesions are uniformly benign and
usually discovered incidentally. In unusual cases, very large osteomas
of the paranasal sinuses can cause obstruction of the nasal air ducts
or other cranial symptoms. The radiographic hallmark is dense,
ivory-like mineralization with smooth margins. Unless there are
obstructive symptoms, observation is appropriate.
Pathogenesis
Etiology
-
Unknown
-
Component of Gardner’s syndrome
Epidemiology
-
Estimated prevalence: 0.4%
-
Male:female ratio: 2:1
-
Location: ethmoid and frontal sinuses of the cranium most common
Pathophysiology
-
Osteomas are benign, indolent lesions.
-
Capacity for slow growth but no malignant potential
Diagnosis (See Algorithm 5.1-1)
Clinical Features
-
Typically incidental findings on dental or other cranial radiography
-
Occasionally, very large osteomas cause
sinus obstruction, loss of smell, or even in rare cases invasion into
intracranial structures.
Radiographic Features
-
Plain radiographs and computed tomography
(CT): smooth-contoured, densely mineralized lesion, within the
medullary bone with no cortical invasion
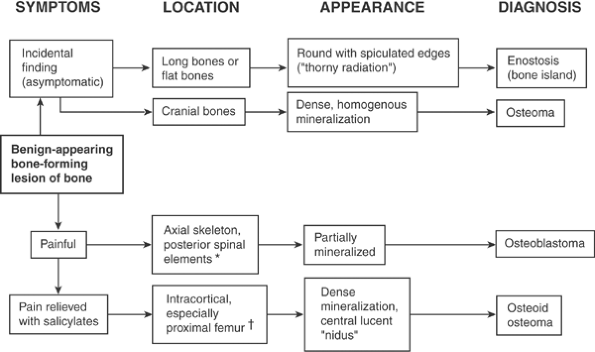 |
|
Algorithm 5.1-1.
Diagnosis of benign-appearing bone-forming lesions of bone. *Most common;may also occur in long bones, but rarely. †Most common;may also occur in posterior spinal elements and flat bone. |
Histologic Features
-
Mature lamellar bone with decreased marrow component
-
Sometimes haversian canals are seen within areas resembling cortical, compact bone.
Differential Diagnosis
Cemento-ossifying Fibroma (Ossifying Fibroma of Craniofacial Bones)
-
This is also another ossifying lesion of the cranium, but more often found in the mandible than the maxilla and cranial bones.
-
It must be distinguished from osteoma as it may have a more aggressive course and more often requires excision.
-
It appears as a well-circumscribed ossifying matrix, occasionally with expansile remodeling.
-
In contrast to dense ossification of osteoma, the periphery of these lesions is not always completely mineralized.
-
The histological appearance is also
different, showing a mix of fibrous and fibromyxoid stroma in addition
to combination of woven and lamellar bone and sometimes cementum.
Osteoid Osteoma
-
These have been described in the head and
neck and also appear as small, densely mineralized lesions but with the
important difference of a central radiolucent nidus and often nocturnal
pain relieved by salicylates, in contrast to incidental, asymptomatic
osteoma.
P.119
Chronic Sclerosing Osteomyelitis
-
Low-grade bone infections, presumably of
oral origin, occur in the maxilla and mandible and appear as sclerotic
lesions but are usually discovered due to symptoms.
Parosteal Osteosarcoma
-
A rare juxta-cortical variant of osteoma, parosteal osteoma, is sometimes confused with parosteal osteosarcoma.
-
The distinguishing feature of parosteal
osteoma is the presence of dense, homogenous mineralization, whereas
the mineralization in parosteal osteosarcoma may be incomplete and
heterogeneous.
Treatment
Surgical Treatment
-
Surgical intervention is rare as the vast majority of osteomas are asymptomatic, incidental findings.
-
Excision has been occasionally reported for rare lesions with obstructive symptoms of the sinuses.
Results and Outcome (Prognosis)
-
Observed lesions may progress very slowly.
Enostosis
Enostosis or “bone island” is a small, asymptomatic,
densely mineralized lesion found within the cancellous bone. It is
typically an incidental finding; the important diagnostic challenge
often is ruling out the possibility of the lesion representing a small
bone metastasis. The characteristic radiating thick trabeculae seen on
imaging studies and absence of uptake on bone scan are distinguishing
features.
densely mineralized lesion found within the cancellous bone. It is
typically an incidental finding; the important diagnostic challenge
often is ruling out the possibility of the lesion representing a small
bone metastasis. The characteristic radiating thick trabeculae seen on
imaging studies and absence of uptake on bone scan are distinguishing
features.
