Classification of Fractures
for nearly as long as people have identified fractures; they certainly
predate the advent of radiography. Even in the earliest written
surviving medical text, the Edwin Smith Papyrus, there was a
rudimentary classification of fractures. If a fracture could be
characterized as “having a wound over it, piercing through”—in other
words, an open fracture—it was determined to be an “ailment not to be
treated.” This early form of one of the earliest systems of fracture
classification served both to characterize the fracture and to guide
the treatment.
classification have served numerous purposes: to characterize fractures
as far as certain general and specific features, to guide treatment,
and to predict outcomes. This chapter will review the purposes and
goals of fracture classification, the history of the use of such
systems, and the general types of fracture classification systems in
common use today. This chapter will also provide a critical analysis of
the effectiveness of fracture classification systems, as well as some
of the limitations of these systems. Finally, it will comment on the
possible future of fracture classification systems.
not unique to orthopaedics or to fractures. Taxonomy is a universal
phenomenon that occurs in all fields of science and art. One clear and
simple example is the system of taxonomy that has been used to divide
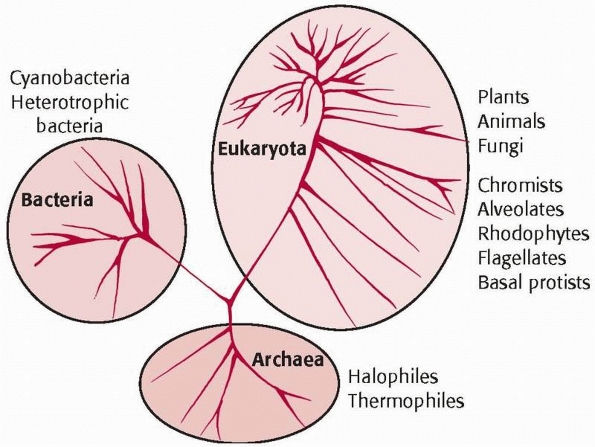
the natural world into three kingdoms: animals, plants, and bacteria (Fig. 2-1).
This taxonomy, though simple, is a perfect example of the kinds of
classification that permeate the world of arts and sciences and of the
first general purpose of classification systems—to name things.
 |
|
FIGURE 2-1 Balloon diagram of taxonomy of the natural world.
|
|
TABLE 2-1 Purposes of Classification Systems
|
||||||
|---|---|---|---|---|---|---|
|
describe things according to characteristics and to provide a hierarchy
of those characteristics. A group of common descriptors are created so
that individual items can be classified into various groups. Groups are
then ordered into a hierarchy according to some definition of
complexity. A simple example of this is the phylogeny used to describe
the animal kingdom; this system describes and groups animals according
to common characteristics, and then orders those groups in a hierarchy
of complexity of the organism. This is, in principle, analogous to many
fracture classification systems, which provide a group of common
descriptors for fractures that are ordered according to complexity.
action or intervention. This feature of classification systems is not
universally seen, and it is generally present only in classification
systems that are diagnostic in nature. This introduces one of the key
distinctions among classification systems—that between systems used for
description and characterization and those used to guide actions and
predict outcomes. For example, the classification system for the animal
kingdom names and classifies animals, but it is descriptive only—it
does not guide the observer in any suggested action. In orthopaedic
practice, however, physicians use fracture classification systems to
assist in making treatment decisions. In fact, many fracture
classification systems were designed specifically for the purpose of
guiding treatment. We should have higher expectations of the validity
and integrity of systems that are used to guide actions than those used
purely as descriptive tools.
assist in predicting outcomes of an intervention or treatment. The
ability to reliably predict an outcome from a fracture classification
alone would be of tremendous benefit, for it would allow physicians to
counsel patients, beginning at the time of injury, about the expected
outcome. This ability would also assist greatly in clinical research,
as it would allow the comparison of the results from one clinical study
of a particular fracture to that of another. It should be clear to the
reader that, for a classification system to reliably predict outcome, a
rigorous analysis of the reliability and validity of the classification
system is necessary. Table 2-1 summarizes the
purposes of classification systems, along with the level of reliability
and validity necessary for high performance of the system.
have radiographs. The Edmund Smith Papyrus, while it did not make a
clear distinction between comminuted and noncomminuted fractures,
clearly classified fractures as open or closed, and provided guidelines
for treatment based on that classification. Open fractures, for
example, were synonymous with early death in the Ancient Egypt, and
these fractures were “ailments not to be treated.”
discovery of radiographs, there were in existence fracture
classification systems that were based on the clinical appearance of
the limb alone. The Colles fracture of the distal radius, in which the
distal fragment was displaced dorsally—causing the dinner fork
deformity of the distal radius—was a common fracture. Any fracture with
this clinical deformity was considered a Colles fracture and was
treated with correction of the deformity and immobilization of the limb.15
The Pott fracture, a fracture of the distal tibia and fibula with varus
deformity, was likewise a fracture classification that was based only
on the clinical appearance of the limb.58
These are but two examples of fracture classifications that were
accepted and in widespread use prior to the development of radiographic
imaging.
systems expanded in number and came into common usage. Radiography so
altered the understanding of fractures and the methods of fracture care
that nearly all fracture classification systems in use today are based
solely on a characterization of the fracture fragments on plain
radiographs. Most modern fracture classification systems are based on a
description of the location, number, and displacement of fracture lines
viewed on radiographs, rather than on the clinical appearance of the
fractured limb. While countless fracture classification systems based
on radiographs have been described in the past century for fractures in
all parts of the skeleton, only the most enduring classification
systems remain in common usage today. Examples of these enduring
classification systems are the Garden30 and Neer51
classification systems of proximal femoral and proximal humeral
fractures, respectively. These and other commonly used classification
systems will be discussed in more detail in a later part of this
chapter.
are based upon having observers—usually orthopaedic physicians —make
judgments and interpretations based on the analysis of plain
radiographs of the fractured bone. Usually, anteroposterior and lateral
radiographs are used, although some fracture classification systems
allow for or encourage the use of additional x-ray views, such as
oblique radiographs, or internal and external rotation radiographs. It
is evident that each decision made in the process of classifying a
fracture is based on a human’s interpretation of the often complex
patterns of shadows evident on a plain radiograph of the fractured
limb. This, in turn, requires that the observer have a detailed and
fundamental understanding of the osteology of the bone being imaged and
of the fracture being classified. The observer must have the ability to
accurately and completely identify all of the fracture lines,
understand the origin and nature of all of the fracture fragments, and
delineate the relationship of all of the fracture fragments to one
another. Finally, the procedure of fracture classification requires
that the observer very accurately quantify the amount of displacement
or angulation of each fracture fragment from the location in which it
should be in the nonfractured situation.
been added by many observers to assist in classifying fractures. In
most cases, the CT scan data has been used and applied
to
a classification system that was devised for use with plain radiographs
alone. There are a few classification systems, however, that are
specifically designed for use with CT imaging data. The most well-known
example of such a system is the Sanders classification system for
fractures of the calcaneus,61 which was designed for use with a carefully defined semicoronal CT sequence through the posterior facet of the subtalar joint.
 |
|
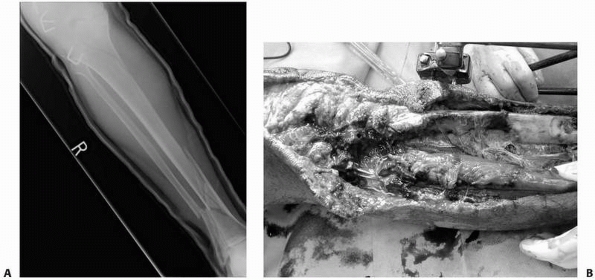
FIGURE 2-2 Tibial fracture as seen on radiograph (A) and intraoperatively (B). The x-ray appearance greatly underestimates the overall severity of the injury.
|
relied solely on radiographic images to classify the fracture, guide
treatment, and predict outcomes. It is becoming increasingly
appreciated, however, that nonradiographic factors such as the extent
of soft tissue injury when there are other injuries (skeletal or
nonskeletal), medical comorbidities, and various other nonradiographic
factors have a large effect on treatment decisions and on the outcomes
of fracture treatment.23,42 These factors, however, are not accounted for in radiographic systems for fracture classification.
