Diarthrodial Joints
Editors: Tornetta, Paul; Einhorn, Thomas A.; Damron, Timothy A.
Title: Oncology and Basic Science, 7th Edition
Copyright ©2008 Lippincott Williams & Wilkins
> Table of Contents > Section IV – Basic Science > 21 – Diarthrodial Joints
21
Diarthrodial Joints
Thomas V. Smallman
Kris Shekitka
Donald Flemming
In tribute to the late Donald E. Sweet, Director of
Department of Orthopaedic Pathology, Armed Forces Institute of
Pathology, Washington, DC.
Department of Orthopaedic Pathology, Armed Forces Institute of
Pathology, Washington, DC.
The objective of this chapter is to review briefly the
development of joints and their normal structure and function and then
to outline concisely the pathophysiology of arthritic disorders.
Infectious arthritis is fully covered in Chapter 27,
Infectious Disorders of Bone and Joints, and is not discussed here. The
structure and function of hyaline cartilage and the cell biology of
cartilage repair are covered in Chapter 20, Articular Cartilage Structure, Composition, and Repair. The reader might wish to review the key elements in this chapter prior to studying this section.
development of joints and their normal structure and function and then
to outline concisely the pathophysiology of arthritic disorders.
Infectious arthritis is fully covered in Chapter 27,
Infectious Disorders of Bone and Joints, and is not discussed here. The
structure and function of hyaline cartilage and the cell biology of
cartilage repair are covered in Chapter 20, Articular Cartilage Structure, Composition, and Repair. The reader might wish to review the key elements in this chapter prior to studying this section.
Background
The articular system consists of:
-
Diarthrodial joints: mobile joints
consisting of hyaline cartilage covering juxtaposed bone ends
surrounded by a capsule, the innermost layer of which is synovium-
Factors to consider in joint motion
-
Joint geometry
-
Loading environment
-
Soft tissue and articular surface load sharing
-
-
Loading is accomplished through deformation of the whole joint complex.
-
-
Amphiarthroses: less mobile structure
consisting of a fibrocartilaginous plate between two bone ends covered
by hyaline cartilage with an intervening “disc” material surrounded by
a fibrous capsule-
Examples include the symphysis pubis, the
posterior superior two thirds of the sacroiliac joint (anterior
inferior third is synovial), and the intervertebral disc (see Chapter 23). -
Nature of intervening material determines the degree of motion achieved.
-
-
Syndesmoses: nonmobile junction between bone ends, consisting of a simple fibrous connection
-
Examples include the distal tibiofibular joint and the skull sutures.
-
Disorders of joints are numerous. The essential steps of
history, physical examination, problem definition, and investigation
will usually lead the clinician to an ordered approach, a listing of
options that leads to further inquiry, and usually an ultimate
diagnosis. An understanding of normal growth and development of joints
provides a basis on which to develop models for the pathophysiology of
disease.
history, physical examination, problem definition, and investigation
will usually lead the clinician to an ordered approach, a listing of
options that leads to further inquiry, and usually an ultimate
diagnosis. An understanding of normal growth and development of joints
provides a basis on which to develop models for the pathophysiology of
disease.
Classification
The classification of joint disorders is shown in Box 21-1.
Development of Diathrodial Joints
(Limb bud development is reviewed in Chapter 14, Growth and Development of the Musculoskeletal System.)
Box 21-1 Classification of Joint Disorders
-
Osteoarthritis
-
Primary
-
Secondary
-
-
Inflammatory Arthritis
-
Rheumatoid arthritis
-
Juvenile rheumatoid arthritis (Still’s disease)
-
Spondyloarthropathies
-
Ankylosing spondylitis
-
Reactive arthritis
-
Psoriatic arthritis
-
Arthritis associated with inflammatory bowel disease
-
Undifferentiated
-
Infectious Arthritis-
Bacterial
-
Granulomatous (tubercular, fungal, sarcoidosis)
-
Lyme arthritis
Metabolic-
Gout
-
CPPD crystal deposition disease
-
Ochronosis
-
Hemophilia
Circulatory Disorders Affecting Joints-
Neuropathic arthropathy
-
Avascular necrosis
Anomalies-
Epiphysealis hemimelica
-
P.419
-
Beginning during the sixth week,
condensed cells in central mesenchyme within each limb bud become
chondroblasts, making very primitive cartilage, so-called cartilage
models of each bone. -
By end of the eighth week within each
limb bud, beyond the central cartilage, mesenchymal cells continue to
transform into all elements of the limb (skeletal muscle, ligaments,
tendons, fat, vascular elements). -
Dense interzones, darker areas between the chondrified bone models, appear at 6 weeks and will become the joints (Fig. 21-1).
-
Cavities within interzones appear at 10 weeks (Fig. 21-2).
-
Mechanism of cavity formation:
genetically determined enzymatic destruction with or without simple
elaboration of synovial fluid; does not require activity of muscles,
often occurs before adjacent muscles have formed or attached -
Persistence and maintenance of joints,
once formed, require muscle action. Formation of the unique anatomy of
each joint unfolds locally. -
All of the knee structures are present at
14 weeks. Menisci form as fibrocartilaginous semicircles with
peripheral attachment; at no time in normal development are they
complete discs separating the bone ends. -
All elements of the hip are formed at 4.5 months.
Normal Structure and Function of Joints
-
Homeostasis in a diarthrodial joint
involves important relationships between the three key components of
the joint: articular cartilage, synovium, and subchondral bone.-
Three important vectors of circulatory,
mechanical, and metabolic influences are in constant interplay at any
given time, affecting each of these components.
-
Articular Cartilage
-
Articular cartilage is aneural and
avascular, with diffusion from synovial fluid through the extracellular
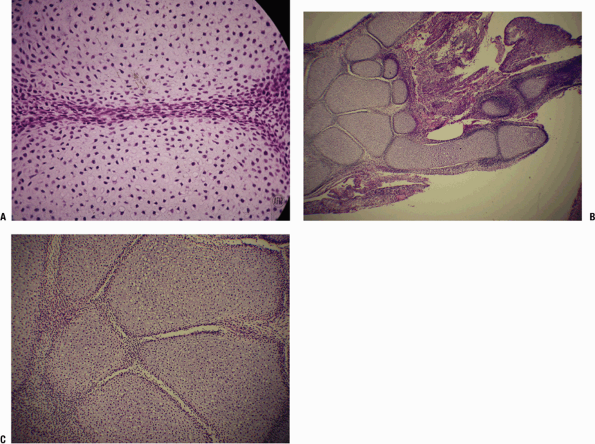
matrix being the route for its nutrition.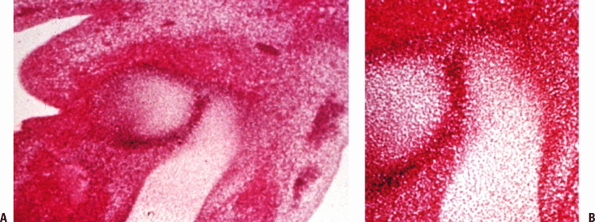 Figure 21-1
Figure 21-1
Limb bud. (A) Cross-section of limb bud shows early cartilage models,
forming from mesenchymal cells that have modulated to form early
cartilage. The dark area, the interzone between the cartilage models,
is the joint anlage. (B) Higher-power view of the interzone shows that
at this time (6 weeks), the interzone is a collection of primitive
cells that have coalesced between the lighter-staining cartilage model
bones.![]() Figure 21-2
Figure 21-2
Synovial joint development. (A) Cells in the interzone flatten out and
begin to secrete synovial fluid. The primitive cells, densely packed,
are pushed apart by the fluid thus elaborated, and ultimately a joint
space is created. On either side of the joint space the cells flatten
out. (B) Section of the end of the limb but in an embryo (estimated 10
weeks). Portions of the thumb, index, and little finger as well as the
carpal bone cartilage models are seen. Some joints have well-developed
joint spaces, and some are at an earlier stage or the cut has missed
the joint space. (C) At a higher power, some of the cartilage cells are
beginning to hypertrophy. The interosseous ligaments between trapezium
and capitate and between lunate and triquetrum are seen. -
Chondrocyte mediates all processes for articular cartilage: synthesis, maintenance, degradation, and repair.
-
Repair is problematic (see Chapter 14)
because the replacement material that results from the repair process
lacks the normal architecture of the articular cartilage (see Fig. 20-1A in Chapter 20). -
Each joint is unique, reflecting its
biomechanical tasks of motion, weight transmission, and stability.
Inherent in the structure of each joint is conforming bony and
articular surface anatomy, the synovial lining layer, ligaments and
capsule with unique local geometry determined by the motion required,
allowing recesses for accumulation and disposition of debris, and
specialized fluid transport channels. The joint remodels throughout
life, shedding material and cells from the surface of the articular
cartilage, and replacing these cells with those arising from a growth
zone located adjacent to the tide-mark. -
Motion and activity within physiologic
limits are essential. An appropriate exercise pattern throughout life
promotes through Wolff s law the development and maintenance of optimal
structure of bone, articular cartilage, ligaments, muscles, and tendons.
P.420
Synovium
-
The role of the synovium is to maintain
joint homeosta-sis and to provide the appropriate response in disease
states. Synovium differs from true epithelium because it has no
basement membrane, thus facilitating exchange of nutrients and other
substances between the vascular system and the joint fluid. -
Three histologic types are described:
-
Areolar synovium
-
Structure
-
Surface compact layer of collagen (types I and III) and synovial cells
-
Deeper layer is of fibrovascular matrix,
vessels, nerves, migratory cells (mast cells and mononu-clear cells),
and ground substance, including dermatan sulfate and hyaluronic acid.
-
-
Three cell types exist:
-
Phagocytic cells (type A synoviocytes)
-
Maintenance cells (type B synoviocytes)
-
Intermediate forms (type C synoviocytes)
-
-
-
Fibrous synovium
-
Fatty or adipose synovium
-
Prototypical location is the fat pad of the knee; acts as mobile packing tissue, changing shape to match joint motion
-
Structure: Surface layer is collagen,
surface lining cells, and neurovascular elements forming a continuous
array with only a few very small gaps a few microns wide. -
Deeper layer is fat and fibrous septa containing occasional synovial cells, vessels, and nerves.