Pathogenesis
-
Etiology is unknown.
Epidemiology
-
Most often found in adults undergoing
testing for other reasons, although presumably develops at a younger
age during skeletal maturation -
No gender predilection or inheritance pattern
-
Distribution
-
Found within cancellous bone
-
Preference for pelvis, femur, and other long bones
-
Relatively rare in the spine
-
Pathophysiology
-
Disorder of endochondral ossification
-
An error in resorption of the secondary
spongiosum during bone formation leads to an island of heavily
calcified matrix left within the spongy, cancellous bone.
-
-
Enostoses do not change in size over time and have no malignant potential.
Diagnosis (See Algorithm 5.1-1)
Clinical Features
-
Asymptomatic, incidental findings, usually discovered during evaluation for other reasons
Radiologic Features
Radiographs
-
Round or ovoid, homogeneously dense, sclerotic focus in cancellous bone
-
Distinctive finding is the presence of radiating bony streaks (“thorny radiation”) emanating from the sclerotic center.
-
Almost always small, ranging from 1 mm to 2 cm
-
“Giant” bone islands, measuring up to 6 cm, have been reported, but large lesions should raise suspicion for other diagnoses.
Computed Tomography/Magnetic Resonance Imaging
-
Distinctive “brush borders” are more clearly seen on CT and are diagnostic.
-
Appearance on magnetic resonance imaging (MRI) examination should emulate cortical bone.
Bone Scan
-
Most important radiographic feature of
enostosis is the usual absence of uptake on bone scan, which—when
present—differentiates it from metastatic disease. -
Occasional scintigraphically active
enostoses have been described, suggesting caution in using bone scan as
the only modality for diagnosis, but bone scan remains very important
in diagnosis.
Histologic Features
-
Features include mature lamellar pattern with haversian canals within a focus of compact (cortical) bone.
-
Thickened, radiating trabeculae merge with the surrounding trabeculae in the periphery of the lesion (Fig. 5.1-1).
Differential Diagnosis
Metastasis
-
Differentiating enostosis from bone metastases is the most important diagnostic challenge.
-
Small blastic lesions can be seen in breast cancer and prostate cancer metastases.
-
Metastases usually show increased uptake on bone scan.
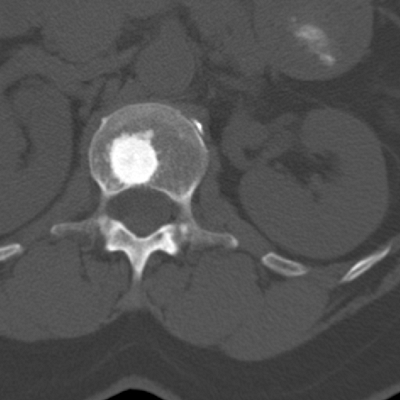 |
|
Figure 5.1-1 Enostosis in vertebral body. Note dense mineralization and thickened, radiating trabeculae.
|
Osteopoikilosis
-
Osteopoikilosis is an inherited autosomal
dominant disorder in which numerous enostoses are found, usually at the
ends of long bones. -
Similar to isolated enostosis, no uptake is seen on bone scan.
Osteopetrosis (Albers-Schönberg Disease)
-
This is typically a polyostotic disorder caused by an error in resorption of the primary spongiosum.
-
The hallmark is dense osteosclerosis filling the entire medullary canal, as opposed to isolated bone islands.
Enchondroma
-
A small enchondroma may be difficult to distinguish from an enostosis.
-
CT imaging should help distinguish the pattern of mineralization.
-
Enchondromas demonstrate incomplete
mineralization with a speckled or whorled pattern in contrast to the
dense, homogenous mineralization of enostoses. -
Enchondromas almost always demonstrate uptake on bone scan.
Treatment
Surgical Treatment
-
No surgery is indicated; the main role of the consulting surgeon is to exclude other diagnostic possibilities.
Prognosis
-
Observed lesions rarely progress.
