Elbow Arthroplasty: Surgical Technique
IV – Elbow Reconstruction > Part C – Operative Treatment Methods
> 58 – Elbow Arthroplasty: Surgical Technique
treatment for the appropriately selected patient. The advent of
reliable devices in the 1980s allowed this joint to be replaced with
outcomes similar to other joints. Not that total elbow arthroplasty is
without its potential complications or problems. At least as important
as in other joints, if not more important because of potential
pitfalls, proper surgical technique is paramount for a successful
arthroplasty.
approach and various technical details; although much about total elbow
arthroplasty surgical technique is not implant design specific. Current
devices are most easily categorized as being either linked or unlinked.
Constraint of the implant is important, but is not dependent on
linkage. A linked implant is best thought of as a hinge, with the ulnar
and humeral components coupled, or linked. Initial designs had no play
or toggle between the components, and these devices failed quickly.
Modern-design linked implants allow for angular and rotational play
between the humeral and ulnar components. Unlinked components have no
direct connection between the components. The amount of congruity or
conformity between the articulating surfaces of the implants dictates
the constraint of the device. A highly congruent articulation has more
constraint than an articulation with little conformity. Thus, a linked
or unlinked device can be either constrained or unconstrained. An
unlinked device with little constraint has the highest risk of
dislocation; particular attention has to be given to both pre-existing
deformity and adequate soft tissue repair, especially the ligaments. A
linked device cannot, by design, dislocate; but, attention to
pre-existing deformity is just as important as when considering an
unlinked design.
will be discussed in the following chapters. General indications for a
total elbow arthroplasty include a painful elbow joint that does not
have less-extensive reconstructive options available. Adequate bone
stock for reconstruction with the chosen prosthesis is necessary, and
the surgeon should have familiarity with the technical limitations of
the prosthesis considered for implantation. Bone deficit can be
compensated for by some devices, but each device has limitations.
Custom devices are rarely if ever needed. A functional elbow flexor is
necessary. A competent extensor mechanism is considered important; but,
a functional limb can be reconstructed without, if the patient is
willing to accept a compromise. Gravity is then used for elbow
extension, and overhead function with the involved limb is not
possible. An adequate soft tissue envelope is necessary, especially
with the higher risk of infection in total elbow arthroplasty.
Understanding of and compliance with the restrictions inherent to any
total elbow arthroplasty is a mandatory requisite for surgery.
Longevity of newer implants has improved to parallel hip arthroplasty
in rheumatoid patients. Failures have been noted in patients placing
high demands on their reconstructed joint. Because of this, most
surgeons have placed the permanent restriction on their patients of not
lifting >10 pounds as a single, occasional event, and 1 to 2 pounds
as a repetitive event.
approaches to the elbow, but a posterior incision allows for much
freedom in the deeper exposure. Although a laterally based skin
incision (and deeper exposure) has been used in elbow arthroplasty
(especially some unlinked designs), it does have inherent limitations.
This author routinely uses a universal posterior skin incision for
elbow arthroplasty. Multiple approaches can be easily accomplished, and
future revision surgery is not compromised by other skin incisions.
Fewer problems with the cutaneous nerves are encountered when the
incision is placed posteriorly. Many elbows requiring arthroplasty have
pre-existing deformity, which can be challenging to correct. Laterally
based approaches can make a challenging deformity nearly impossible to
correct.
arthroplasty, there are three structures that require consideration
regardless of the implant used. These include the triceps, the ulnar
nerve, and the collateral ligaments. The implant used may dictate
certain needs, but each of these three factors must be considered in
every elbow arthroplasty.
complications with triceps reattachment after elbow arthroplasty.
Postoperative weakness has been attributed to failure of the repair
site of triceps to the olecranon. This has caused a scrutiny of current
techniques and reconsideration of alternative methods to deal with the
triceps. Most available implants for elbow arthroplasty recommend that
the triceps be released or transected at some level. The ideal method
of release is unknown. Although more challenging, implants can be
placed through a medial and/or lateral deep exposure. The triceps can
then be left alone. Visualization is more difficult and extreme care
must be taken to ensure proper implant orientation.
occurs when treating chronic nonunions with a linked prosthesis. As
described by Morrey, these are readily approached by excising the
distal humeral fragment(s). The epicondyles are, by default, excised,
but this does not compromise the result. By excising the distal
humerus, the triceps can be left attached to the olecranon and the
components can be implanted through the gap from the resection. This
has the advantage of allowing full triceps activity without the need to
protect a repair. If surgical exposure is adequate from this approach,
the risk of extensor weakness should be eliminated. The potential
drawback of the approach is the limitation of exposure and the
potential inability to correct significant deformity. A similar
technique has also been described for routine elbow linked arthroplasty
by Matsen.