-
Function of the fat pad of the knee may be much more complex than simple padding and protection, and may include roles in:
-
Paracrine function through the elaboration of cytokines
-
Mechanical function to condition motion between the patella, femur, and tibia
-
Neural function in proprioception, or in reactive neural inflammation through substance P fibers
-
-
-
Subchondral Bone
Each joint is a unique engine of force transmission. Dr.
Sweet used an analogy in which one can think of the articular cartilage
as representing the tire, the subchondral bone the rim, and the
cancellous array the spokes. Cortical bone then acts as a deformable
unit, accepting force and transmitting it to the metaphyseal cancellous
array, which distributes the force uniformly to the articular cartilage
(Fig. 21-3).
Sweet used an analogy in which one can think of the articular cartilage
as representing the tire, the subchondral bone the rim, and the
cancellous array the spokes. Cortical bone then acts as a deformable
unit, accepting force and transmitting it to the metaphyseal cancellous
array, which distributes the force uniformly to the articular cartilage
(Fig. 21-3).
-
Changes in the articular cartilage
diminish its ability to absorb compressive loads, causing an alteration
to the subchondral bone. -
Changes in the structure of subchondral
bone can result in loss of structural support, exposing the cartilage
chondrocytes to forces exceeding their tolerance.-
In avascular necrosis (AVN), death of a
portion of the subchondral plate leads to a reactive zone of
granulation tissue in which osteoclastic activity leads to a loss of
the bony supporting arch for cartilage, often leading to collapse and
progressive arthritis. -
In osteoarthritis, subchondral bone stiffens; it is not known whether this is primary or secondary to cartilage change.
-
Summary
-
A diarthrodial joint can be considered a functioning unit, the integral elements of which are:
-
Articular cartilage
-
Synovium
-
Subchondral bone
-
Capsule/ligaments
-
Synovial fluid
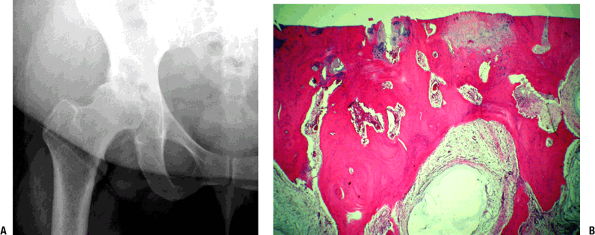
 Figure 21-3
Figure 21-3
Proximal femur. Specimen from a patient with renal osteodystrophy,
selected to show the bony structure: deformable cortex (which is
osteopenic) linked to cancellous array to subchondral plate. Mechanical
force from the cortex is linked to the cancellous trabeculae, which
form a honeycomb-like pattern of interconnected bony plates, passing
the force to the superiorly attached subchondral plate. Mechanical
force is thus transmitted uniformly to the acetabulum. Not shown is a
similar arrangement on the acetabular side where analogous cancellous
trabecular arcs pass the force through the ilium to the sacroiliac
joint. -
Any arthropathy will inevitably affect each of these elements.
-
Beyond these inherent elements, to
function appropriately, a joint requires motor support (e.g., skeletal
muscle with intact innervation). Primary muscle (muscular dystrophy,
inflammatory myopathies) and nerve disorders, both peripheral (nerve
injury, peripheral neuropathies) and central (stroke), may lead to loss
of motion and disordered joint function. In both instances, secondary
atrophic changes occur in all of the elements of the associated joints;
late degenerative disease may then follow. Disease in a joint leads to
loss of function. Pain, instability, and loss of range of motion then
lead to secondary changes in the supporting muscle groups, with disuse
atrophy and weakness.
-
Osteoarthritis
Definition
-
Osteoarthritis includes a heterogeneous
group of conditions of multifactorial etiology, characterized by joint
signs and symptoms, which are associated with defective integrity of
articular cartilage, in addition to related changes in the underlying
bone. -
Symptoms include aching, intermittent pain, stiffness, loss of motion, swelling (usually), and crepitus.
Epidemiology
Classification
Table 21-1 summarizes the classification of osteoarthritis.
Mechanisms of Joint Destruction
(Also see the introduction to this topic in Chapter 14.)
Pathologic Sequence
-
Hallmark finding (late): loss of
cartilage with underlying bony changes consisting of sclerosis of
subchondral bone, bone cysts, and osteophyte formation (Fig. 21-4) -
The growth zone adjacent to the tidemark
contributes to articular cartilage growth throughout life. Normal aging
changes the contours of joints. Remodeling can add length
(progressive), subtract from bone length (regressive), and add diameter
(circumferential).-
When the process is balanced, there is no disease.
-
When remodeling is unbalanced, the disease process of osteoarthritis occurs.
-
-
Early
-
Often focal, osteoarthritis starts with
superficial fibrillation and loss of small proteoglycans, decorin and
biglycan (usually associated with fibrils at articular surface), and
aggrecan (Fig. 21-5). -
Increased type II collagen cleavage occurs by action of collagenase, aggrecan cleavage, and degradation of small proteoglycans.
-
-
Intermediate
-
Continued loss of proteoglycans, with increased fibrillation down to subchondral bone (see Fig. 21-5)
-
Increase in number of degenerating chondrocytes, with cloning and regeneration of cells
-
-
Late (see Fig. 21-4)
-
Mechanical abrasion of degenerative
cartilage continues, with progressive loss of coverage so that the
surface becomes eburnated. The loss of cartilage allows increased force
to be transmitted to the subchondral plate. Increased bone formation
(Wolff’s law) then occurs, and over time the subchondral bone becomes
sclerotic. -
Subchondral cysts develop from two potential mechanisms:
-
Focal mesenchymal proliferation and accumulation of “stromal mucin” (myxoid change) in marrow fat
-
Surface cracks allow forced entry of joint fluid into the subchondral bone.
-
-
Proliferation of tufts of metaplastic
cartilage along denuded bone create a new, mechanically inferior,
cartilaginous joint surface (mechanism of “healing” seen after
realignment osteotomy). -
Cartilage fragments shed from surface may
grow as metaplastic cartilage in the joint space concentrically; this
may evoke secondary metaplastic cartilaginous change and mimic primary
synovial osteochon-dromatosis. -
Breakdown of cartilaginous fragments may induce an inflammatory response, including chondrogranu-lomas.
-
Hemorrhage of friable synovium may occur, producing hemosiderin staining of synovium over time.TABLE 21-1 Classification of Osteoarthritis
Type Association Presumed Mechanism Primary Genetic Subtle abnormalities of articular cartilage Secondary Mechanical Post-traumatic; post-slipped capital femoral epiphysis Damage to articular cartilage; joint incongruity Epiphyseal dysplasias Abnormal articular cartilage; abnormal joint shape Blount’s disease Mechanism not known Overuse Damage to articular cartilage Paget’s disease Joint incongruity; altered subchondral bone Joint instability (Ehlers-Danlos) Damage to articular cartilage Metabolic Ochronosis; gout; CPPD crystal deposition disease Abnormal metabolite (crystal) deposition in cartilage Hemochromatosis Not known Acromegaly Overgrowth of articular cartilage produces joint incongruity; cartilage may be abnormal. Postinflammatory Septic or inflammatory arthritis; hemophilia Destruction of articular cartilage ![]() Figure 21-4
Figure 21-4
Osteoarthritic hip. (A) Radiograph showing advanced osteoarthritis with
complete loss of surface cartilage, exposing dense subchondral bone.
Numerous cysts are present filled with amorphous myxoid material. The
bone adjacent to the cysts is sclerotic, representing altered
mechanical force transmission around the area of the cyst normally
filled with cancellous bone. (B) Photomicrograph showing advanced
osteoarthritis with complete loss of surface cartilage, exposing dense
subchondral bone. There is no definable articular cartilage present.
Numerous cysts are present filled with amorphous myxoid material; the
bone adjacent to the cysts is sclerotic, representing altered
mechanical force transmission around the area of the cyst normally
filled with cancellous bone.
-
P.423
Radiologic Findings (see Fig. 21-4)
-
The classic findings are of joint space
narrowing (reflecting loss of cartilage), subchondral sclerosis
(increased bone formation secondary to increased force across the joint
surface), osteophyte formation (progressive remodeling), and
subchondral cyst formation (mechanism controversial, secondary to break
in subchondral bone or arising as a result of metaplasia of subchondral
marrow in the environment of increased mechanical force).
Inflammatory Arthritis
Mechanism
-
Inflammatory arthritis is associated with specific recognition of antigenic triggers that are not fully known.
-
Specific immune response occurs via two
cellular mechanisms and requires initiation by antigen presenting
cells, which present antigens to the lymphocytes.-
Humoral immunity
is mediated by B lymphocytes, which produce antibodies (Ab) responsible
for recognition of foreign antigens, and their elimination with the aid
of complement. -
Cellular immunity is mediated by T lymphocytes, which recognize and eliminate intracellular antigens.
-
CD4 T cells (CD4 glycoprotein surface molecule) secrete cytokines.
-
CD8 T cells (CD8 glycoprotein surface molecule) kill antigen-bearing target cells.
-
This process is governed by the MHC region of genes (HLA genes).
-
-
Cytokines
-
A complex network of cytokines functions in initiating and perpetuating arthritis.
-
These polypeptide signaling substances
are produced by a host of cells, including synovial cells,
chondrocytes, monocytes, macrophages, T lymphocytes, B lymphocytes,
other connective tissue cells, and endothelial cells.
Rheumatoid Arthritis
Definition
-
Rheumatoid arthritis is an
immunologically mediated inflammatory disorder of synovial joints. It
can have systemic manifestations, characterized by multiple joint
involvement beginning with synovitis and later arthritis that can
become chronic and disabling.
Epidemiology
-
There is considerable variation of disease occurrence.