to fully appreciate the extent of soft tissue damage that has occurred,
and the image provides no information about the patient’s medical
history. For example, if one views a radiograph of the transverse
tibial shaft fracture shown in Figure 2-2, one
may conclude that this is a simple, low-energy injury. In this example,
however, the fracture occurred as a result of very high energy, and the
patient sustained extensive soft tissue damage. In addition, the
patient was an insulin-dependent diabetic with severe peripheral
neuropathy and skin ulcerations on the fractured limb. There is no way,
from view of the plain radiographs or application of a fracture
classification based on radiographs alone, to account for these
additional factors. The patient in this example required amputation, a
treatment that would not be predicted by review of the radiographs
alone. Some discussion of the role of classifying the soft tissue
injury in characterizing fractures will take place later in this
chapter.
can be characterized into three broad categories: (i) those that are
fracture specific, which evolved around and were generated for the
classification of a single fracture in a single location in the
skeleton; (ii) those that are generic or universal fracture
classification systems, which apply a single, consistent methodology to
the classification of fractures in all parts of the human skeleton; and
(iii) those that attempt to classify the soft tissue injury. It is
beyond the scope of this chapter to discuss individually all the
fracture classification systems now in common usage, but it is
important for the reader to understand the differences between the
general types of classification systems. For that reason, some examples
of each of the three types of fracture classification systems will be
discussed.
is a longstanding fracture classification system that describes the
displacement and angulation of the femoral head on anteroposterior (AP)
and lateral radiographs of the hip (see Fig. 47-2).
The classification is essentially a descriptive one, describing the
location and displacement of the fractured femoral neck and head. The
fracture types are ordered, however, to indicate increasing fracture
severity, greater fracture instability, and higher risk of
complications with attempts at reduction and stabilization of the
fracture. This feature of ordering fracture types by severity takes the
classification system from a nominal system to an ordinal system.
Garden types 1 and 2 fractures are considered to be stable injuries and
are frequently treated with percutaneous internal fixation. Garden 3
and 4 fractures have been grouped as unstable fracture patterns and,
while closed reduction and internal fixation are used in some
circumstances, most Garden 3 and 4 fractures in elderly patients are
treated with arthroplasty.
is an example of another descriptive classification system that has
been widely utilized and is based on the location of the major fracture
line in the proximal tibia and the presence or absence of a depressed
segment of the articular surface of the proximal tibia (see Fig. 53-9). This fracture classification is not dependent on the amount of displacement or depression
of the articular fractures, but only on the location of the fracture
lines. The Schatzker classification seems very simple, but it also
demonstrates some of the areas of confusion that can result from
fracture classifications. For example, Schatzker V and VI fractures are
distinct fracture types in the system, but observers have a great deal
of difficulty in distinguishing these two fracture types from one
another when viewing fracture radiographs. Also, the Schatzker VI
fracture group includes fractures classified as types C1 and C3 by the
AO/OTA system (described below), thus demonstrating an area of
inconsistency between two commonly used but different systems for
classifying the same fracture that can lead to confusion among
observers.
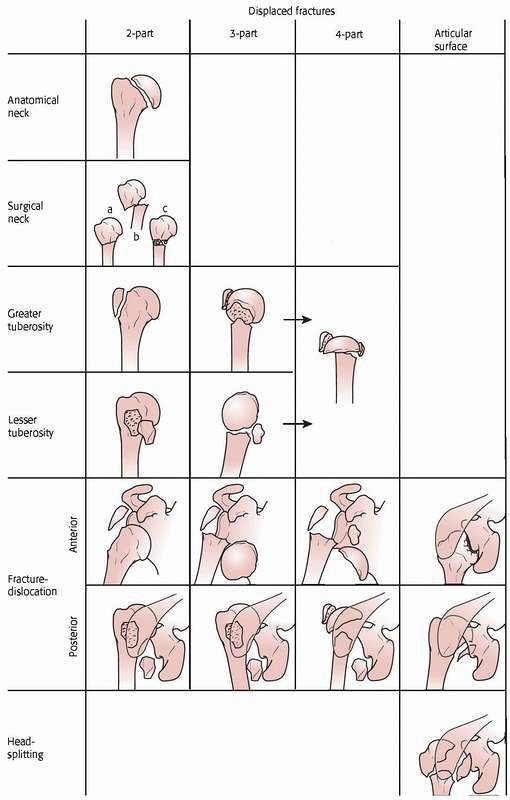
It is based on how many fracture “parts” there are—a part is defined as
a fracture fragment that is either displaced more than one centimeter
or angulated more than 45 degrees. The Neer classification groups
fractures into nondisplaced (one-part), two-part, three-part, or
four-part fractures. Nondisplaced fractures in the Neer system involve
several fracture lines, none of which meet the displacement or
angulation criteria to be considered a “part.” Two-part fractures in
the Neer system can represent either a fracture across the surgical
neck of the humerus or greater tuberosity fracture that is displaced.
Three-part fractures classically involve the humeral head, in greater
tuberosity fragments being displaced or angulated. Four-part fractures
involve displacement or angulation of the humeral head and greater and
lesser tuberosities. The reader should note that, in addition to
correct identification of the fracture fragments, this classification
system requires the observer to make careful and accurate measurements
of fragment displacement and angulation to determine if a fragment
constitutes a part.
is an example of a widely used system that is based primarily on the
mechanism of injury. The system makes use of the fact that particular
mechanisms of injury to the ankle will result in predictable patterns
of fracture to the malleoli. The appearance of the fracture on the
radiographs, then, is used to infer the mechanism of the injury. The
injuries are classified according to the position of the foot at the
time of injury and the direction of the deforming force at the time of
fracture. The position of the foot is described as pronation or
supination, and the deforming force is categorized as external
rotation, inversion, or eversion. This creates six general fracture
types, which are essentially nominal—they are not ordered into
increasing injury severity. Within each fracture type, however, there
is an ordinal scale, with varying degrees of severity being assigned to
each type (1-4) according to the fracture pattern. With this
classification system, correct determination of the fracture type can
guide the manipulations necessary to affect fracture reduction —the
treating physician must reverse the direction of the injuring forces to
achieve a reduction. For example, internal rotation is required to
achieve reduction of a supination external rotation fracture pattern.
is essentially the only generic or universal system in wide usage
today. It is universal in the sense that the same fracture
classification system can be applied to any bone within the body. This
classification system was devised through a consensus panel of
orthopaedic traumatologists who were members of the Orthopaedic Trauma
Association and is based upon a classification system initially
developed and proposed by the AO/ASIF group in Europe.49,50
The Orthopaedic Trauma Association believed there was a need for a
detailed universal system for classification of fractures to allow for
standardization of research and communication among orthopaedic
surgeons. The AO/OTA fracture classification system is an alphanumeric
system that can be applied to most bones within the body.
-
Which bone? The major bones in the body are numbered, with the humerus being #1, the forearm #2, the femur #3, the tibia #4, and so on (Fig. 2-4).
-
Where in the bone is the fracture?
The answer to this question identifies a specific segment within the
bone. The second number of the coding system is applied to the location
in the bone. In most long bones, the diaphyseal segment (2) is located
between the proximal (1) and distal (3) segments. The dividing lines
between the shaft segment and the proximal and distal segments occur in
metaphysis of the bone. The tibia is assigned a fourth segment, which
is the malleolar segment. An example of the application of answering
the first two questions of the AO/OTA classification is that a fracture
of the midshaft of the femur will be given a numeric classification of
32 (3 for femur, 2 for the diaphyseal segment) (Fig. 2-4). -
Which fracture type?
The fracture type in this system can be A, B, or C, but these three
types are defined differently in diaphyseal fractures and fractures at
either end of the bone. For diaphyseal fractures, the type A fracture
is a simple fracture with two fragments. The type B diaphyseal fracture
has some comminution, but there can still be contact between the
proximal and distal fragments. The type C diaphyseal fracture is a
highly comminuted or segmental fracture with no contact possible
between proximal and distal fragments. For proximal and distal segment
fractures, type A fractures are considered extra-articular, type B
fractures are partial articular (there is some continuity between the
shaft and some portion of the articular surface), and Type C fractures
involve complete disruption of the articular surface from the
diaphysis. An example of this portion of the classification system is
shown in Figure 2-4. -
Which group do the fractures belong to?