Osteoid Osteoma
Osteoid osteoma is a small, painful bone lesion that
most commonly occurs in the cortices of long bones, with a prediction
for the proximal femur. The pain pattern is usually distinctive, with
nightly pain rapidly alleviated with salicylates (aspirin). The lesion
has a characteristic small, radiolucent central nidus measuring up to 1
to 2 cm and thick, surrounding reactive bone. Standard treatment is
percutaneous radiofrequency ablation.
most commonly occurs in the cortices of long bones, with a prediction
for the proximal femur. The pain pattern is usually distinctive, with
nightly pain rapidly alleviated with salicylates (aspirin). The lesion
has a characteristic small, radiolucent central nidus measuring up to 1
to 2 cm and thick, surrounding reactive bone. Standard treatment is
percutaneous radiofrequency ablation.
Pathogenesis
Etiology
-
Unknown
-
Postulated developmental error or vascular malformation rather than a true neoplasm
Epidemiology
-
Age of presentation: average 19 years (typical range 10 to 35 years)
-
Youngest patient: age 2 years
-
Male preponderance
Anatomic Location
-
Most commonly found in the long bones, especially the femur and tibia
-
Predilection for the proximal femur
-
Less common, subperiosteal lesions have been described on the surface of long bones.
-
-
Juxta-articular lesions: most common in the hip but have been described at many different articulations
-
Spine lesions
-
Lumbar more than cervical
-
Most common in posterior elements, typically in laminae
-
Pathophysiology
-
Presence of nerve fibers
-
Detected in the reactive zone and nidus of osteoid osteomas
-
Not seen in other bone tumors (osteoblastoma, osteosarcoma, etc.)
-
-
Increased levels of prostaglandins also detected within nidus
-
Increased vascular pressure due to prostaglandins may stimulate afferent nerves in nidus, causing pain.
-
May explain why salicylates provide such dramatic pain relief
-
Natural History
The nidus does not increase in size over time, although
reactive bone may become more prominent. Some authors have stated that
some osteoid osteomas “burn out” over time. Infrequent painless cases
have been described (1.6%
reactive bone may become more prominent. Some authors have stated that
some osteoid osteomas “burn out” over time. Infrequent painless cases
have been described (1.6%
P.121
in one review of 860 patients). Nevertheless, the actual incidence of spontaneous regression is difficult to document.
Diagnosis (Algorithm 5.1-1)
Clinical Features
-
Clinical hallmark of osteoid osteoma is pain at rest and especially night pain.
-
The pain is dramatically relieved by
salicylates (aspirin) typically within 20 to 25 minutes. While this is
a reliable sign, the absence of immediate relief does not rule out the
possibility of osteoid osteoma, as it occurs in only approximately 70%
of patients.
-
-
Symptoms typically occur for weeks to years before the patient seeks medical attention.
-
The pain may be referred to remote anatomic locations (e.g., femur lesion presenting with leg or knee pain).
-
Over time, symptoms gradually worsen, with an intermittent pain becoming a more constant ache.
-
Lesions in special anatomic locations may have different presentations.
-
Juxta-articular lesions
-
May present with monoarticular arthropathy consisting of joint effusion and synovitis
-
In prolonged cases, degenerative articular changes
-
-
Spinal locations
-
Usually present with painful scoliosis caused by muscle spasm
-
Lesion found at the concavity of the curve
-
-
Radiologic Features
Radiography
-
Small, sclerotic intracortical lesion with a central radiolucent nidus
-
Intracortical lesions: easy to detect on
plain radiography due to thick, dense reactive sclerosis. Often, the
central, radiolucent nidus is visible on radiographs if the appropriate
projection is obtained (Fig. 5.1-3).-
Intramedullary or endosteal lesions: less reactive bone formation and more difficult to diagnose radiographically (see Fig. 5.1-3)
-
Subperiosteal lesions: may be extremely difficult to see on plain radiographs
-
CT
-
CT is the definitive study modality, allowing for exact localization of nidus.
-
Protocol: thin (2-mm) cuts
-
Findings: The combination of thick,
uniform reactive bone surrounding a central, 1- to 2-cm or less
radiolucent nidus is highly suggestive of osteoid osteoma. -
Subperiosteal osteoid osteomas also have
a radiolucent nidus but incite a less dense, nonconfluent (“shaggy”)
reactive bone formation.
Bone Scan
-
Increased uptake is always identified in active osteoid osteomas.
-
Scintigraphy is very sensitive for
detecting the nidus and especially useful in subperiosteal and
intra-articular lesions, which may be missed on plain radiography.