-
Highest prevalence (0.5% to 1%): Northern European, North American, some Native Americans
-
Intermediate prevalence (0.3% to 0.7%): Southern European
-
Low prevalence (0.1% to 0.5%): developing countries, rural Africa
-
-
Multifactorial disease
-
Genetic susceptibility
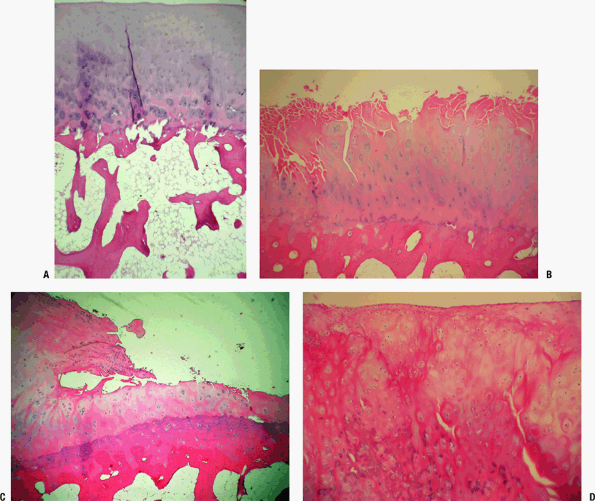 Figure 21-5
Figure 21-5
(A) Loss of surface regularity; cloning of the deeper cartilage cells;
loss of staining pattern of proteoglycans; subchondral plate shows
evidence of turnover with increased cement lines, and Howship’s
lacunae. (B) Further loss of surface regularity, and of proteoglycan
staining pattern; the development of fissures; thickening of
subchondral bone; cloning of chondrocytes in the basal layers. (C)
There is fragmentation of the surface cartilage, with a small piece
flaking off; the surface wear has left just a thin layer of articular
cartilage remaining; the subchondral bone has become a solid plate of
dense cortical bone. (D) The cartilage that flakes off can grow in the
synovial fluid and become a “joint mouse”; calcification of the
senescent cartilage in the loose body makes it radiographically
visible, and it may mimic synovial osteochondromatosis, a metaplastic
condition in which the synovial lining changes and islands of
cartilaginous differentiation develop in the joint lining. This image
shows the fibrocartilaginous surface of a joint mouse; the
darker-staining deeper areas represent calcification of the cartilage.
-
-
Environmental, lifestyle, and personal characteristics
-
Sex (female:male 2 to 3:1)
-
Age (onset fifth decade)
-
Positive factors associated with a reduced incidence
-
Hormonal (parity, pregnancy, oral contraceptives)
-
Dietary (fish, olive oil, cooked vegetables, omega-3 long chain polyunsaturated fatty acids)
-
-
Negative factors associated with an increased incidence
-
Smoking (increased incidence)
-
Infectious agents
-
Socioeconomic (low status worse)
-
Ethnic factors (higher prevalence in Northern Europe and North America)
-
-
P.424
P.425
Etiology
-
Susceptible individual (genetic and immunological predisposition) plus trigger:
-
HLA-DR4 and DW4 are most prevalent phenotypes (80%).
-
Trigger is unknown; possibilities are
environmental antigen (infection most likely) versus nonspecific local
stimuli (trauma, infection, allergic reaction, immunologic materials). -
For infectious model, Epstein-Barr virus or retrovi-ruses are most likely initiators.
-
Pathophysiology
-
Regardless of trigger, IgM is produced,
directed against altered autologous IgG; leukocytes phagocytose the
IgM/IgG complexes along with fibrin and complement; necrosis of these
cells induces release of lysosomal enzymes, which cause an acute
inflammatory reaction. -
Infectious trigger
-
Environmental antigen is attached to an
antigen presenting cell macrophage and presented to T cell (CD4
phenotype is most populous in rheumatoid arthritis pannus). -
CD4 + T cells produce numerous cytokines, the actions of which include:
-
Catabolic effects on cartilage
-
Activation of B cells, which differentiate into plasma cells producing IgG and IgM
-
-
-
Noninfectious trigger
-
Nonspecific stimuli induce local changes
that promote the differentiation of dendritic cells in the rheumatoid
synovium; these cells present an autologous Ag to the T cell, which
then initiate the disease process. -
Progression continues as T cells clone, resulting in recruitment of other effector cells.
-
Clinical Features
-
Diagnostic criteria are given in Box 21-2.
-
Typical: insidious malaise, fatigue,
weakness, and vague arthralgias for weeks, followed by joint pain,
swelling, morning stiffness of several joints, usually wrists, proximal
joints of fingers and toes, usually symmetric distribution; any other
major joint can be involved. -
Pauciarticular: One third of patients
have involvement of one or a few joints; then disease spreads to affect
other joints, usually symmetrically. -
Acute: Some patients may have an acute onset with fever and multiple swollen painful joints.
-
Box 21-2 Diagnostic Criteria for Rheumatoid Arthritis
Diagnosis
Physical Examination
Articular System
-
Early: warmth, tenderness, swelling with
boggy feel to synovium with cutaneous ruddy cyanotic hue; muscle
weakness and atrophy adjacent to involved joints -
Late: Loss of motion, flexion
contractures, ankylosis may occur with deformities of joints of hands
and feet (swan-neck, boutonniere, cock-up toes, ulnar deviation of
wrist and metacarpophalangeal bones). -
Spine: Cervical spine is often involved; lumbar spine is relatively spared.
-
When considering surgery on a patient
with RA, always rule out subluxation of the atlantoaxial joint; obtain
a flexion/extension lateral to show instability of this joint; can
produce compression of the spinal cord by the odontoid; look for
symptoms and signs of cord compression: bladder dysfunction, sphincter
laxity, circumanal hypesthe-sia, and long tract signs.
-
-
Popliteal (Baker’s) cyst, often present,
may rupture and mimic thrombophlebitis; both may coexist; investigation
(Doppler/ultrasound, or venogram) is indicated. -
Rheumatoid nodules (necrobiotic
granulomas) are present in 20% of patients—usually extensor surfaces of
elbows, fingers, and other areas subject to pressure.
Systemic and Unique Local Features
-
Pulmonary: diffuse interstitial fibrosis
-
Reticuloendothelial system: Splenomegaly occurs in 10%; regional lymphadenopathy may also develop.
-
Felty syndrome: rheumatoid arthritis +
splenomegaly + neutropenia; appears in patients with longstanding
disease; mechanism of neutropenia: antibodies to neutrophils,
attachment of immune complexes to neutrophils and subsequent
sequestration in spleen; marrow suppression -
Sjögren syndrome: Keratoconjunctivitis sicca (dry eyes + dry mouth + rheumatoid arthritis) occurs in ~15% of patients.
Synovial Fluid Aspiration
-
Milky turbid appearance; 20,000 to 50,000 inflammatory cells/mm3 with 50% polys; in infection, there are > 100,000 cells/mm3, of which >75% are polys.
Radiologic Findings
The radiological findings mirror the stage of the disease:
-
Periarticular soft tissue swelling
-
Bilateral symmetrical joint involvement
-
Uniform loss of joint space
-
Marginal erosions
-
Juxta-articular osteoporosis
-
Juxta-articular periostitis (not common, is more commonly associated with spondyloarthropathy)
-
Large pseudocysts
-
Joint deformity, including subluxation,
dislocation, articular bony destruction, bony ankylosis, complete
destruction of joint space
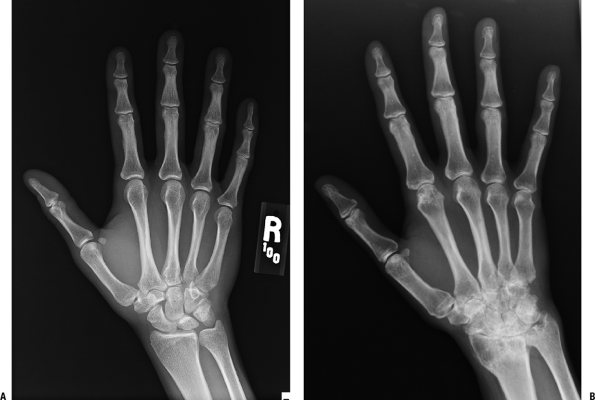
Figure 21-6 A shows the radiology of a normal hand and wrist. This should be carefully examined and compared to Figure 21-6B,
which shows the hand of a patient with rheumatoid arthritis, moderately
advanced in the wrist and mildly involved in the digits.
which shows the hand of a patient with rheumatoid arthritis, moderately
advanced in the wrist and mildly involved in the digits.
 |
|
Figure 21-6
(A) Radiograph of a normal hand shows the numerous small joints of the hand with normal bone density, discrete joint spaces, and normal contour of adjacent soft tissues. (B) Radiograph of a hand with rheumatoid arthritis shows widespread juxta-articular osteopenia and early erosion (irregularity of the cortical surface) of both sides of the index metacarpal head at the level of the joint recess. The carpal joints show loss of cartilage space and a mottled sclerosis of the carpal bones with discrete circular zones of decreased density suggesting cyst formation. There are numerous focal areas of cortical lysis. |
P.427
Pathologic Findings
-
Chronic inflammatory synovitis attacks any synovial surface (joints, bursae, tenosynovial sheaths).
-
Serum and synovial fluid contain
immunoglobulins (mostly IgM) produced as antibodies to autologous IgG
altered in some way in rheumatoid arthritis.-
These immunoglobulins in serum are known as rheumatoid factor (RF) and are present in 70% of patients.
-
Synovium
-
Nonsuppurative chronic inflammation of
synovium with hypertrophy and hyperplasia of the synovial lining cells;
the surface is papillary or villiform (Fig. 21-7) -
Scattered giant cells can be present among the synovial lining cells.
-
Inflammation consists of infiltration of
the synovial membrane by lymphocytes, plasma cells, and some mast
cells; hyperplastic lymphoid follicles and a fibrinous exudate are
present (see Fig. 21-7).-
Plasma cells often contain cytoplasmic eosinophilic inclusions of immunoglobulin (Russell bodies).
-
In acute phase, polymorphonuclear leukocytes predominate.
-
Vasculitis may be prominent.
-
-
Effects of synovial proliferation (Fig. 21-8)
-
Cartilage
-
Inflamed hypertrophied synovium (pannus)
creeps over articular cartilage, depriving it of nutrition and
enzymatically degrading the matrix; Figure 21-8 demonstrates these changes in articular cartilage even in the absence of an overlying pannus. -
With complete loss of cartilage, the end result is fibrous or bony ankylosis.