Grouping further divides the fractures according to more specific
descriptive details. Fracture groups are not consistently defined; that
is, fracture groups are different for each fracture type. Complete
description of the fracture groups is beyond the scope of this chapter. -
Which subgroup?
This is the most detailed determination in the AO/OTA classification
system. As is the case with groups, subgroups differ from bone to bone
and depend upon key features for any given bone in its classification.
The intended purpose of the subgroups is to increase the precision of
the classification system. An in-depth discussion of this fracture
classification is beyond the scope of this chapter, and the reader is
referred to the references for a more detailed description of this
universal fracture classification system.
 |
|
FIGURE 2-3
The Neer four-part classification of proximal humerus fractures. A fracture is displaced if the fracture fragments are separated 1 cm or greater, or if angulation between the fracture fragments is more than 45 degrees. A displaced fracture is either a two-, three-, or four-part fracture. (From Neer CS. Displaced proximal humeral fractures: I. classification and evaluation. J Bone Joint Surg 1970;52A:1077-1089, reprinted with permission from Journal of Bone and Joint Surgery.) |
 |
|
FIGURE 2-4
The AO/ASIF Comprehensive Long Bone Classification applied to proximal humeral fractures. This system describes three types of proximal humerus fractures (types A, B, and C). Type A fractures are described as unifocal extra-articular (two-segment) fractures, type B as bifocal extra-articular (three-segment) fractures, and type C as anatomic neck or articular segment fractures. Each type includes three fracture patterns, with nine subgroups for each type of fracture. The subgroup classification indicates the degree of displacement. (Adapted from Müller ME, Allgower M, Schneider R, et al. Manual of Internal Fixation. New York: Springer-Verlag, 1991, with permission.) |
It is continually evaluated by a committee of the OTA, and is open for
change where appropriate. The reader should note that the AO/OTA
classification system of fractures, and its precursor, the AO/ASIF
system, were designed for delineation and recording of the maximum
possible amount of detail about the individual fracture pattern and
appearance on radiographs. The assumption made during the development
of these classification systems is that with specific
definitions/diagrams and a high degree of detail come greater accuracy
and a superior fracture classification system that could be applied by
any orthopaedic surgeon. It was believed such a system could
potentially result in better prognostic and research capabilities. As
will be discussed later in the chapter, greater specificity and detail
in a fracture classification system does not necessarily correlate well
with good performance of the classification system.
energy of the injury may be reflected in the soft tissue damage to the
extremity involved. If one sees a radiograph demonstrating a comminuted
fracture, it is often thought that it is a high-energy injury. However,
there may be other patient factors that come into consideration and may
lead to a comminuted fracture from a lower-energy mechanism. This may
be evident in an elderly patient with ground-level falls who has a
significantly comminuted distal humerus fracture. The energy of the
injury itself resulted only from a ground-level fall, but led to a
complex fracture type as a result of underlying osteoporotic bone. Some
of the value in the soft tissue classification system is in planning
the treatment as well as in predicting the outcome.
soft tissue injury is the open fracture. Early classification systems
for open fractures focused only on the size of the opening in the skin.
With time, however, it was recognized that the extent of muscle injury,
local vascular damage, and periosteal stripping are also of paramount
significance. Gustilo et al.32,33
developed the classification system now used by most North American
orthopaedists to describe open fractures. This classification system
takes into account the skin wound, the extent of local soft tissue
injury and contamination, and the severity of the fracture pattern (see
Table 12-2). The Gustilo classification system
originally included type I, type II, and type III fractures. However,
this system was modified later to expand the type III open fractures
into subtypes A, B, and C. It is important to note that the type III-C
fracture is defined as any open fracture in which there is an
accompanying vascular injury that requires repair.
The Gustilo classification system has been applied to open fractures in
nearly all long bones. It is important to recognize that this
classification system only can be applied fully after surgical
debridement of the open fracture has been performed. This system
has proven useful in predicting risk of infection in open tibial fractures.32
|
TABLE 2-2 Oestern and Tscherne Classification of Closed Fractures
|
|||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
according to the classification of Gustilo was investigated by Brumback
and Jones,10 who presented
radiographs and videotapes of surgical debridements to a group of
orthopaedic traumatologists who classified the fractures. They reported
an average interobserver agreement of 60%. The range of agreement,
however, was wide, ranging from 42% to 94%. Percentage agreement was
best for the most severe and the least severe injuries, and was poorer
for fractures in the middle range of the classification system. That
the classification system did not have similar reliability across the
spectrum of injury severity has been a criticism of this classification
system as a prognostic indicator for any but the least severe and most
severe injuries.
This system remains the only published classification system for the
soft tissue injury associated with closed fractures. Fractures are
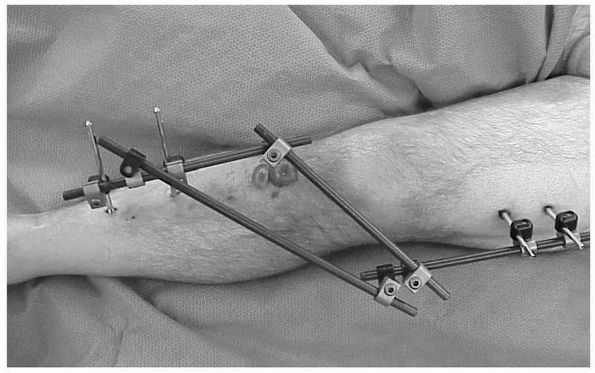
assigned one of four grades, from 0 to 3. Figure 2-5
is an example of a patient with a Tscherne Grade 2 closed tibial
plateau fracture. Deep abrasions of the skin, muscle contusion,
fracture blisters, and massive soft tissue swelling, as in this
patient, may lead the surgeon away from immediate articular
stabilization and toward temporary spanning external fixation. No
studies have been done to determine the interobserver reliability of
the Tscherne system for the classification of the soft tissue injury
associated with closed fractures.
if it can assist in predicting outcome. A prospective study completed
by Gaston et al.31 assessed various
fracture classification schemes against several validated functional
outcome measures in patients with tibial shift fractures. The Tscherne
classification system of closed fractures was more predictive of
outcome than the other classification systems used. The Tscherne system
was most strongly predictive of time to return to prolonged walking or
running.
 |
|
FIGURE 2-5 Example of a Tscherne II fracture of the proximal tibia.
|
Reliability reflects the ability of a classification system to return
the same result for the same fracture radiographs over multiple
observers or by the same observer when viewing the fracture on multiple
occasions. The former is termed interobserver reliability, or the
agreement between different observers using the classification system
to assess the same cases. The latter is termed intraobserver
reproducibility—the agreement of the same observer’s assessment, using
the classification system, for the same cases on repeated occasions.
The validity of a classification system reflects the accuracy with
which the system describes the true fracture entity. A valid
classification system would correctly categorize the fracture in a
large percentage of cases, when compared to a “gold standard.”
Unfortunately, there is no such gold standard for fracture
classification—not even observation at surgery can be considered
infallible—so the assessment of the performance of fracture
classification systems must be confined to assessing interobserver
reliability and intraobserver reproducibility.
the terms “agreement” and “accuracy” in reference to the performance of
fracture classification systems, as well as which of these terms is the
best measure of a system’s performance. The term “accuracy” implies
that there is a correct answer or a gold standard against which
comparisons can be made, validated, and determined to be true or false.
However, the term “agreement” indicates that there is no defined gold
standard and that unanimous agreement among all observers that might
classify a given fracture is the highest measure of performance of a
classification system. These two terms are not congruent and they are
not interchangeable. Each is tested by a vastly different statistical
method and to optimize each would require a radically different method
for generating and validating a fracture classification
system.