Increased radiotracer activity is seen on immediate and delayed phases. -
“Double-density” sign: small focus of
increased activity corresponding to nidus surrounded by larger, less
intensely active region corresponding to the surrounding reactive bone
MRI
-
MRI is of questionable value, as the large area of marrow and soft tissue edema can obscure diagnosis.
-
It may be helpful in suspected cases of
juxta-articular osteoid osteoma, where intense marrow edema in only one
bone may be the initial clue.
Histologic Features
-
The nidus of osteoid osteoma is a small,
discrete lesion with disorganized seams of unmineralized or partially
mineralized osteoid with prominent osteoblastic and osteoclastic
activity. -
The spaces between the osteoid seams contain highly vascular, fibrous stroma.
-
Toward the periphery, the osteoid is more mature and dense with smaller osteoblasts.
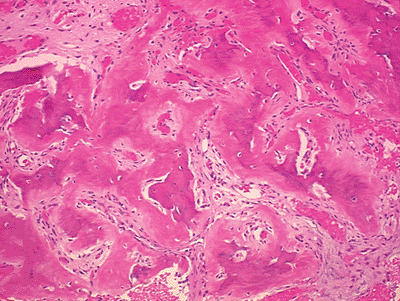 |
|
Figure 5.2-1
Histology for both osteoid osteoma and osteoblastoma show similar disorganized woven bone with intervening fibrovascular stroma. |
Differential Diagnosis
Stress Reaction
-
Differentiating stress reaction (stress fracture) from osteoid osteoma is not an uncommon dilemma in young patients.
-
Both often present with unicortical periosteal reaction and pain.
-
The pain pattern is typically diurnal and
related to weight bearing on the affected extremity in stress reaction
and nocturnal and nonmechanical in osteoid osteoma. -
Reactive bone formation runs transverse to long axis of bone in stress reaction rather than parallel to it in osteoid osteoma.
P.122
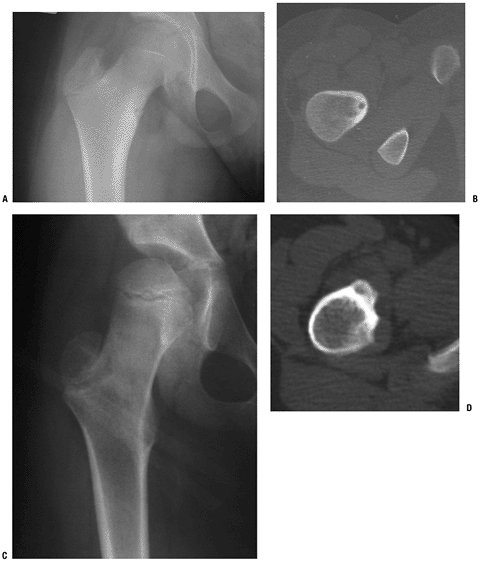 |
|
Figure 5.1-3 Osteoid osteoma in proximal femur. (A,B)Classic intracortical osteoid osteoma with central nidus and thick, reactive bone formation. (C,D)Subperiosteal variant with almost normal plain radiographs and less vigorous reactive bone formation.
|
Chronic Osteomyelitis (Brodie’s Abscess)
-
Chronic osteomyelitis in bone may present with thick reactive bone formation and central lucency.
-
If there is a radiolucent central region, it is often larger than 2 cm.
-
Bone scan should be beneficial,
demonstrating center photopenia in infection, representing the necrotic
sequestrum, versus intense central uptake representing the nidus in
osteoid osteoma. -
MRI may not be helpful in distinguishing
the two as both infection and osteoid osteoma may demonstrate extensive
soft tissue and marrow edema.
Enostosis
-
Enostosis also appears as a sclerotic intraosseous lesion but should be painless and lacking uptake on scintigraphy.
-
Also helpful should be the brush borders of thickened trabeculae emanating from central, sclerotic core seen on CT in enostosis.
P.123
Osteoblastoma
-
Although osteoblastoma is commonly
grouped in the differential diagnosis of osteoid osteoma, it is a
radiographically and histologically distinct entity. -
It is larger, with the nidus of osteoid
osteomas rarely exceeding 2 cm in size, and comparatively only
partially mineralized with a thin, expansile rim of sclerotic bone.
Intracortical Hemangioma
-
Hemangioma of bone is most common in the vertebral bodies, where it presents with vertical, thickened trabeculae.
-
It can also present as single or multiple sclerotic appendicular skeletal lesions.