-
-
Ligaments/capsule
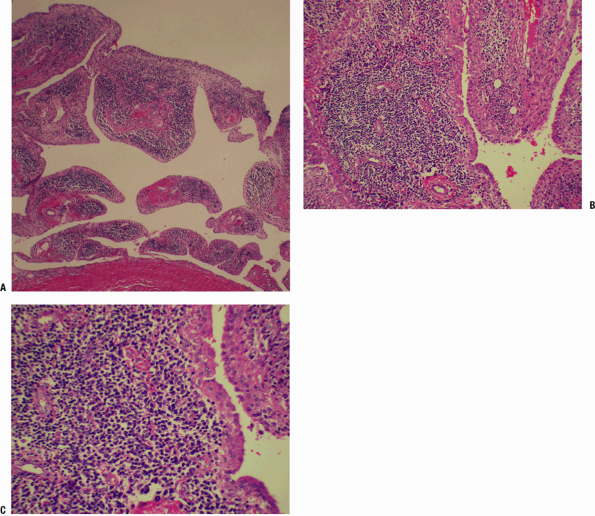 Figure 21-7
Figure 21-7
Rheumatoid arthritis. (A) Low-power view of nonspecific follicular
synovitis with several rounded oval dark lymphoid aggregates beneath
the synovial membrane. (B) Medium-power view showing the hyperplastic,
nodular lymphoid aggregates typical of rheumatoid synovitis. Multiple
lymphoid aggregates can produce a villonodular appearance to the
synovium. Inflammation can vary but often is quite pronounced and
accompanied by abundant fibrinous exudates (not evident in this field).
These exudates can fill the inflamed joint, presenting as “rice bodies”
at arthrotomy. The synovium is proliferative, appearing many cells
thick. (C) High-power field showing cells involved in the lymphoid
aggregate, a mixture of predominant lymphocytes, and a few plasma cells
and histiocytes.![]() Figure 21-8
Figure 21-8
Rheumatoid arthritis in subchondral bone. (A) The chronic inflammatory
process has reached the bone adjacent to the articular cartilage. Bone
has been deleted (osteoclastic removal associated with the active
hyperemia of the inflammatory process), and an inflammatory focus has
replaced the marrow and bone. This has the appearance of a rheumatoid
nodule with chronic inflammatory cells (type not apparent at this
power) surrounding a zone of fibrinoid necrosis. There is a collection
of fibrinous material in the joint space above. The articular cartilage
is affected; it appears porous and shows increased cloning of cells and
abnormal staining suggesting loss of proteoglycans. (B) View of severe
rheumatoid arthritis with pannus destroying the articular surface and
involving the medullary space. There is no recognizable articular
surface. Shards of subchondral bone, with divot-like irregularities in
the surfaces, Howship’s lacunae, indicative of clast activity. -
Inflammatory tissue mass (pannus) is
highly vascular, with an activated cellular mass producing cytokines as
well as clastic cells, which themselves produce cytokines and enzymes
producing tissue lysis. -
Destruction and weakening of the ligaments and capsule with resulting instability, subluxation, and dislocation of the joint
P.428 -
-
Subchondral bone
-
Subchondral bone is attacked by the pannus at the joint margins, producing radiographically apparent marginal erosions.
-
The active hyperemic flush pervades the
entire juxta-articular region, inducing clastic removal of bone and
explaining the juxta-articular osteopenia that accompanies inflammatory
arthritides. -
Vasculitis and inflammation can extend
throughout the subchondral bone, inducing lysis of articular cartilage
from below in addition to the effects of the pannus above (see Fig. 21-8).
-
Rheumatoid Nodule
-
Can be the first indication of rheumatoid
arthritis; locations: subcutaneous extensor surfaces, gastrointestinal
tract, lungs, heart, and any synovial membrane -
Associated with vasculitis
-
Irregularly shaped central necrobiotic
zone of necrotic fibrinoid material, surrounded by a zone of pallisaded
histiocytes and chronic inflammatory cells encircled by dense fibrosis;
vasculitis may be seen in surrounding tissue (Fig. 21-9)
Juvenile Inflammatory Arthritis/Rheumatoid Arthritis
Definition
-
A chronic inflammatory arthritis of
children similar to rheumatoid arthritis; often less severe, but with
analogous radiologic and histologic findings
Diagnosis
Clinical Features
-
Presentation varies:
-
25% systemic presentation with self-limited course
-
Male > female
-
Median age of onset 5 years
-
High fever, nonpruritic erythematous macular or maculopapular rash in association with rash
-
Reticuloendothelial system involvement with lymphadenopathy, hepatosplenomegaly, pneumonitis, and pleuritis
-
Cardiac involvement with pericarditis and/or myocarditis (rarely)
-
25% of patients have severe, destructive polyarthropathy.
-
Seronegative and antinuclear antibody (ANA) negative
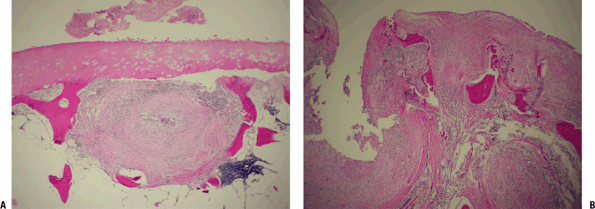
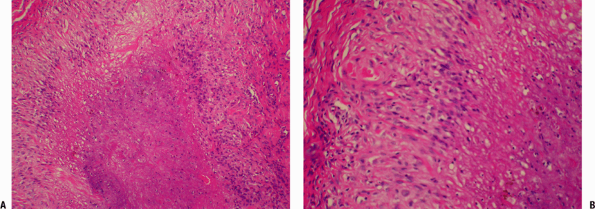 Figure 21-9
Figure 21-9
Rheumatoid nodule. (A) Close-up view of palisades of histiocytes of the
rheumatoid nodule surrounding a central zone of amorphous fibrinous
material. On the far left and right are spindle cells within the
fibrous capsule. (B) Close-up of the transition: capsule on the left,
palisading histiocytes in the middle, central zone of necrosis on the
right. This picture of a rheumatoid nodule might be confused with a
caseating granuloma of tuberculosis. Mycobacterial and other infectious
granulomas generally have multinucleated giant cells and caseous
necrosis (tends to appear more granular than the fibrinoid necrosis
seen here).
-
-
-
30% pauciarticular disease
-
Female > male
-
Involvement of one or more joints, usually knees or ankles
-
Mild arthritis, excellent prognosis
-
Eye involvement is a problem, with 50% of
patients having chronic iridocyclitis, which may progress to band
keratopathy, cataracts, and loss of vision. -
RF negative
-
ANA present in 50% to 60%
-
-
25% polyarthritic without systemic disease with good prognosis
-
Insidious onset, in small joints of hands and feet
-
Mainly affects young females (median onset 2 years)
-
RF negative
-
25% are ANA positive.
-
Special techniques demonstrate IgG and IgA rheumatoid factors in more than half.
-
-
5% to 10% polyarticular with positive RF
-
Female > male
-
Median age at onset is 12.
-
Analogous to adult rheumatoid arthritis in clinical presentation and course.
P.429
Juvenile inflammatory arthritis/Ankylosing Spondylitis
-
May also present in childhood
-
Represents 20% of children who present with juvenile-onset arthritis
-
Male > female
-
Median onset 10 years
-
Present with early hip girdle symptoms
-
Often iridocyclitis
-
RF and ANA negative
-
HLA-B27 positive in 75%, as compared to negative finding in other types of childhood arthritis
Spondyloarthropathies
The spondyloarthropathies are a heterogeneous group of
inflammatory diseases characterized by spinal and peripheral joint
oligoarthritis, inflammation of the attachments of ligaments and
tendons to bones (enthesitis), and, at times, mucocutaneous, ocular,
and/or cardiac manifestations.
inflammatory diseases characterized by spinal and peripheral joint
oligoarthritis, inflammation of the attachments of ligaments and
tendons to bones (enthesitis), and, at times, mucocutaneous, ocular,
and/or cardiac manifestations.
-
They include:
-
Ankylosing spondylitis
-
Reactive arthritis, known previously as Reiter’s syndrome
-
Psoriatic arthritis
-
Enteropathic arthritis (related to inflammatory bowel diseases, ulcerative colitis, or Crohn’s disease)
-
Juvenile spondyloarthritis
-
Undifferentiated spondyloarthropathies
(patients expressing elements but failing to fulfill accepted criteria
for any of the above)
-
-
Pathogenesis involves an inherent “early
warning system” of the synovium that is acutely responsive to
deposition of immunogenic fragments. This involves a humorally mediated
process that initiates a cascade. -
Offending fragments could be immune complexes, mRNA/DNA from the initiating organism, or actual fragments of the microbe.
-
The process of crossing vessel walls and
synovial membrane can be assisted by adhesion molecules (integrins and
selectins) on white blood cells carrying ingested pathogens or parts.-
P-selectin shows affinity for attachment
to synovium; may act as bridge allowing pathogenetic material into the
cells, precipitating the synovial process.
-
-
Presented with this immunogenic stimulus,
antigen-specific CD4 T cells, synovial intimal lining macrophages, and
B cells all participate in the production of keycytokines (tumor
necrosis factor alpha [TNF-α], interleukins [ILs]), which initiate the
inflammatory process.
P.430
Ankylosing Spondylitis (Rheumatoid Spondylitis, Marie-Strumpell Disease)
Definition
Ankylosing spondylitis is a chronic and usually
progressive inflammatory disease involving the spine and adjacent soft
tissues, sacroiliac joints almost always, hips and shoulders often, and
other joints less frequently.
progressive inflammatory disease involving the spine and adjacent soft
tissues, sacroiliac joints almost always, hips and shoulders often, and
other joints less frequently.
Epidemiology
-
Male:female, 3 to 10:1
-
Concordance rate in identical twins as high as 63% (compared to 23% in nonidentical twins)
-
Histocompatibility testing shows HLA-B27 in >90% of patients versus 7% in the normal white population.
-
Less frequent incidence of ankylosing spondylitis and HLA-B27 in blacks
-
Prevalence of ankylosing spondylitis 1% in white population
-
Ankylosing spondylitis is a disorder of the young, with onset in second or third decades.
-
20% of people who carry HLA-B27 will develop ankylosing spondylitis.
Diagnosis
Clinical Features
-
Constitutional symptoms are mild in the absence of advanced disease.