It has been unclear at times whether those developing and applying
classification systems today are expecting the classification to serve
as a gold standard or they are attempting to develop the classification
to achieve optimal agreement among observers.
appear in the orthopaedic literature assessing the interobserver
reliability of various fracture classifications systems.2,3,28,39,53,59,67,73 In a controversial editorial published in 1993, Fracture Classification Systems: Do They Work and Are They Useful, Albert Burstein, Ph.D., arrayed some important issues and considerations for fracture classification systems.11
He stated that classification systems are tools, and that the measure
of whether such a tool works is if it produces the same result, time
after time, in the hands of anyone who employs the tool. Dr. Burstein
went on to say that “any classification scheme, be it nominal, ordinal,
or scalar, should be proved to be a workable tool before it is used in
a discriminatory or predictive manner.” He emphasized that the key
distinction for a classification system was between its use to describe
and characterize fractures and its use to guide treatment or predict
outcomes. It is the latter use that requires a system to be proven to
be a valid tool; the minimum criteria for acceptable performance of any
classification system should be a demonstration of a high degree of
interobserver reliability and intraobserver reproducibility.
editorial appeared, and nearly all concluded that fracture
classification systems had substantial interobserver variability.
Classification systems for fractures of the proximal femur,2,28,53,56,64,73 proximal humerus,7,9,39,67,68 ankle,16,52,59,64,73 distal tibia,22,45,71 and tibial plateau,12,46,75,76,77
among others, were all shown to have poor to slight interobserver
reliability. The earliest of these studies looked only at the observed
percentage of agreement —the percentage of times that individual pairs
of observers categorized fractures into the same category. Subsequent
studies, however, have most frequently used a statistical test known as
the kappa statistic, a test that analyzes pair-wise comparisons between
observers applying the same classification system to a specific set of
fracture cases. The kappa statistic was originally introduced by Cohen
in 1960,13 and the kappa statistic
and its variants are the most recognized and widely used methods for
measuring reliability for fracture classification systems. The kappa
statistic adjusts the proportion of agreement between any two observers
by correcting for the proportion of agreement that could have occurred
by chance alone. Kappa values can range from + 1.0 (perfect agreement)
to 0.0 (chance agreement) to – 1.0 (perfect disagreement) (Table 2-3).
are only two choices of fracture categories or when the fracture
classification system is nominal—all categorical differences are
equally important. In most situations, however, there are more than two
categories into which a fracture can be classified, and fracture
classification systems are ordinal—the categorical differences are
ranked according to injury severity, treatment method, or presumed
outcome. In these cases, the most appropriate variant of the kappa
statistic to be used is the weighted kappa statistic, described by
Fleiss,26,27
in which some credit is given to partial agreement and not all
disagreements are treated equally. For example, in the Neer
classification of proximal humeral fractures, disagreement between a
nondisplaced and a two-part fracture has far fewer treatment
implications than disagreement between a nondisplaced and a four-part
fracture. By weighting kappa values, one can account for the different
levels of importance between levels of disagreement. However, the most
appropriate use of the weighted kappa statistic should include a clear
explanation of the weighting scheme selected, since the results of the
kappa statistic will vary—even with the same observations—if the
weighting scheme varies.29 Thus,
without specific knowledge of the weighting scheme used, it is
difficult to compare the results of fracture classification system
reliability across different studies.
|
TABLE 2-3 Range of the Kappa Statistic
|
||||||||
|---|---|---|---|---|---|---|---|---|
|
have been used to categorize kappa values; values less than 0.00
indicate poor reliability, 0.01 to 0.20 indicate slight reliability,
0.21 to 0.40 indicate fair reliability, 0.41 to 0.60 indicate moderate
reliability, 0.61 to 0.80 indicate substantial reliability, and 0.81 to
1.00 indicate nearly perfect agreement. Although these criteria have
gained widespread acceptance, the values were chosen arbitrarily and
were never intended to serve as general benchmarks. A second set of
criteria, also arbitrary, have been proposed by Svanholm et al.: less
than 0.50 indicate poor reliability; 0.51 to 0.74 indicate good
reliability, and greater than 0.75 indicate excellent reliability.69
been found to be a limitation of many fracture classification systems.
Many studies have documented only fair to poor intraobserver
reliability for a wide range of fracture classification systems.
Systems tested have included, among others, the Neer fracture
classification system of proximal humeral fractures,7,8,39,67 the Garden classification systems of proximal femoral fractures,2,28,53,56,64 the Rüedi and Allgöwer and AO classification systems of distal tibial fractures,22,45,71 the Lauge-Hansen and Weber classification of malleolar fractures,16,52,72,73 and the Schatzker and AO fracture classification system of proximal tibial fractures.12,46,75,76,77
Even the Gustilo-Anderson classification system for classifying open
fractures has been shown to have only fair interobserver reliability.10
Additionally, studies have shown observer variability in classifying
various other orthopaedic injuries, such as fractures of the acetabulum,6,57,74 the distal radius,1,3,36 the scaphoid,20 the spine,5,55 the calcaneus,35,41 and gunshot fractures of the femur.66
sources of this variability, but the root cause for the variability has
not been identified. It remains unknown if any system for the
classification of fractures can perform with excellent intraobserver
reliability when it will be used by many observers. A methodology for
validation for fracture classification systems has been proposed, but
it is highly detailed and extremely time consuming and it is unknown if
it can be practically applied.4
the reliability of fracture classification systems should clearly state
the weighting scheme used. Methodological issues such as this were
evaluated in a systematic review of 44 published studies assessing the
reliability of fracture classification systems.4
Various methodological issues were identified, including a failure to
assure that the study sample of fracture radiographs was representative
of the spectrum and frequency of injury severity seen for the
particular fracture in 61% of the studies, a failure to justify the
size of the study group in 100% of the studies, and inadequate
statistical analysis of the data in 61% of the studies. While the
authors of this study used very rigid and, some would argue, unfairly
rigorous criteria to evaluate these studies, the authors’ conclusion
that reliability studies of fracture classification cannot be easily
compared to one another is valid and appropriate. The development and
adoption of a systematic methodological approach to the development and
validation of new fracture classification systems seems appropriate and
is needed.
a fracture classification scheme correlates well with outcomes
following fracture care.70 In a
prospective, multicenter study, 200 patients with unilateral isolated
lower extremity fractures (acetabulum, femur, tibia, talus, or
calcaneus) underwent various functional outcome measurements at 6 and
12 months, including the Sickness Impact Profile and the AMA Impairment
rating. The AO/OTA fracture classification for each of these patients
was correlated with the functional outcome measures. While the study
indicated some significant differences in functional outcome between
type C and type B fractures, there was no significant difference
between type C and type A fractures. The authors concluded that the
AO/OTA code for fracture classification may not be a good predictor of
6- and 12-month functional performance and impairment for patients with
isolated lower extremity fractures.
some of the reasons for interobserver variation in the classification
of fractures. These studies have generally focused on a few specific
variables or tasks involved in the fracture classification process.
Some of those which have been investigated are discussed in the
following paragraphs.
clinical practice and may affect the observer’s ability to accurately
or reproducibly identify and classify the fracture. Many have
attributed observed intraobserver variability in fracture
classifications systems to variations in the quality of radiographs.1,7,28,37,39,67
Studies looking specifically at this variable, however, have not
demonstrated it to be a significant source of intraobserver variability.16,22
In one such study involving classification of tibial plafond fractures
using the Rüedi and Allgöwer system, observers were asked to classify
the fractures, but also asked to make a determination of whether the
radiographs were of adequate quality to classify the fracture.22
In that study, observers agreed less on the quality of the radiographs
(mean kappa 0.38 + 0.046) than on the classification of the fractures
themselves (mean kappa 0.43 + 0.048). In addition, the extent of
interobserver agreement on the quality of the radiographs had no
correlation with the extent of agreement in classifying the fractures.