-
While some patients may experience pain, the nocturnal pattern and response to salicylates help distinguish the two.
-
The lesions tend to be less well circumscribed, without the typical central lucent nidus.
Treatment
Surgical Management
-
Although there may be some capacity for
osteoid osteomas to spontaneously regress, in general definitive
operative or medical treatment is indicated for all symptomatic osteoid
osteomas.
Preferred Treatment
-
Radiofrequency ablation (RFA) is
performed as an outpatient procedure under CT guidance. Heat generated
in the tissues near the tip of the probe necroses a small portion of
bone and, presumably, the nidus. -
Contraindications include spinal lesions
near cord or nerve root, subchondral lesions near articular surface,
and inaccessible lesions.
Alternative Treatment
-
Intralesional resection of the nidus (curettage)
-
Indications include recurrent tumors not amenable to repeat RFA and lesions for which the diagnosis is in doubt.
-
The main surgical challenge is locating the small nidus intraoperatively. Several techniques have been described:
-
Preoperative CT-guided localization via an implanted wire
-
Intraoperative nuclear scintigraphy (preoperative dye administration)
-
Preoperative fluorescent tetracycline labeling
-
Medical Treatment
-
Salicylates or nonsteroidal anti-inflammatory agents (NSAIDs)
-
Median duration of treatment before resolution of symptoms: 30 months
Results and Outcome
-
The success rate of RFA is 80% to 90%. If initial treatment is unsuccessful, a second procedure is successful in most instances.
-
Success rate for open curettage has been reported to be approximately 90%.
-
Scoliosis induced by osteoid osteoma corrects after treatment of osteoid osteoma.
-
The use of NSAIDs alone was reported to
result in resolution of symptoms in approximately 70% of patients after
a mean of 2.5 years in one study.
Osteoblastoma
Osteoblastoma is a benign, locally aggressive bone
neoplasm in which abundant, plump osteoblasts produce disorganized and
immature osteoid. It appears as a radiolucent, sometimes expansile,
lesion with hazy mineralization and a thin sclerotic rim. It has a
predilection for the spine and tends to recur if not completely excised.
neoplasm in which abundant, plump osteoblasts produce disorganized and
immature osteoid. It appears as a radiolucent, sometimes expansile,
lesion with hazy mineralization and a thin sclerotic rim. It has a
predilection for the spine and tends to recur if not completely excised.
Pathogenesis
-
Etiology is unknown.
Epidemiology
-
1% of primary bone tumors and 3% of benign bone tumors
-
Peak occurrence is in the first to third decades, with 80% of cases occurring in patients under 30 years old.
-
Male:female ratio 2:1
-
Distribution
-
Predilection for the spine (60% to 70% of cases)
-
Equally in cervical, thoracic, and lumbar regions
-
More commonly in the posterior elements (66% in one series)
-
Nonaxial tumors can be found in round or flat bones as well as cranial bones
-
Within the long bones, metaphyseal = diaphyseal, proximal femur >mt distal femur, proximal tibia
-
Also in juxtacortical or periosteal
locations, with an appearance somewhat analogous to osteoid osteoma,
except with a much larger, central lucent area
-
-
Natural History
Although histologically similar in some respects to
osteoid osteoma, osteoblastoma is a distinct entity with a different
natural history—one of progression rather than stasis or regression. A
range of manifestations has been observed, from indolent growth to
aggressive local growth.
osteoid osteoma, osteoblastoma is a distinct entity with a different
natural history—one of progression rather than stasis or regression. A
range of manifestations has been observed, from indolent growth to
aggressive local growth.
In aggressive tumors, there is a tendency for multiple
recurrences over a long period of time. Disease-free periods of up to
17 years have been reported. One patient had 11 surgeries over 30
years. However, even with multiple recurrences,
recurrences over a long period of time. Disease-free periods of up to
17 years have been reported. One patient had 11 surgeries over 30
years. However, even with multiple recurrences,
P.124
the tumor typically remains benign. Nevertheless, rare cases of sarcomatous transformation have been described.
Diagnosis (See Algorithm 5.1-1)
Clinical Features
-
Pain is a typical symptom.
-
Often it is dull and aching.
-
Duration of 6 to 12 months before diagnosis
-
Rarely discovered incidentally
-
Nocturnal pain, like that seen in osteoid osteoma, is rare.
-
Not relieved by salicylates or NSAIDs
-
-
Physical examination reveals tenderness over tumor site.