-
Spine
-
Low back pain with stiffness, worse in morning; nocturnal back pain
-
Mimics sciatica in 10%
-
No evidence of nerve root irritation or compression initially
-
Progressive rigidity of the spine
-
Atlantoaxial subluxation and spinal cord compression less common than in rheumatoid arthritis
-
Cervical spine that has undergone multiple-level fusion is susceptible to fracture.
-
-
Peripheral joints
-
25% of patients develop peripheral arthritis.
-
Hips can be severely involved with arthritis indistinguishable from osteoarthritis.
-
-
Knees and heels may be involved.
-
Anterior chest pain and pleuritic pain
may occur (inflammation at insertion sites of costosternal and
costovertebral muscles and at manubriosternal and sternoclavicular
joints).
-
-
Extra-articular manifestations
-
Ocular: Acute anterior uveitis (20% to 30%) may be initial indication of ankylosing spondylitis.
-
Cardiac: aortic valve incompetence (3%), may require replacement; cardiac conduction defects, may require pacemaker
-
Pulmonary: apical pulmonary fibrosis, chronic productive cough, and dyspnea
-
Renal: Amyloidosis may cause uremia.
-
Osseous: osteoporosis
-
Neurological: cauda equine syndrome
(rare) late complication, possibly due to arachnoiditis or ischemia;
slowly progressive; treatment problematic
-
Pathologic Findings
-
Diarthroidal joints
-
Synovitis resembling rheumatoid arthritis
begins in the apophyseal joints of the spine, costovertebral joints,
and sacroiliac joints. -
Same pathological findings in the hip, shoulder, and other peripheral joints
-
Bony erosions and cartilage destruction ensue, followed by ankylosis.
-
-
Amphiarthrial joints
-
Intervertebral discs, manubriosternaljoint, and symphysis pubis are invaded by granulation tissue.
-
The cartilage is destroyed, and local fibrosis and then ankylosis occur.
-
Radiologic Findings (Figs. 21-10 and 21-11)
-
Vertebral bodies have squared margins (marginal erosion) or central cyst suggesting infection.
-
Syndesmophytes at margins of vertebral
bodies produce “bamboo spine” appearance (ossification of outer layers
of annulus fibrosis); differential diagnosis with diffuse idiopathic
skeletal hyperostosis: lack of ossification of anterior and posterior
longitudinal ligaments in ankylosing spondylitis (see Fig. 21-11) -
Diffuse osteoporosis
-
Fusion of sacroiliac joints
-
Erosions and ossification of insertions
of ligaments, tendons, and capsules into bone (spinous processes,
greater trochanters, pelvis, and heels) -
Involvement of peripheral joints resembles osteoarthritis late.
Reactive Arthritis
Definition
-
An arthritis that is preceded by an immunologic sensitization
-
The term “Reiter’s syndrome” applies only to the classic triad of arthritis, urethritis, and conjunctivitis.
Classification
For some organisms, the link between infection in locations other than the joints is clear. For others an association
P.431
exists, but the role of the offending organism as a triggering agent of
reactive arthritis is not well established; these organisms are marked
with (?) in the classification.
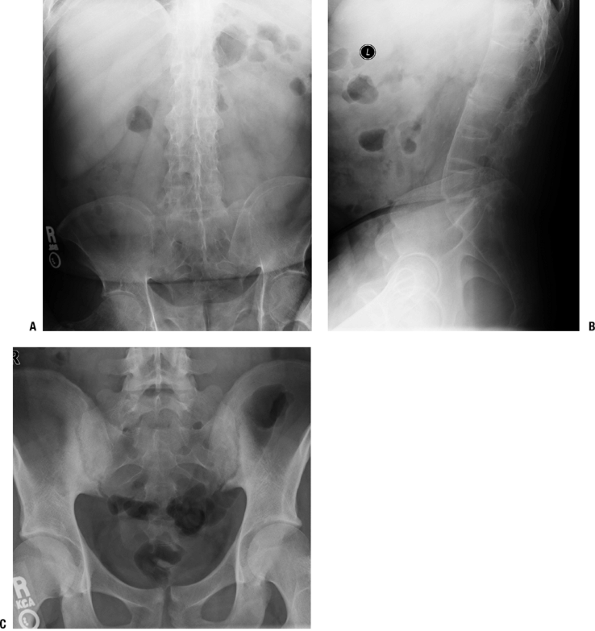 |
|
Figure 21-10
Ankylosing spondylitis. (A) Anterior-posterior view of lumbar spine shows ankylosis of the sacroiliac joints and the lumbar spine. There is joint space narrowing of the hip joints indicative of early secondary osteoarthritis. (B) Lateral view of the lumbar spine demonstrating ossification of the anterior longitudinal ligament, preservation of the disc spaces, and osteopenia of the vertebral bodies, the margins of which are “squared off.” (C) A 27-year-old man with low back pain and ankylosing spondylitis. Anterior-posterior view of the pelvis shows erosions and sclerosis in a bilateral, symmetrical distribution typical of sacroiliitis of ankylosing spondylitis. |
-
Urogenic with bacterial associations
-
Chlamydia trachomatis
-
Mycoplasma genialium (?)
-
Ureaplasma urealyticum (?)
-
Neisseria gonorrhoeae (?)
-
-
Enterogenic with bacterial associations
-
Salmonella (various types)
-
Shigella (various types)
-
Yersinia (various types)
-
Campylobacter (various types)
-
Clostridium difficile
-
-
Respiratory tract associated with bacterial associations
-
Beta-hemolytic Streptococcus (?)
-
Chlamydia pneumoniae
-
-
Idiopathic: typical clinical presentation without a clear association with an established infection
-
The idiopathic group becomes smaller as more and more new associations are being reported.
-
Epidemiology
-
Risk of reactive arthritis in an enteric infection caused by gram-negative microbes is 1% to 15%.
-
HLA-B27 is detected with 5 to 10 times greater frequency in reactive arthritis patients than in the general population.
-
Patients are usually young adults (mean age 30 to 40); disease is uncommon in children.
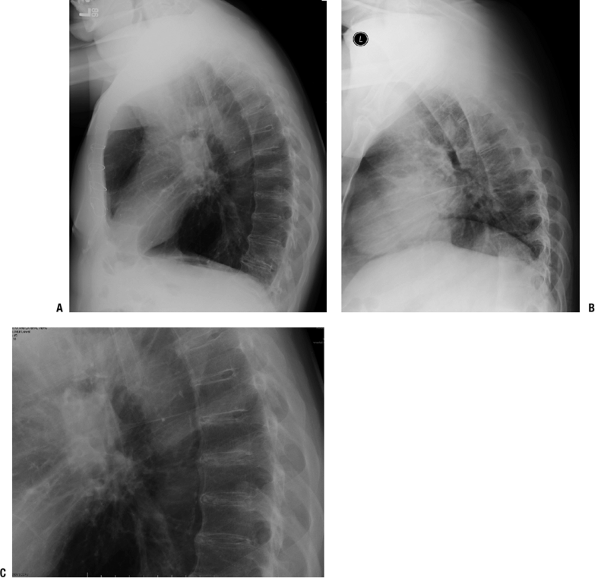 Figure 21-11
Figure 21-11
Diffuse idiopathic skeletal hyperostosis (DISH). (A) A 73-year-old man
with typical manifestations of DISH in the thoracic spine. Lateral view
of the thoracic spine shows anterior ossification extending over more
than four contiguous vertebral bodies. The ossification is separated
from the vertebral bodies by a lucency, indicating the density is in
the anterior longitudinal ligament and soft tissues anterior to it. (B)
Differential diagnosis: DISH versus ankylosing spondylitis. In this
lateral view of ankylosing spondylitis, the anterior ossification
merges with the vertebral body. The disc spaces are preserved. In DISH
there is extensive calcification of the disc space, as is shown in the
magnified view in (C).
P.432
Pathophysiology
-
Organisms or bacterial antigens disseminate to the joint, causing inflammation.
-
CD4 + T-cell response drives and supports the arthritic process.
-
Persistence of the microbial structures may be a function of:
-
Impaired elimination by a deficient cytokine reaction, such as poor T-helper response
-
Defective or aberrant function of HLA B-27
-
Diagnosis
Clinical Features
-
Onset of arthritis is 1 to 4 weeks after infection; triggering infection may be asymptomatic.
-
Asymmetric oligoarthritis, often large
joints of lower extremities; 50% have arthritis in upper limbs;
occasionally present in small joints of hands or feet -
Associated features
-
Enthesitis/bursitis may be associated with arthritis or may be the initial presentation.
-
Eye findings are conjunctivitis (usually) or acute anterior uveitis (less frequently).
-
Skin rashes can present with several forms.
-
Nail changes may mimic those of psoriasis.
-
30% have spondylitis with acute low back pain and buttock radiation.
-
-
Duration of the acute arthritis is a few months; in a small percentage, the symptoms become intermittent or chronic.
-
There are no specific radiologic or pathologic findings; the findings are analogous to mild rheumatoid arthritis.
-
P.433
Psoriatic Arthritis
Definition
-
Psoriatic arthritis is a unique
inflammatory arthritis associated with psoriasis; the pathology is
analogous to that of rheumatoid arthritis.
Epidemiology
-
Prevalence of psoriasis is 2% to 3%; prevalence of inflammatory arthritis among patients with psoriasis is ~30%.
-
Genetic epidemiology suggests a unique status:
-
Psoriatic arthritis is associated with HLA class 1 al-leles, rheumatoid arthritis with class 2 alleles.
-
-
Patients with psoriatic arthritis are at increased risk of death; standardized mortality ratio of 1.62.
Diagnosis
Clinical Features
-
The longer the duration of the psoriatic arthritis, the more it tends to become polyarticular.
-
Arthritis may precede rash by many years.
-
Five clinical patterns most relevant at time of disease onset
-
Distal predominant pattern
-
Oligoarticular asymmetrical
-
Polyarticular rheumatoid arthritis-like:
20% of patients over time develop a destructive arthritis; much more
aggressive than previously thought. -
Spondylitis
-
Arthritis mutilans
-
-
The features of psoriatic arthritis and rheumatoid arthritis are compared in Table 21-2.
-
Pathology of psoriatic arthritis is analogous to that of rheumatoid arthritis.