The authors concluded that, based on the results of their
investigation, it appeared that improving the quality of plain
radiographic images would be unlikely to improve the reliability of
classification of fractures of the tibial plafond.
as CT or magnetic resonance imaging (MRI) scanning, in which
high-quality images should always be obtained, have generally not
demonstrated improved intra-observer reliability over studies that have
used plain radiographs alone. Bernstein et al. found that CT scans did
not improve interobserver agreement for the Neer classification of
proximal humerus fractures.7 Chan et
al., in a study of the impact of a CT scan on determining treatment
plan and fracture classification for tibial plateau fractures, found
that viewing the CT scans did not improve interobserver agreement on
classification, but did increase agreement regarding treatment plan.12
Two studies investigating the effect of adding CT information to plain
radiographs on the interobserver agreement in classifying fractures of
the tibial plateau and tibial plafond failed to show a significant
improvement in agreement after the addition of CT scan information.12,46,47 Katz et al.,35
studying fractures of the distal radius, found the addition of a CT
scan occasionally resulted in changes in treatment plans and also
increased agreement among observers on the surgical plan in treating
these injuries. A study investigating the use of three-dimensional CT
scanning in distal humeral fractures concluded that three-dimensional
CT did not improve interobserver reliability over plain radiographs or
two-dimensional CT scans, but that it did improve intra-observer
reproducibility.25 These authors and
others have concluded that CT scan information may be a useful adjunct
in surgical planning for a severe articular fracture, but is probably
not required for the purpose of fracture classification.
Thirty-five distal radius fractures that had been classified as AO/OTA
A2 and A3 (extra-articular types) after radiographic review underwent
CT scanning. The scans revealed that 57% of the fractures had an
intra-articular component and had been inappropriately classified at
AO/OTA type A fractures. The reader should note that this study did not
attempt to determine interobserver reliability of the classification,
but simply that a single observer reviewing the CT scans disagreed with
the original fracture classification in 57% of cases. It remains
unproven whether CT scanning is a useful adjunct to improve
interobserver agreement in the classification of fractures.
interobserver reliability of classification of tibial plateau fractures
according to the Schatzker classification system.77
Three orthopaedic trauma surgeons classified tibial plateau fractures
first with plain radiographs, and then with either the addition of a CT
and an MRI scan. Kappa values averaged 0.68 with plain radiographs
alone, 0.73 with the addition of a CT scan, and 0.85 with addition of
an MRI scan. No statistical analysis was reported to indicate whether
the addition of CT and MRI information resulted in a statistically
significant improvement in reliability.
diagnostic image, usually a radiograph, on which the observer must make
observations, measurements, or both. Even with high-quality
radiographs, however, overlapping osseous fragments or densities can
make the accurate identification of each fracture fragment difficult.
Osteopenia can also increase the difficulty in accurate classification
of fractures. Osteopenic bone casts a
much
fainter “shadow” on radiographic films, making the delineation of fine
trabecular or articular details a much more difficult task for the
observer. Osteopenia represents a physiologic parameter that may affect
treatment plans and outcomes, but is not mentioned in any
classification system.
accurately classify with plain radiographs. Articular fractures tend to
occur in areas of the skeleton with complex three-dimensional
osteology, may be highly comminuted, and the classification systems
used for these fractures are predicated on the accurate identification
of each fracture fragment and determination of its relationship to the
other fragments and/or its position in the nonfracture situation.
Observer variability in the identification of these small fracture
fragments in complex fractures would be expected to lead to poorer
interobserver reliability of the fracture classification system.
Dirschl et al. investigated the observers’ ability to identify small
articular fragments in classifying tibial plafond fractures according
to the Rüedi and Allgöwer classification.22
Observers classified 25 tibial plafond fractures on radiographs and
then on line drawings that had been made from those radiographs by the
senior author; interobserver reliability was no different in the two
situations. At a second classification session, observers were asked to
first mark, on the fracture radiographs, the articular fragments and
then to classify the fractures; in a final session, the observers
classified the radiographs after the fracture fragments had been
premarked by the senior author. Having observers mark the fracture
fragments resulted in no improvement in interobserver reliability of
the fracture classification system. When identification of the
articular fragments was removed from the fracture classification
process, however, by having the fragments premarked by the senior
author, the interobserver reliability was significantly improved (mean
kappa value increased from 0.43 to 0.54, P<0.025).
The authors believed the results of this study indicated that observers
classifying fractures of the tibial plafond have great difficulty
identifying the fragments of the tibial articular surface on
radiographs. They went on to postulate that fracture classification
system predicated on the identification of the number and displacement
of small articular fragments may inherently perform poorly on
reliability analyses, because of observer difficulty in reliably
identifying the fracture fragments.
particularly articular fragments, has long been felt to be important in
characterizing fractures and has been used by many to make decisions
regarding treatment. Additionally, some classification systems for
fractures are predicated on the observer accurately identifying the
amount of displacement and/or angulation of fracture fragments; the
Neer classification system for proximal humeral fractures is an
example. Finally, the quality of fracture care has frequently been
judged by measuring the amount of displacement of articular fracture
fragments on posttreatment radiographs.
variability among observers in making measurements on radiographs and
that this may be a source for variability in fracture classification.
One such study assessed the error of measurement of articular
incongruity of tibial plateau fractures.47
In this study, five orthopaedic traumatologists measured the maximum
articular depression and the maximum condylar widening on 56 sets of
tibial plateau fracture radiographs. For 38 of the cases, the observers
also had a computed tomography scan of the knee to assist in making
measurements. The results of the study indicated that the 95% tolerance
limits for measuring maximum articular depression were ± 12
millimeters, and for measuring maximum condylar widening were ± 9
millimeters. This result indicates that there is substantial
variability in making these seemingly simple measurements.
of measurements decreases (the range of articular depression in the
study above was 35 millimeters). Thus, it would be expected that lower
tolerance limits would result from the measurement of reduced tibial
plateau than those observed in the reported study, which measured
injury films. However, in a study looking at the tolerance limits for
measuring articular congruity in healed distal radial fractures,
tolerance limits of ± 3 millimeters were identified, when the range of
articular congruity measurements was only 4 millimeters.36
reliability of measurement of articular fracture displacements. In one
study of intra-articular fractures of the distal radius, there was poor
correlation between measurement of gap widths or step deformities on
plain radiographs as compared to CT scans.14
Nearly one third of measurements made from plain radiographs were
significantly different than those made from CT scans. Another study
extended these findings by examining known intra-articular
displacements made in the hip joints of cadaveric specimens.8
The authors observed that CT-generated data were far more accurate and
reproducible than were data obtained from plain films. Moed et al.
reported on a series of posterior wall acetabular fractures treated
with open reduction and internal fixation in which reduction was
assessed on both plain radiographs and on CT scans.48
Of 59 patients who were graded as having an anatomic reduction based on
plain radiographs and for whom postoperative CT scans were obtained, 46
had a gap or step-off greater than 2 millimeters. These results may not
be characteristic of all fractures, since the posterior wall of the
acetabulum may be more difficult to profile using plain radiographs
than most areas of other joints.
observer variability in the routine measurement of articular
incongruity on radiographs. It also seems highly unlikely that
observers using plain radiographs can reliably measure small amounts of
incongruity. This suggests that improvements in our ability to reliably
assess the displacement of fracture fragments are necessary to reduce
variability in articular fracture assessment.