-
Spinal osteoblastomas can cause painful scoliosis.
-
Objective neurologic findings in 29% of patients in one series
-
Deficits mostly minimal (e.g., muscle
weakness, sensory disturbance, or abnormal reflexes), although two
patients had neurogenic bladders and paraparesis
-
Radiologic Features
Radiographs
-
Typical appearance is of radiolucent lesion with faint densities.
-
A large range of radiographic appearances have been noted.
-
Cortical expansion is common, especially in the spine (Fig. 5.1-4), where there may be a significant aneurysmal component.
-
Dense mineralization may be seen in longstanding lesions or after radiotherapy.
-
Reactive bone formation is seen in lesions arising in cortex and less so with intramedullary lesions.
-
CT
-
CT scanning shows more detail, outlining
more clearly the sclerotic rim surrounding and separating the lesion
from adjacent soft tissues or surrounding bone. -
CT is also better for showing the sometimes subtle hazy mineralization of the lesion itself (see Fig. 5.1-3).
Histologic Features (See Fig 5.1-2)
-
Disorganized seams of abundant osteoid production in the form of woven bone
-
Similar to that of the central nidus in
osteoid osteoma but without the tendency toward more organized, mature
trabeculae seen toward the periphery -
“Epithelioid” osteoblasts (rounded and larger than normal)
-
-
Frequently seen: vascular spaces and giant cells, the latter of which are not normally seen in osteoid osteoma
P.125
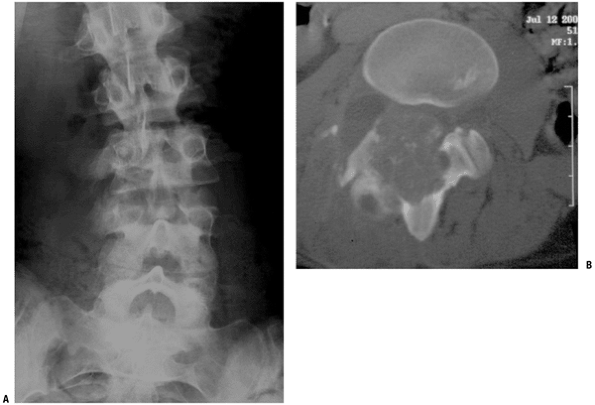 |
|
Figure 5.1-4 Osteoblastoma in lumbar spine. (A) Plain radiograph showing expansile, osteoblastic lesion with scoliosis.The lesion is in concavity. (B) CT scan showing expansile lesion in posterior spinal elements with faint calcifications and thin, incomplete sclerotic rim.
|
Differential Diagnosis
Osteoid Osteoma
-
Clinically, the characteristic nocturnal pain of osteoid osteoma is not typical of osteoblastoma.
-
No distinct small nidus is seen on imaging studies.
-
Histologically, the large vascular spaces and giant cells seen in osteoblastoma are not common in osteoid osteoma.
Aneurysmal Bone Cyst
-
These can resemble osteoblastoma radiographically and histologically.
-
In particular, osteoblastomas have a known propensity for secondary aneurysmal cyst formation.
-
If osteoblastoma is suspected in a
resected aneurysmal bone cyst, thorough sampling of the tumor is
necessary to locate what may be only a small, focal area of
osteoblastoma. -
Disorganized osteoid seams and osteoblastic proliferation should be absent in primary aneurysmal bone cyst.
Giant Cell Tumor of Bone
-
The similarity in the age of presentation
of the two tumors and the presence of giant cells on histological
examination of osteoblastoma may be a source of confusion. -
No mineralization is found in giant cell tumor.
-
Further, giant cell tumor is less common in the spine and more common anteriorly, in the vertebral bodies, when it does occur.
Chondroblastoma
-
Chondroblastoma may also present as an intraosseous lesion with stippled radiodensities.
-
It is almost exclusively seen in the epiphyses of long bones.
-
Histology also shows immature cartilage (chondroid), not seen in osteoblastoma.
Low-Grade Osteosarcoma
-
Imaging studies should be distinctive,
with conventional osteosarcoma rarely showing a sclerotic rim and
dense, surrounding, reactive bone formation. -
Disorganized and immature osteoid is seen
in osteosarcoma, but the hallmarks of malignancy (e.g., cellular and
nuclear pleomorphism and mitoses) are absent in osteoblastoma.