Enteropathic Arthritis (Associated with Ulcerative Colitis and Crohn’s Disease)
-
Enteropathic arthritis is a form of
chronic, inflammatory arthritis associated with the occurrence of
inflammatory bowel disease, usually ulcerative colitis or Crohn’s
disease.Table 21-2 Comparison of Psoriatic Arthritis and Rheumatic ArthritisFeature Psoriatic Arthritis Rheumatoid Arthritis Male:female ratio Equal distribution More common in women Hand involvement Distal joints; arthritis mutilans (resorption of phalanges, metacarpals, or metatarsals) Metacarpophalangeal/proximal interphalangeal Ray involvement All joints of a single digit affected (on just one side, asymmetrical) Same joints affected in both hands (symmetrical) Erythema of affected joints More Less Spinal involvement Paravertebral ossification in lower thoracic, and lumbar regions, plus sacroiliac joints (40% have spondylitis) Cervical spine most affected Enthesitis Yes No Tenderness Mild More severe Rheumatoid nodules No Yes Rheumatoid factor 13% >80% Radiologic features Shortening of digits because
of bone lysis, most severe form being telescoping of digits; bony
fusion may occur, but usual feature is pencil-in-cup deformity;
periosteal reactions; dactylitis (inflammation of entire digit)
inflammation affecting both joints and tendonsEarly: soft tissue swelling,
juxta-articular osteopenia. Later: marginal erosions, joint space
narrowing. Advanced: destruction of subchondral bone, diffuse
osteoporosis, subluxation dislocation.Extra-articular features Are those of the spondyloarthropathies: mucous membrane lesions, iritis, urethritis, diarrhea, aortic root dilatation, HLA-B27 Occur more often in men than
women, late (“burned out”), in seropositive patients with more severe
joint disease: include rheumatoid nodules, pleural and pulmonary
inflammation leading to fibrosis, pericarditis, Sjogren syndrome (dry
eyes, dry mouth, peripheral neuropathy, rheumatoid arthritis), Felty
syndrome (rheumatoid arthritis, splenomegaly, granulocytopenia)In general, severity of skin changes parallels the severity of the arthritis. -
The peak age of incidence is 15 to 35.
-
About one in five people with Crohn’s or
ulcerative colitis will develop enteropathic arthritis; the arthritis
can be peripheral or axial.
P.434
Peripheral
-
10% to 20% of patients with inflammatory bowel disease
-
Male:female 1:1
-
HLA-B27 association uncommon
-
Coincides with inflammatory bowel disease activity
-
Mainly affects lower limbs
-
Asymmetric migration, relapsing oligoarthritis
-
Lasts 2 to 6 weeks
-
Radiographic changes rare
Axial
-
2% to 7% of patients with inflammatory bowel disease
-
Male > female for spondylitis
-
HLA-B27 association 50% to 70%
-
Independent of inflammatory bowel disease activity; often precedes inflammatory bowel disease symptoms
-
Gradual onset of low back pain
-
Morning stiffness
-
Pain worse with prolonged standing or sitting
-
Pain improved by moderate activity
-
Lasts <6 months
Undifferentiated Spondyloarthropathy
-
An arthritis that lacks the specific
features listed above for the other types of spondyloarthropathy is
considered undifferentiated.
Metabolic
Pathophysiology of Crystal Arthropathies
-
Monosodium urate and calcium pyrophosphate dihy-drate (CPPD) crystals are responsible for:
-
Acute synovial inflammation
-
Interaction between crystals and synoviocytes (types A and B, infiltrating leukocytes)
-
Secretion of pro-inflammatory agents
(IL-lβ, TNF-α), and chemotactic factors (IL-8), leading to neutrophil
invasion, activation, and further inflammatory response
-
-
Cartilage degradation
-
Bone lesions within the joint
-
-
Cascades of activated proteins involve
cytoplasmic membrane-related proteins (FAK complex, Src family tyrosine
kinases), but also MAPK and NF-kB pathways, leading to nitric oxide,
prostanoid, and cytokine production, and protease activation.
Gout
Definition
-
Inflammatory arthritis triggered by
crystallization of uric acid within involved joints, characterized
clinically by attacks of acute inflammation, and often associated with
hyperuricemia
Epidemiology
-
Prevalence <2% in men >30 years of
age and in women >50 years of age; increased to 9% in men and 6% in
women >80 years of age -
Incidence of primary gout (no diuretic
exposure) has doubled over the past 20 years, probably due to the
increasing incidence of obesity and other lifestyle factors.
Pathophysiology
Role of Urate Levels
-
Uric acid exists as soluble urate at physiologic pH but precipitates at concentrations >8.0 ng/dL.
-
There is a direct positive association between serum urate levels and a future risk of gout.
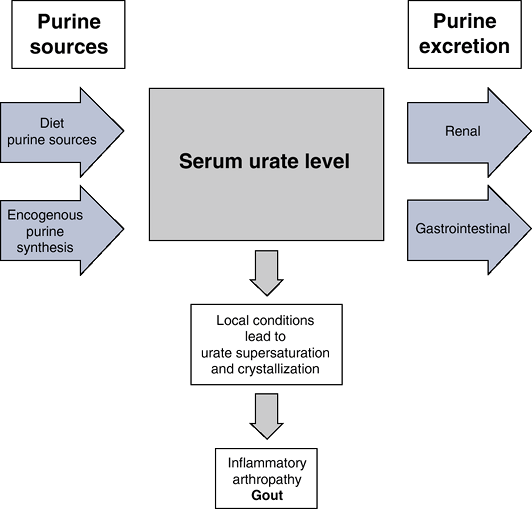
Urate Metabolism
-
Urate balance depends on changes in diet, synthesis, and rate of secretion (Fig. 21-12).
-
Dietary effects
-
Red meat, seafood, beer (+ +), liquor (+), and other sources: all increase serum level of uric acid
-
Vitamin C has a uricosuric effect.
-
-
Increased synthesis can be primary or secondary.
-
Primary hyperuricemia (rare): due to an
inherited error of metabolism resulting from an enzymatic defect of
purine synthesis and/or in the renal excretion of uric acid -
Secondary hyperuricemia: nongenetic
disorders that increase the production of uric acid or that decrease
the excretion of uric acid-
Increased cell turnover in proliferative and inflammatory disorders (hematologic cancer and psoriasis)
-
-
-
Excretion of urate is predominantly through the kidneys, less through gastrointestinal tract.
Diagnosis
Clinical Features
Acute Gout
-
Usually monoarticular and, in the lower extremity, often the metatarsophalangeal joint of the great toe
-
Characterized by severe pain, swelling, low-grade fever, and leukocytosis
-
Precipitating events include trauma, recent surgical procedure, intercurrent illness.
Intercritical Gout
-
Long asymptomatic periods occur, despite increased urate levels
![]() Figure 21-12
Figure 21-12
Gout is the result of supersaturation and crystallization of uric acid
within the joints. The amount of urate in the body depends on the
balance between dietary intake, synthesis, and excretion. Hyperuricemia
results from the overproduction of urate (10%), underexcretion of urate
(90%), or often a combination of the two.
P.435
Chronic Gout
-
Eventually widespread crystal deposition occurs throughout the body, particularly in the kidneys and adjacent to joints.
Radiologic Findings
-
Articular distribution of soft tissue
swelling, marginal punched-out lesions with overhanging edges, with no
regional osteopenia or reactive sclerosis (Fig. 21-13)
Pathologic Findings
-
To preserve crystals for identification,
fixation in alcohol, hand processing, and using unstained sections are
preferred over formalin fixation (urate crystals are water-soluble). -
Synovial fluid (see Fig. 21-13)
-
Inflammatory exudate mimics infection.
-
Examination under polarized light reveals needle-like crystals with a strong negative birefringence.
-
Often crystals lie within polymorphonuclear leukocytes.
-
Synovium (see Fig. 21-13)
-
Inflammation, crystals lying within leukocytes
-
Tophi appear as granulomatous deposits of
amorphous, proteinaceous material, bordered by fibrous tissue,
histiocytes, and multinucleated giant cells.
-
Cppd Crystal Deposition Disease
Definition
-
Joint disorder induced by the deposition
of calcium pyrophosphate dihydrate (CPPD) crystals in hyaline
cartilage, fibrocartilage, and other soft tissue structures,
characterized by acute, subacute, or chronic joint inflammation -
Box 21-3 covers some terminology.
Epidemiology
-
Frequency is 1:1,000.
-
Rare before age 30; incidence increases with age, approaches 50% in patients >80
-
Female > male
Classification
-
Sporadic: middle-aged to elderly patient, with no sexual predominance
-
Hereditary: female predominance, occurs at an early age, more severe arthropathy
-
Secondary: hyperparathyroidism, hypothyroidism, gout, hemochromatosis, hypomagnesemia, hypophosphatasia
Diagnosis
Clinical Features
-
Six patterns have been described (Table 21-3).
Radiologic Findings
-
Calcification is seen in fibrocartilage, articular cartilage, capsule; appearance is punctuate or linear (Fig. 21-14).
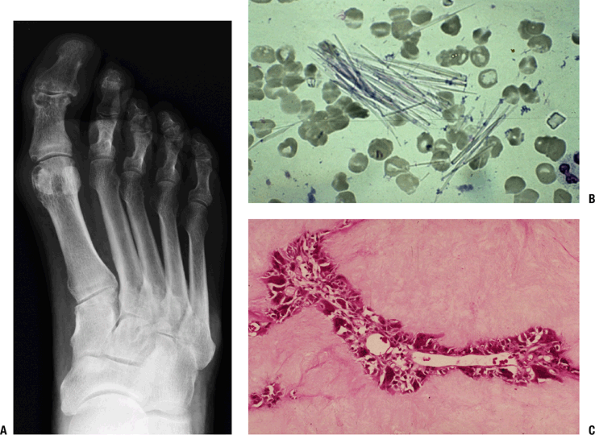 Figure 21-13 Gout. (A)
Figure 21-13 Gout. (A)
A 58-year-old man with pain in the great toe and gout.
Anterior-posterior view of the foot shows soft tissue swelling at the
medial aspect of the first metatarsophalangeal and interphalangeal
joints. Well-corticated erosions are seen at the medial aspect of these
articulations as well. Note the relative preservation of joint space
and mineralization. (B) Uric acid crystals. Synovial fluid sample contains needle-like crystals of uric acid. (C)
Inflammatory reaction in gout. Surrounding and within a deposit of
crystalline material is a thin layer of mononuclear and giant cells. A
small vessel courses through this reaction. -
Figure 21-13
Gout. (A) A 58-year-old man with pain in the great toe and gout.