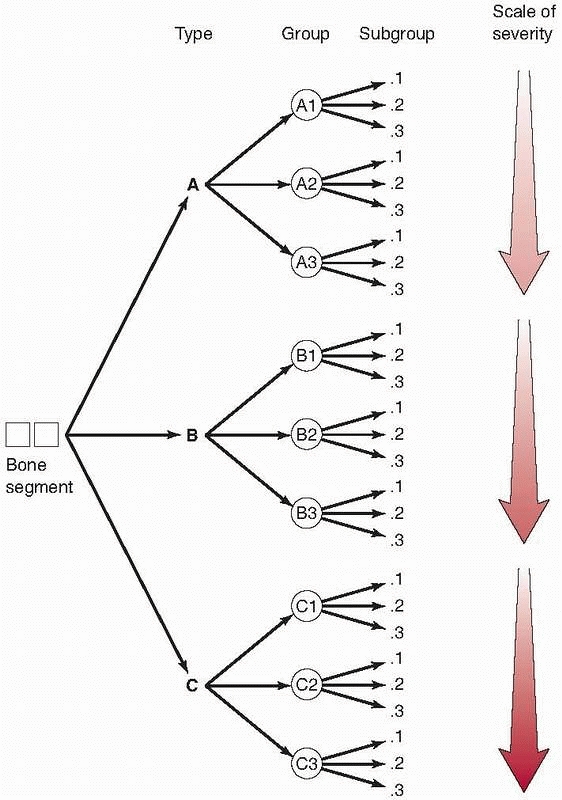
requiring the observer to choose between many possible categories in
characterizing a fracture. The AO/OTA system, for example, has up to 27
possible classifications for a fracture of a single bone segment (there
are three choices each for fracture type, group, and subgroup). It
seems reasonable that observers would find it easier to classify a
fracture if there were fewer choices to be made, and studies of the
AO/OTA fracture classification system have confirmed this. In nearly
all cases, for various fractures, classification of type can be
performed much more reliably than classification into groups or
subgroups.16,36,43,45,56,71,75 These studies concluded that, for optimal reliability, the use
of this classification beyond characterization of type was not recommended.
choices to no more than two for any step in the classification of
fractures would improve the ability of the observer to classify the
fracture and would improve interobserver reliability. In 1996, the
developers of the AO/ASIF comprehensive classification of fractures
(CCF) modified to incorporate binary decision-making.50
The reasoning was that, if observers could answer a series of “yes or
no” questions about the fracture, they could more precisely and
reliably classify the fracture. The modification was planned,
announced, and implemented without any sort of validation that the
modification would achieve the desired outcomes or that binary decision
making would improve reliability in fracture classification.
fracture types have evaluated whether binary decision making improves
reliability in the classification of fractures. The first of these
studies developed a binary modification of the Rüedi and Allgöwer
classification of tibial plafond fractures and had observers classify
25 fractures according to the original classification system and the
binary modification.22 The binary
modification was applied rigidly in fracture classification sessions
that were proctored by the author; observers were forced to make binary
decisions about the fracture radiographs, and not permitted to jump to
the final fracture classification. The results of this study indicated
that the binary modification of this classification system did not
perform with greater reliability than the standard classification
system (mean kappa 0.43 ± 0.048 standard and 0.35 ± 0.038 binary).
Another investigation compared the interobserver reliability of
classification of malleolar fractures of the tibia (segment 44)
according to the classic and binary modification of the AO/ASIF CCF.16
Six observers classified 50 malleolar fractures according to both the
standard and binary systems, and no difference in interobserver
reliability could be demonstrated between the two systems (mean kappa
0.61 standard and 0.62 binary). The authors concluded that strictly
enforced binary decision-making did not improve reliability in the
classification of malleolar fractures according to the AO/ASIF CCF. The
results of these two studies cast doubt on the effectiveness of binary
decision making in improving interobserver reliability in the
classification of fractures.
amount of information provided an observer could be overwhelming and
limit reliability of fracture classification.35
This group tested the Sanders classification of calcaneal fractures
and, rather than providing observers with the full CT scan data for
each of the 30 cases, they provided each observer with only one
carefully selected CT image from which to make a classification
decision. The results indicated that the overall interobserver
reliability was no better with only one CT cut than with the full
series of CT cuts. The results clearly showed, however, that
interobserver agreement was much better for the most and least severe
fractures in the series and poorest for fractures in the midrange of
severity. This finding is probably applicable to all classification
schemes, in which observers are much better at differentiating the best
from the worst than they are at cases in the middle of the spectrum of
injury severity.
are categorical; regardless of the nature or complexity of the
classification system, each group’s fractures are grouped into discreet
categories. Injuries to individual patients, however, occur on a
continuum of energy and severity of injury; fractures follow this same
pattern, occurring on a spectrum of injury severity. The process of
fracture classification can therefore be said to be a process by which
a continuous variable, such as fracture severity, is made a categorical
one. This “categorization” of a continuous variable may be a source of
intraobserver variability in fracture classification systems.29,45
One recent study concluded that “it has become clear that these
deficiencies are related to the fact that the infinite variation of
injury is a continuous variable and to force this continuous variable
into a classification scheme, a dichotomous variable, will result in
the discrepancies that have been documented.”29
The authors further suggested that “multiple classifiers, blinded to
the treatment selected and clinical outcomes, and consensus methodology
should be used to optimize the utility of injury classification schemes
for research and publication purposes.”
proposed that, instead of classifying fractures, perhaps fractures
should merely be rank ordered from the least severe to the most severe.
This would serve as a means to preserve the continuum of fracture
severity and has been proposed as a means of potentially improving
interobserver reliability. An initial study using this methodology in
tibial plafond fracture showed promise.19
Twenty-five tibial plafond fractures were ranked by three orthopaedic
traumatologists from the least severe to the most severe, and the group
demonstrated outstanding interobserver reliability, with a Cronbach
alpha statistic17 of 0.94 (nearly
perfect agreement). In a subsequent study, the rank order concept was
expanded and a series of 10 tibial plafond fractures were ranked by 69
observers.21 The intraclass
correlation coefficient was 0.62, representing substantial agreement,
but also represented some deterioration from the results with only
three observers. Based on these results, which are superior to those of
most categorical fracture classification systems that have been
evaluated, further study of this sort of classification system appears
to be warranted.
fracture classification system that ranks cases on a continuum of
injury severity would be to approach the matter much in the same way as
clinicians determine bone age in children.21
A series of radiographs would be published that represent the spectrum
of fracture severity, from the least severe to the most severe, and
then an observer would simply review these examples and determine where
the fracture under review lay on this spectrum of severity. This
concept is markedly different from any scheme used to date to classify
fractures, would be unlikely to completely replace other systems of
fracture classification, and may have weaknesses that have not yet been
determined. Such a system will require extensive testing and validation
before it could be widely used.
Recently, many have come to question whether any system for fracture
classification that relies solely on radiographic data will be highly
reliable or highly predictive of the outcome of severe fractures. There
is strong evidence that the
extent
of injury to the soft tissues (cartilage, muscle, tendon, skin, etc.),
the magnitude and durations of the patient’s physiologic response to
injury, the presence of comorbid conditions, and the patient’s
socioeconomic background and lifestyle may all play critical roles in
influencing outcomes following severe fractures.
articular cartilage is a critical and significant contributor to the
overall severity of an articular fracture, as evidenced by studies
documenting poor outcomes after osteochondritis dissecans and other
chondral injuries. The information present in the orthopaedic clinical
literature indicates that the severity of injury to the articular
surface during fracture has an important bearing on outcome and the
eventual development of posttraumatic osteoarthrosis. A better
understanding of the impaction injury to the articular cartilage and
the prognosis of such injury will be critical to improving our
assessment and understanding of severe intra-articular fractures.
Unfortunately, there currently are no imaging modalities that have been
validated to indicate to the clinician the extent of injury to the
cartilage of the articular surface and/or the potential for repair or
the risk of posttraumatic degeneration of the articular cartilage.
Plain radiographs and CT scans provide very little information about
the current and future health of the articular cartilage in a joint
with a fracture.
well-trained, will have some level of variability in applying any
tool—no matter how reliable—in classifying fractures. The magnitude of
the “baseline” level of inherent human variability in fracture
classification is entirely unknown. As such, it is extremely difficult
for investigators to know with precision what represents excellent
interobserver reliability in fracture classification. There is
disagreement over the best statistical analysis to use in assessing
reliability or what level of agreement is acceptable in studies on
fracture classification. Statistics such as the intraclass correlation
coefficient are very good as indicators of when a laboratory test, such
as the hematocrit or serum calcium level, has acceptable reliability
and reproducibility. Whether the same threshold level of reliability
should be applied to a process such as fracture classification is
unknown. Similarly, the interpretation of the weighted kappa statistic
for fracture classification is somewhat difficult, since there are few
guidelines to aid in interpreting their results. Landis and Koch admit
that their widely accepted reference intervals for the kappa statistic
were chosen arbitrarily. Additionally, a recent investigation seemed to
indicate that using the kappa statistic with a small number of
observers introduces the possibility of “sampling error” causing an
increased variance in the kappa statistic itself.4,57
Having many different observers causes stabilization of the kappa value
around a “mean value” for the agreement among the population of
observers. Invariably, however, using more observers results in a lower
mean kappa value and indicated poorer interobserver reliability of the
classification system being tested. Therefore, studies with a small
amount of observers that reported excellent reliability in fracture
classification systems may be reporting spuriously high results for the
kappa statistic—results that would be much lower if more observers were
used. Unfortunately, there are currently no better or more reliable
methods for reporting and interpreting interobserver reliability than
the use of the ICC or the kappa statistic.