Treatment
Surgical Treatment
Recommended Treatment
-
Complete intralesional resection is recommended.
-
The use of a physical adjuvant, such as phenol or liquid nitrogen, may help reduce the risk of recurrence.
-
Wide resection may be considered in expendable bones such as rib, fibula, or metacarpals.
Indications
-
All tumors undergo surgical resection due to natural history of progressive growth and worsening symptoms in untreated tumors.
Adjuvant Treatments
-
Adjuvant radiation has been used but
probably should be reserved for recurrent or inoperable lesions.
Radiation may increase the risk of sarcomatous transformation.
Results and Outcome
Complete excision is typically curative. Local
recurrence is higher in difficult anatomic locations, such as the
spine. In one series of 23 patients across multiple institutions, two
recurrences were reported, one patient with a disease-free interval of
17 years and one patient who had 11 operations over 27 years. In
another series of 53 patients with more than 1 year of follow-up, 3 of
27 of spinal lesions recurred, 2 in patients who received adjuvant
radiation, and 4 of 15 extremity lesions recurred, 1 in a patient who
received postoperative radiation.
recurrence is higher in difficult anatomic locations, such as the
spine. In one series of 23 patients across multiple institutions, two
recurrences were reported, one patient with a disease-free interval of
17 years and one patient who had 11 operations over 27 years. In
another series of 53 patients with more than 1 year of follow-up, 3 of
27 of spinal lesions recurred, 2 in patients who received adjuvant
radiation, and 4 of 15 extremity lesions recurred, 1 in a patient who
received postoperative radiation.
Cases of mortality and amputation have been reported due
to aggressive and repeated local growth of the tumor in the spine and
extremities.
to aggressive and repeated local growth of the tumor in the spine and
extremities.
Acknowledgments
I gratefully acknowledge research assistance by Brittany
L. Rice, MLS, AHIP, and Smita Jhaveri at the Suburban Hospital Medical
Library.
L. Rice, MLS, AHIP, and Smita Jhaveri at the Suburban Hospital Medical
Library.
Suggested Reading
Beauchamp CP, Duncan CP, Dzus AK, et al. Osteoblastoma: experience with 23 patients. Can J Surg 1992;35:199–202.
Greenspan
A. Benign bone-forming lesions: osteoma, osteoid osteoma, and
osteoblastoma. Clinical, imaging, pathologic, and differential
considerations. Skel Radiol 1993;22:485–500.
A. Benign bone-forming lesions: osteoma, osteoid osteoma, and
osteoblastoma. Clinical, imaging, pathologic, and differential
considerations. Skel Radiol 1993;22:485–500.
Greenspan A. Bone island (enostosis): current concept—a review. Skel Radiol 1995;24:111–115.
Ilyas I, Younge DA. Medical management of osteoid osteoma. Can J Surg 2002;45:435–437.
Marsh BW, Bonfiglio M, Brady LP, et al. Benign osteoblastoma: range of manifestations. J Bone Joint Surg [Am] 1975;57:1–9.
Nemoto O, Moser RP, Van Dam BE, et al. Osteoblastoma of the spine: a review of 75 cases. Spine 1990;15:1272–1280.
O’Connell JX, Nanthakumar SS, Nielsen GP, et al. Osteoid osteoma: the uniquely innervated bone tumor. Mod Pathol 1998;11:175–180.
Rimondi
E, Bianchi G, Malaguti MC, et al. Radiofrequency thermoablation of
primary non-spinal osteoid osteoma: optimization of the procedure. Eur Radiol 2005;15:1393–1399.
E, Bianchi G, Malaguti MC, et al. Radiofrequency thermoablation of
primary non-spinal osteoid osteoma: optimization of the procedure. Eur Radiol 2005;15:1393–1399.
Sciubba JJ, Younai F. Ossifying fibroma of the mandible and maxilla: review of 18 cases. J Oral Pathol Med 1989;18:315–321.
Shankman S, Desai P, Beltran J. Subperiosteal osteoid osteoma: radiographic and pathologic manifestations. Skel Radiol 1997;26:457–462.
Torriani M, Rosenthal DI. Percutaneous radiofrequency treatment of osteoid osteoma. Pediatr Radiol 2002;32:615–618.
Vanhoenacker FM, De Beuckeleer LH, Van Hul W, et al. Sclerosing bone dysplasias: genetic and radioclinical features. Eur Radiol 2000;10:1423–1433.