Anterior-posterior view of the foot shows soft tissue swelling at the
medial aspect of the first metatarsophalangeal and interphalangeal
joints. Well-corticated erosions are seen at the medial aspect of these
articulations as well. Note the relative preservation of joint space
and mineralization. (B) Uric acid crystals. Synovial fluid sample
contains needle-like crystals of uric acid. (C) Inflammatory reaction
in gout. Surrounding and within a deposit of crystalline material is a
thin layer of mononuclear and giant cells. A small vessel courses
through this reaction.Table 21-3 Patterns of Cppd Deposition DiseasePattern Prevalence Description Asymptomatic 10% to 20% Osteoarthitis 35% to 60% Chronic, progressive,
frequently bilateral and symmetrical, with distribution in knee, hip,
metacarpophalangeal, elbow, ankle, wrist, and glenohumeral joints;
flexion contractures of knee and elbow are commonPseudogout 10% to 20% May occur spontaneously or in
association with trauma, concomitant medical illness (stroke,
myocardial infarction, surgery, transfusion, intravenous fluid
administration, institution of thyroxine replacement therapy, and joint
lavage); mimics gout or infection with pain (less severe than gout),
erythema, tenderness, and raised erythrocyte sedimentation rate;
attacks are self-limited (duration days to weeks)Rheumatoid arthritis 2% to 6% Morning stiffness, fatigue,
symmetric polyarthropathy, with synovial thickening, loss of range of
motion, raised erythrocyte sedimentation rate; duration weeks to monthsCharcot joint <2% Rapid, progressive destruction of large joint Rare presentations Ankylosing spondylitis, rheumatic fever, and trauma ![]() Figure 21-14
Figure 21-14
Knee with CPPD crystal deposition disease. Calcification of menisci,
capsule, and articular cartilage accompanied by early osteoarthritic
change (medial osteophyte formation).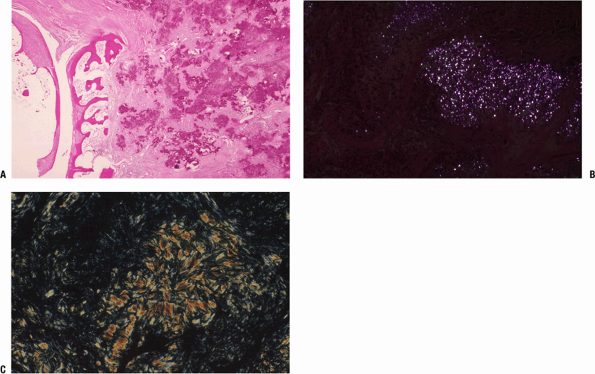 Figure 21-15
Figure 21-15
(A) Morphology of CPPD crystal deposition disease. Deposition of CPPD
crystals in capsule adjacent to cortex, joint recess, and articular
surface. (B) Photomicrograph of section using polarized light;
refractive characteristics of rhomboidal CPPD crystals are shown. (C)
For comparison, this section using polarized light shows the
characteristics of uric acid crystals. -
May also be seen in symphysis pubis and intervertebral discs
P.436
P.437
P.438
Box 21-3 Cppd Crystal Deposition Disease Terms
|
Pathologic Findings
-
Gross: chalky white deposits
-
Micro (Fig. 21-15):
In vascularized tissue, basophilic calcific deposits are surrounded by
chronic inflammatory and giant cell reaction; in nonvascularized
tissue, the reaction is absent. -
Crystal identification (see Fig. 21-15): rhomboidal-shaped crystals; weakly positive birefringence; crystals are best seen with polarized light on unstained sections.
Ochronosis
Definition
-
Ochronosis produces darkening of the
connective tissues of the body and is caused by brown-black pigment
composed of excess homogentisic acid (HGA) in patients with
alkaptonuria. -
Musculoskeletal involvement leads to a severe arthropathy.
-
Alkaptonuria: rare, autosomal-recessive
disorder (prevalence < 1:250,000 in most populations, except
Dominican Republic and the Piestany region in Slovakia), characterized
by the inability to metabolize HGA.
Metabolism
-
Dietary phenylalanine and tryrosine are metabolized in liver and kidneys through HGA to acetoacetic acid and fumaric acid.
-
In alkaptonuria, the enzyme homogentisic
acid oxidase, a liver enzyme, is missing, allowing HGA to accumulate as
alkapton, a black pigment; in urine, alkapton turns black on exposure
to air; other connective tissues become progressively darkly stained
(skin, sclerae, intervertebral discs, articular cartilage). -
Mechanism of arthropathy is not clear.
Genetics
-
In somatic cell hybrids, HGA is located on chromosome 3, mapping to 3q21–q23 by fluorescent in situ hybridization.
Diagnosis
Clinical Features
-
General: a progressive degenerative
arthropathy, mainly affecting axial and weight-bearing joints,
associated with extra-articular manifestations -
Spine: Insidious low back pain and
stiffness begin in the 30s; occasionally an acute disc herniation
provides the initial event leading to diagnosis. -
Peripheral joints: Knees, hips, and
shoulders are most commonly involved; the usual presentation is of an
aggressive osteoarthritis; occasionally, the process produces acute
inflammation; small joints of the hands and feet are rarely involved. -
Tendinopathy: The tendons also become
sites of alkapton deposition; tendinitis, rupture, with tendon
calcification visible radiographically; Achilles tendon, rotator cuff
are most commonly involved. -
Extra-articular manifestations:
Pigmentation of sclerae and external ears allows early clinical
diagnosis; alkap-ton pigment deposition in heart valves may lead to
dysfunction, failure, and surgical replacement; stones may form in
prostate and kidneys.
P.439
Radiologic Findings
-
Spine: marked degenerative disc disease
(disc space narrowing, endplate sclerosis, and osteophytosis), with
excessive calcification of multiple intervertebral discs -
Peripheral joints: similar to osteoarthritis
Pathologic Findings
-
Gross: brown-black discoloration of cartilage and disc
-
Micro: disorganization similar to Charcot
joint, with collections of brown-black shards of cartilage surrounded
by fibrosis, histiocytic reaction, and chronic inflammation
Hemophilia
Definition
-
Hemophilic arthropathy produces a form of secondary osteoarthritis, the result of repeated hemorrhage into a joint.
Diagnosis
Clinical Features
-
Acute hemorrhage: may be precipitated by
minor trauma; typically involves knees, elbows, and ankles;
presentation is of a hot, swollen, painful joint -
Chronic arthritis: a form of secondary osteoarthritis primarily affecting knees, elbows, ankles
Radiologic Findings
-
Acute: joint effusion, soft tissue swelling
-
Chronic: narrowed joint space, loss of
cartilage, marginal erosions, multiple juxta-articular cysts, and
progressive osteophyte formation; regional osteopenia (Fig. 21-16)
Pathologic Findings
-
Chronic: Gross finding is dark-brown
(mahogany) staining (from hemosiderin), with papillary proliferation of
the synovial lining; hemosiderin staining within the synovium as well
as the subsynovial tissue, which also shows chronic inflammation and
fibrosis.![]() Figure 21-16
Figure 21-16
Hemophilia. A 34-year-old man with knee pain and hemophilia.
Anterior-posterior view of the knees shows enlargement of the distal
end of the femur and the proximal end of the tibia in response to
hyperemia during development of the arthrosis. Severe joint space
narrowing and secondary degenerative changes are seen. An erosion is
present at the medial aspect of the left medial femoral condyle.
Circulatory Disorders Affecting Joints
Neuropathic Arthropathy (Charcot Joint)
Definition
-
Neuropathic arthropathy is a chronic,
progressive disease of bone, characterized ultimately by severe loss of
joint architecture in a patient who has a neuropathy affecting the
extremity. -
It can be rapidly progressive (within weeks), painful or painless, and can occur without weight bearing.
Epidemiology
-
Distribution of joint involvement
-
*Diabetes: interphalangeal, metatarsophalangeal, and tarsal joints
-
*Tabes dorsalis: knees, hips, ankles, and lumbar spine
-
*Syringomyelia: shoulders, elbows, and cervical spine
-
-
Other conditions associated with
neuropathic arthropathy include *meningomyelocele, *congenital
insensitivity to pain, *leprosy, and *peripheral nerve injuries.
P.440
Etiology
-
Idiopathic: enigmatic association with indomethacin administration and intra-articular steroid injection
-
I Neuropathic: See the conditions listed above under Epidemiology (marked with *).
Pathophysiology
-
The primary abnormality is a loss of
vasomotor control in the region of a joint, secondary to the underlying
neurologic disorder. -
The sequence then is:
-
A loss of coordination between arteriolar inflow and venous outflow occurs, with a marked increase on the arteriolar side.
-
Osteoclastic deletion of bone occurs in response to the high-flow state.
-
Juxta-articular osteopenia profoundly
weakens the sub-chondral bone, leaving the articular cartilage without
adequate bony support. -
Fractures occur, liberating shards of bone and cartilage into the joint.
-
Aberrant articular cartilage incites a
profound inflammatory response in soft tissue; a positive feedback
cycle then ensues with further propagation of inflammation. -
Inflammatory response also drives the hyperemic state, leading to further deletion of bone.
-
Repair is attempted with new bone
formation about the joint and reinforcement of existing bone structures
in the presence of ongoing weight bearing; this leads to visible
periosteal reaction about the joint and sclerosis of existing
juxta-articular bone.
-
Diagnosis
Differential Diagnosis
-
Acute: cellulitis, acute gout, thrombophlebitis, osteomyelitis
-
Chronic: osteomyelitis (may coexist with neuropathic arthropathy, especially in the presence of ulceration)
-
Neuropathic neuropathy can mimic a neoplastic process clinically and radiographically.
Clinical Features
-
Insidious presentation, usually in one joint, then progressing to others; an acute and painful onset suggests fracture
-
Key clinical finding is that the degree of discomfort is mild compared to the structural abnormality.