describing fractures; this has been one of the best uses for fracture
classification systems. Using a well-known fracture classification to
describe a fracture to an orthopaedist or colleague who cannot
immediately view the fracture radiographs immediately invokes in the
orthopaedist a visual image of the fracture. This visual image, even if
it is not highly reliable to statistical testing, enhances
communication between orthopaedic physicians.
educational tools. Educating orthopaedic trainees in systems of
fracture classification is highly valuable, for many systems are
devised from the mechanism of injury or from the anatomical alignment
of the fracture fragments. These are important educational tools to
assist orthopaedic trainees in better understanding the osteology of
different parts of the skeleton and the various mechanisms of injury
that can result in fractures. Educational systems using fracture
classification methodologies can assist orthopaedic trainees in
formulating a context in which to make treatment decisions, and can
also provide an important historical context of fracture care and
fracture classification in orthopaedics.
treatment, and it is clearly the intent of many fracture classification
systems to do so. It is unclear, however, from much of the literature
that has been published, whether fracture classification systems are
valid tools to guide treatment. The fact that there is so much observer
variability in fracture classification adds an element of doubt to
comparative clinical studies that have used fracture classification as
a guide to treatment.
be useful in predicting outcomes following fracture care. The
orthopaedic literature to date, however, does not seem to clearly
indicate that fracture classification systems can be used to predict
patient outcomes in any sort of valid or reproducible way. The
interobserver variability of many fracture classification systems is
one of the key reasons that the literature cannot clearly show this
correlation. One exception to this, however, is that most fracture
classification system have good reliability in characterizing the most
severe and least severe injuries—those that correlate with the best and
worst outcomes. It is in the midrange of injury severity that
classification systems demonstrate the poorest reliability and the
poorest ability to predict outcomes.
determination of injury severity than merely classifying a fracture
according to plain radiographs. It has become clear in recent years
that variables other than radiograph appearance of the fracture play a
huge role in determining patient outcome, and these variables will be
utilized in new systems of determining injury severity in patients with
fractures. Objective measures of energy of injury include CT scans,
finite element models or volumetric measures,
measures
of the extent of injury to soft tissues, objective measures of the
patient’s physiologic reserve and response to injury, and serum lactate
levels. An assessment of overall health status and the existence of
comorbid conditions are other ways that may be used to make more
comprehensive the determination of fracture severity. These factors
will likely be combined with the radiographic appearance of the
fracture to better guide treatment and to better predict outcomes of
fracture care.
and more reliably determining and characterizing the injury severity in
patients with fractures. Newer uses for CT scanning and MRI imaging and
ultrasound will be instrumental in providing the treating surgeon more
information about the extent of soft tissue injury, the health of the
bone and cartilage, and the biology at the fracture site. In addition,
we may gain additional information about the patient’s ability to heal
well. All of these will advance the orthopaedist’s ability to determine
injury severity. For example it is possible with very high-energy MRI
scans to determine the proteoglycan content of articular cartilage.
Since articular cartilage is not imaged on CT or plain radiographic
imaging, its health has been generally excluded from the classification
of fractures. However, the long-term health of the articular cartilage
is crucial to the patient’s outcome following a severe articular
injury. In the future, the ability to use advanced imaging modalities
to better characterize the current health and predict the future health
of the articular cartilage will be a great advancement in our ability
to accurately classify fractures and to use fracture classification as
a predictive measure.
that will better assure that fractures can be measured and
characterized on a continuum, which is how they occur. These new
classification systems will better represent the continual aspects of
injury severity than do systems in use today, many of which were based
simply on anatomical consideration rather than on injury severity.
Ideas such as rank-ordering fractures, putting fractures on a
continuum, sending fractures to a fracture classification clearing
house (for classification by one or just a few observers) are but a few
possible future approaches to advancing and making more reproducible
the classification of fractures.
process of validation a fracture classification system should undergo
before becoming available for general use. Most classification systems
in general use have had no formal validation. Most of them have come
into general use because of the reputation or influence of the person
or group that devised them, or perhaps because the system has been in
use so long that it has simply become part of the vernacular in
fracture classification and fracture care. One study has proposed a
formal, detailed, and very time-consuming methodology for the
validation of all fracture classification systems, very similar to that
which was performed for patient-based outcome measures, such as the
short form 36 and the musculoskeletal functional assessment.4
It is as of yet unclear whether such validation methods would improve
the interobserver reliability of fracture classification systems. It is
clear, however, that such methods would be exhaustive and very time
consuming, and that many orthopaedic surgeons do not believe that such
detailed validation is necessary for fracture classification systems.
will advance our understanding and ability to classify fractures.
Advances in imaging processes and image analysis, perhaps when coupled
with neural nets and other learning technologies, may make it possible
for computers to be taught to classify fractures with a high degree of
reliability and reproducibility. One could envision a system by which
digital images of a fracture are classified according to any of several
classification systems and that will be done automatically by a
computer system at the time the radiographs are obtained, much as
electrocardiograph (EKG) readings are currently generated by a computer
at the time the patient’s cardiac tracing is obtained.
fracture classification systems. Rigorous statistical methods—or at
least consensus statistical methodologies—will be developed and
implemented that, while detailed, time consuming, and involved, will
result in greatly improved validation of many fracture classification
systems.
DJ, Blair WF, Steyers CM, et al. Classification of distal radius
fractures: an analysis of interobserver reliability and intraobserver
reproducibility. J Hand Surg 1996; 21A:574-582.
E, Jorgensen LG, Hededam LT. Evans classification of trochanteric
fractures: an assessment for the interobserver reliability and
intraobserver reproducibility. Injury 1990;21:377-378.
GR, Rasmussen JB, Dahl B, et al. Older’s classification of Colle
fractures: good intraobserver and interobserver reproducibility in 185
cases. Acta Orthop Scan 1991; 62:463-464.
L, Bhandari M, Kellam J. How reliabile are reliability studies of
fracture classifications? A systematic review of their methodologies.
Acta Orthop Scand 2004;75:184-194.
L, Anderson J, Chesnut R, et al. Reliability and reproducibility of
dens fracture classification with use of plain radiography and
reformatted computer-aided tomography. J Bone Joint Surg Am
2006;88:106-112.
PE, Dorey FJ, Matta JM. Letournel classification for acetabular
fractures: assessment of interobserver and intraobserver reliability. J
Bone Joint Surg Am 2003;85: 1704-1709.
J, Adler LM, Blank JE, et al. Evaluation of the Neer system of
classification of proximal humeral fractures with computerized
tomographic scans and plain radiographs. J Bone Joint Surg Am
1996;78:1371-1375.
J Jr, Goldfarb C, Catalano L, et al. Assessment of articular fragment
displacement in acetabular fractures: a comparison of computed
tomography and plain radiographs. J Orthop Trauma 2002;16:449-456.
S, Bagger J, Sylvest A, et al. Low agreement among 24 doctors using the
Neer classification; only moderate agreement on displacement, even
between specialists. International Orthop 2002;26:271-273.
RJ, Jones AL. Interobserver agreement in the classification of open
fractures of the tibia. J Bone Joint Surg Am 1994;76:1162-1166.
PSH, Klimkiewicz JJ, Luchette WT, et al. Impact of CT scan on treatment
plan and fracture classification of tibial plateau fractures. J Orthop
Trauma 1997;11:484-489.
RJ, Bindra RR, Evanoff BA, et al. Radiographic evaluation of osseous
displacement following intraarticular fracture of the distal radius:
reliability of plain radiographs versus computed tomography. J Hand
Surg (Am) 1997;22:792-800.