-
Erythema, local warmth, swelling, some tenderness, crepitus, instability, deformity
Radiologic Findings
-
Plain films demonstrate the gross anatomy
but are neither sensitive nor specific to separate neuropathic
arthropathy from infection. Figure 21-17 shows neuropathic arthropathy of the foot in a diabetic.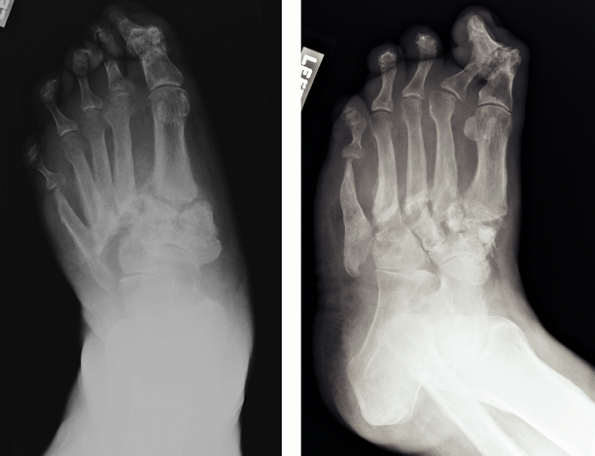 Figure 21-17
Figure 21-17
Neuropathic foot. Two radiographic views of the foot of a patient with
polyneuropathy related to diabetes show extensive destruction of
several joints. There is a loss of normal architecture of the midfoot
with lateral subluxation of all of the metatarsals, subchondral
sclerosis of the periarticular surfaces, as well as adjacent deposition
of new bone. The great toe interphalangeal joint and the second toe
proximal interphalangeal joints are subluxed; there has been
destruction of bone adjacent to several joint surfaces. The fifth
metatarsal shows “penciling” of the distal end, the end result of an
inflammation-driven clastic removal of the outer layer of the cortex
and the unbalanced deposition of endosteal bone to oppose this deletion. -
Regardless of the joint involved, the general features are:
-
Regional osteopenia, bone destruction, and periosteal reaction
-
Could be neuropathic arthropathy, osteomyelitis, or fracture without infection
-
-
Over time the appearance is one of:
-
Joint swelling
-
Subluxation or frank dislocation
-
Joint dissolution with debris and deformity
-
Regional osteopenia, local sclerosis
-
-
P.441
Pathologic Findings
-
Mass-like, exuberant inflammatory
process, usually mixed acute and chronic inflammation of synovium and
subsynovial soft tissue with rich vascularity (granulation tissue-like) -
Numerous shards of bone and articular cartilage entrapped in soft tissue
Avascular Necrosis
-
The importance of avascular necrosis is as a cause of secondary osteoarthritis.
-
It can occur in multiple sites and in both children (e.g., Legg-Calve-Perthes disease) and adults. (Also see Chapter 19.)
Anomalies
Dysplasia Epiphysealis Hemimelica (Trevor Disease)
Definition
-
Dysplasia epiphysealis hemimelica is a
rare congenital anomaly affecting the growth plates of the knee, talus,
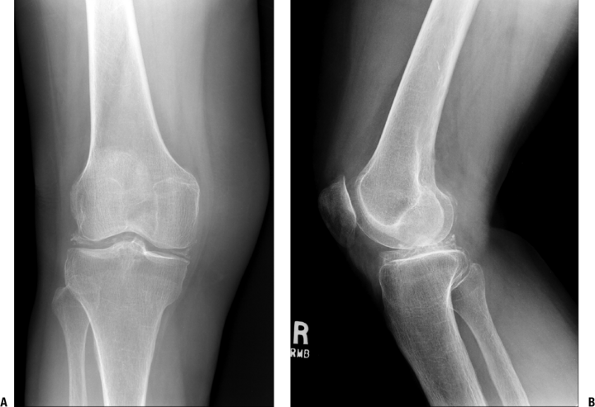
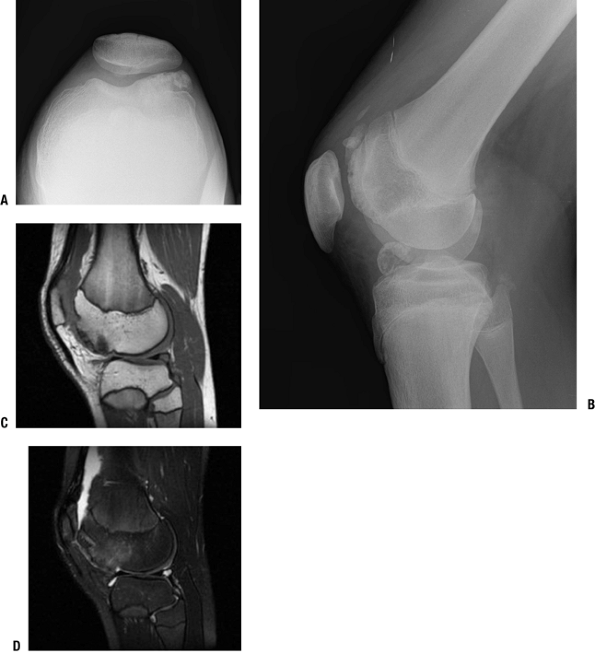
and tarsal navicular and first cuneiform joints.![]() Figure 21-18
Figure 21-18
Dysplasia epiphysealis hemimelica (Trevor disease). A 14-year-old boy
with atraumatic knee pain and locking and epiphyseal osteochondroma.
Sunrise (A) and lateral (B) views of the knee show an osseous
excrescence extending from the anterior aspect of the lateral femoral.
Multiple osteochondral fragments project in the joint. Note the
elongation of the inferior pole of the patella in response to
chronically abnormal patellofemoral mechanics. Sagittal T1-weighted (C)
and T2-weighted (D) images through the lateral femoral condyle show the
lesion at the anterior aspect of the femoral condyle, with cortical and
medullary continuity with underlying bone. -
It essentially consists of an epiphyseal osteochondroma that protrudes into the joint space.
-
The medial side of the epiphysis is most commonly affected.
P.442
Epidemiology
-
Incidence: 1:1,000,000
-
No race predilection
-
Male:female 3:1
Pathophysiology (Intra-articular Osteochondroma)
-
Abnormality of the regulation of cartilage proliferation in the affected epiphysis, resulting in a cartilaginous exostosis.
-
The morphology is that of benign cartilage over a base that resembles an osteochondroma.
Diagnosis
Differential Diagnosis
-
Chondroblastoma, osteochondroma, enchondroma
Clinical Features
-
Swelling and mechanical symptoms occur as the growing osteocartilaginous mass distorts the joint.
-
Secondary changes include loss of motion, deformity, and atrophy of the supporting muscles.
Radiologic Findings (Fig. 21-18)
-
The gross anatomy is of an exostosis,
projecting and contiguous to the subchondral surface, covered by
cartilage. The radiograph shows an abnormal projection of normal bone
arising from the subchondral surface. Arising in childhood, the
abnormal forces of this focal projection may cause secondary local
changes in the adjacent articular structure. -
Magnetic resonance imaging will show the
continuity of the base of the exostosis with the subchondral bone as
well as abnormal signal in the cartilage cap in both the T1-and
T2-weighted images.
Suggested Reading
Adams A, Lehman TJ. Update on the pathogenesis and treatment of systemic onset juvenile rheumatoid arthritis. Curr Opin Rheumatol 2005;17:612–616.
Bonnet CS, Walsh DA. Osteoarthritis, angiogenesis and inflammation. Rheumatology (Oxford) 2005;44:7–16.
Buckwalter JA, Brown TD. Joint injury, repair, and remodeling: Roles in post-traumatic osteoarthritis. Clin Orthop Rel Res 2004:7–16.
Buckwalter JA, Martin JA. Sports and osteoarthritis. Curr Opin Rheumatol 2004;16:634–639.
Choi HK, Mount DB, Reginato AM. Pathogenesis of gout. Ann Intern Med 2005;143:499–516.
Davis JC Jr. Understanding the role of tumor necrosis factor inhibition in ankylosing spondylitis. Semin Arthritis Rheum 2005;34: 668–677.
Gladman DD, Antoni C, Mease P, et al. Psoriatic arthritis: Epidemiology, clinical features, course, and outcome. Ann Rheum Dis 2005; 64:14–17.
Halverson PB, Derfus BA. Calcium crystal-induced inflammation. Curr Opin Rheumatol 2001;13:221–224.
Hammaker D, Sweeney S, Firestein GS. Signal transduction networks in rheumatoid arthritis. Ann Rheum Dis 2003;62:86–89.
Haugeberg G, Orstavik RE, Kvien TK. Effects of rheumatoid arthritis on bone. Curr Opin Rheumatol 2003;15:469–475.
Kim TH, Uhm WS, Inman RD. Pathogenesis of ankylosing spondylitis and reactive arthritis. Curr Opin Rheumatol 2005; 17:400–405.
Kuo RS, Bellemore MC, Monsell FP, etal. Dysplasia epiphysealis hemimelica: Clinical features and management.J Pediatr Orthop 1998; 18:543–548.
Liu-Bryan
R, Liote F. Monosodium urate and calcium pyrophosphate dihydrate (CPPD)
crystals, inflammation, and cellular signaling. Joint Bone Spine 2005;72:295–302.
R, Liote F. Monosodium urate and calcium pyrophosphate dihydrate (CPPD)
crystals, inflammation, and cellular signaling. Joint Bone Spine 2005;72:295–302.
Mannoni A, Selvi E, Lorenzini S, et al. Alkaptonuria, ochronosis, and ochronotic arthropathy. Semin Arthritis Rheumatism 2004;33: 239–248.
Mohammed FF, Smookler DS, Khokha R. Metalloproteinases, inflammation, and rheumatoid arthritis. Ann Rheum Dis 2003;62:43–47.
Muller-Ladner U, Gay RE, Gay S. Activation of synoviocytes. Curr Opin Rheumatol 2000;12:186–194.
Reveille JD, Arnett FC. Spondyloarthritis: Update on pathogenesis and management. Am J Med 2005;1 18:592–603.
Steinbach LS, Resnick D. Calcium pyrophosphate dihydrate crystal deposition disease: Imaging perspectives. Curr Probl Diagn Radiol 2000;29:209–229.
Toivanen A, Toivanen P. Reactive arthritis. Curr Opin Rheumatol 2000;12:300–305.