III, WL, Dirschl DR. An assessment of the effectiveness of binary
decision-making in improving the reliability of the AO/ASIF
classification of fractures of the ankle. J Orthop Trauma
1998;12:280-284.
HC, Franck WM, Sabauri G, et al. Incorrect classification of
extra-articular distal radius fractures by conventional x-rays:
comparison between biplanar radiologic diagnostics and CT assessment of
fracture morphology. Unfallchirurg 2004;107(6):491-498.
TA, Willis MC, Marsh JL, et al. Rank order analysis of tibial plafond
fracture: does injury or reduction predict outcome? Foot and Ankle
International 1999;20:44-49.
VV, Davis TRC, Barton NJ. The prognostic value and reproducibility of
the radiological features of the fractured scaphoid. J Hand Surg Br
1999;5:586-590.
DR, Ferry ST. Reliability of classification of fractures of the tibial
plafond according to a rank order method. J Trauma 2006;61:1463-1466.
DR, Adams GL. A critical assessment of methods to improve reliability
in the classification of fractures, using fractures of the tibial
plafond as a model. J Orthop Trauma 1997;11:471-476.
DR, Marsh JL, Buckwalter J, et al. The clinical and basic science of
articular fractures. Accepted by J American Acad Orthop Surg, September
2002, 30 pages.
J, Lindenhovius A, Kloen P, et al. Two- and three-dimensional computed
tomography for the classification and management of distal humeral
fractures. J Bone Joint Surg Am 2006;88:1795-1801.
PA, Andersen E, Madsen F, et al. Garden classification of femoral neck
fractures: an assessment of interobserver variation. J Bone Joint Surg
Br 1988;70:588-590.
RB, Anderson JT. Prediction of infection in the treatment of 1025 open
fractures in long bones. J Bone Joint Surg 1976;58A:453-458.
RB, Mendoza RM, Williams DN. Problems in the management of Type III
(severe) open fractures: a new classification of type III open
fractures. J Trauma 1984;24(8): 742-746.
RB, Merkow RL, Templeman D. Current concepts review: the management of
open fractures. J Bone Joint Surg Am 1990;72:299-303.
CA, Dirschl DR, Ellis TJ. Interobserver reliability of a CT-based
fracture classification system. J Orthop Trauma 2005;19:616-622.
MA, Beredjiklian PK, Bozentka DJ, et al. Computed tomography scanning
of intraarticular distal radius fractures: does it influence treatment?
J Hand Surg Am 2001;26(3): 415-421.
HJ, Hanel DP, McKee M, et al. Consistency of AO fracture classification
for the distal radius. J Bone Joint Surg Br 1996;78:726-731.
B, Andersen ULS, Olsen CA, et al. The Neer classification of fractures
of the proximal humerus: an assessment of interobserver variation.
Skeletal Radiol 1988;17: 420-422.
AJ, Inda DJ, Bott AM, et al. Interobserver and intraobserver
reliability of two classification systems for intra-articular calcaneal
fractures. Foot and Ankle International 2006;27:251-255.
JL, Buckwalter J, Gelberman RC, et al. Does an anatomic reduction
really change the result in the management of articular fractures? J
Bone Joint Surg 2002;84-A: 1259-1271.
JS, Marsh JL, Bonar SK, et al. Assessment of the AO/ASIF fracture
classification for the distal tibia. J Orthop Trauma 1997;11:477-483.
J, Marsh JL, Nepola JV, et al. Radiographic fracture assessments: which
ones can we reliably make? J Orthop Trauma 2000;14(6):379-385.
RB, Carr SEW, Watson JT. Open reduction and internal fixation of
posterior wall fractures of the acetabulum. Clin Orthop and Rel Res
2000;377:57-67.
ME. The comprehensive classification of fractures of long bone. In:
Muller ME, Allgower M, Schneider R, et al, eds. Manual of Internal
Fixation: Techniques Recommended by the AO-ASIF Group. 3rd Ed.
Heidelberg: Springer-Verlag, 1991.
JO, Dons-Jensen H, Sorensen HT. Lauge-Hansen classification of
malleolar fractures: an assessment of the reproducibility of 118 cases.
Acta Orthop Scand 1990;61: 385-387.
DA, Jackson KR, Davies MR, et al. The impact of the Garden
classification on proposed operative treatment. Clin Orthop and Rel Res
2003;409:232-240.
HJ, Txcherne H. Pathophysiology and classification of soft tissue
injuries associated with fractures. In: Tscherne H, ed. Fracture With
Soft Tissue Injuries. New York: Springer-Verlag, 1984:1-9.
FC, Ramos LMP, Simmermacher RKJ, et al. Classification of thoracic and
lumbar spine fractures: problems of reproducibility. Eur Spine J
2002;11:235-245.
H, Parker MJ, Pryor GA, et al. Classification of trochanteric fracture
of the proximal femur: a study of the reliability of current systems.
Injury 2002;33:713-715.
BA, Bhandari M, Orr RD, et al. Improving reliability in the
classification of fractures of the acetabulum. Arch Orthop Trauma Surg
2003;123:228-233.
S, Madsen PV, Bennicke K. Observer variation in the Lauge-Hansen
classification of ankle fractures: precision improved by instruction.
Actat Orthop Scand 1993; 64:693-694.
RD, Schork MA. Statistics with Applications to the Biological and
Health Sciences. Englewood Cliffs, NJ: Prentice-Hall, 1970.
J. Fractures of the tibial plateau. In Schatzker M, Tile M, eds.
Rationale of Operative Fractures Care. Berlin: Springer-Verlag,
1988:279-295.
IB, Steyerberg EW, Castelein RM, et al. Reliability of the AO/ASIF
classification for peritrochanteric femoral fractures. Acta Orthop
Scand 2001;72:36-41.
LE, Zalavras CG, Jaki K, et al. Gunshot femoral shaft fractures: is the
current classification system reliable? Clin Orthop and Rel Res
2003;408:101-109.
JL, Zuckerman JD, Lyon T, et al. The Neer classification system for
proximal humeral fractures: an assessment of interobserver reliability
and intraobserver reproducibility. J Bone Joint Surg Am
1993;75:1745-1750.
KA, Gerber C. The reproducibility of classification of fractures of the
proximal end of the humerus. J Bone Joint Surg Am 1993;75:1751-1755.
H, Starklint H, Gundersen HJ, et al. Reproducibility of
histomorphologic diagnoses with special reference to the kappa
statistic. APMIS 97 1989;689-698.
JF, Agel J, McAndrew MP, et al. Outcome validation of the AO/OTA
fracture classification system. J Orthop Trauma 2000;14:534-541.
JF, Sands AK, Agel J, et al. Interobserver variation in the AO/OTA
fracture classification system for pilon fractures: is there a problem?
J Orthop Trauma 1997;11: 467-470.
NOB, Olsen LH, Nielsen ST. Kappa statistics in the assessment of
observer variation: the significance of multiple observers classifying
ankle fractures. J Orthop Sci 2002;7:163-166.
NOB, Overgaard S, Olen LH, et al. Observer variation in the
radiographic classification of ankle fractures. J Bone and Joint Surg
Br 1991;73:676-678.
B, Chobrangsin P, Ketmalasiri B, et al. Evaluation of Letournel and
Judet classification of acetabular fracture with plain radiographs and
three-dimensional computerized tomographic scan. J Orthop Surg
2000;8:33-37.
NP, Harish S, Roberts C, et al. AO or Schatzker? How reliable is
classification of tibial plateau fractures? Arch Orthop Trauma Surg
2003;123:396-398.
S, Blaser PF, Blanc CH, et al. Comparison between standard radiography
and spiral CT with 3D reconstruction in the evaluation, classification
and management of tibial plateau fractures. Eur Radiol
2000;10:1227-1232 (showed change in surgical plan in 59% of cases, but
no diff in classification).
SV, Nevins RT, Sallis JG, et al. Impact of MRI on treatment plan and
fracture classification of tibial plateau fractures. J Orthop Trauma
2002;16:632-637.
and dislocation compendium (Orthopaedic Trauma Association Committee
for Coding and Classification). J Orthop Trauma 1996;10:Suppl. 1.