needs to be released from the olecranon is some fashion. Early
techniques (Campbell/Van Gorder) involved leaving a tongue of triceps
tendon attached to the olecranon, detaching the tendon proximally in a
“V” fashion. This did allow for a V-Y lengthening if needed and also
did not disturb the Sharpey fibers attachment of the triceps. This
approach has been essentially abandoned for modern elbow arthroplasty,
but as concern regarding the healing potential of the triceps to the
olecranon is raised, it could once again be considered.
 |
|
Figure 58-1
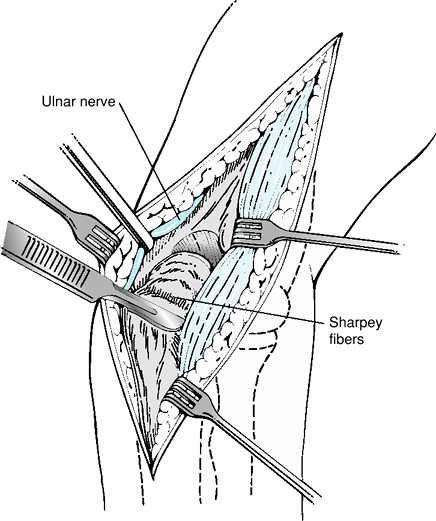
The Bryan-Morrey, or Mayo, approach reflects the triceps with the anconeus laterally. The distal extent continues to the extensor musculature and fascia of the forearm, which is kept in continuity with the triceps. |
reflection of the triceps laterally off of the olecranon. The anconeus
is kept in continuity with the triceps, and the whole sleeve of tissue,
with the extensor fascia, is reflected laterally (Fig. 58-1).
tunnels to pass sutures. This technique allows for complete separation
of the olecranon from the triceps. If the reattachment does not heal
completely, there will be at minimum triceps weakness and possibly
complete extensor mechanism failure. The tissue distal to the Sharpey
fibers can be quite atrophic, resulting in a defect of the sleeve of
soft tissue. This approach can also be performed leaving a small wafer
of bone on the triceps to allow for precise attachment back to the
olecranon and also allow for bone-to-bone healing of the extensor
mechanism. Unfortunately, in clinical practice, this addition did not
bear out any advantage to the all–soft tissue approach.
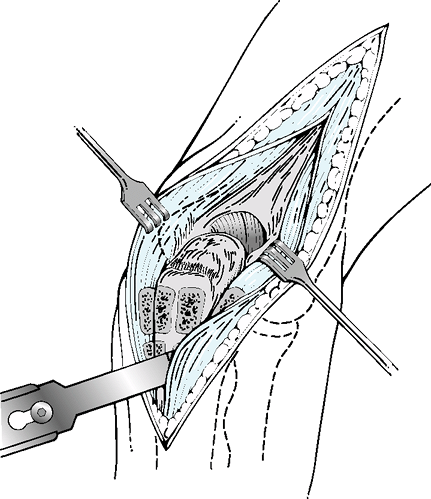
This is a midline triceps splitting approach. As the distal dissection
is undertaken, the split continues along the subcutaneous border of the
ulna.
As originally described, an osteotome is then used to remove the medial
and lateral cortex of the ulna with the soft tissue attachments. The
collateral ligaments can be left in situ, but releasing them allows for
significantly more exposure. The split and the bone wafers are
reattached through drill holes at closure. This approach can be done
strictly as a soft tissue exposure by carrying the dissection along the
medial and lateral ulna, forgoing any bone resection.
 |
|
Figure 58-2 The Gschwend approach splits the triceps and continues distally, osteotomizing triceps attachment with bone from the olecranon.
|
of the ulnar nerve should be documented. If there has been previous
surgery, attempts should be made to study the old operative records. In
cases of old trauma or surgery, one should never assume the location of
the ulnar nerve. It must be identified and protected throughout the
procedure. The proximal medial triceps border is usually a reliable
location to identify the nerve in an elbow with previous surgery.
standard practice by many surgeons regularly performing elbow
arthroplasty. The nerve can be safely removed from the primary surgical
field and should not cause further difficulty in the future, should a
revision be needed. Although there is always risk when transposing the
ulnar nerve, the risk is far less than that incurred by leaving it in
the cubital tunnel. A transposition was not originally described by
Gschwend in his triceps splitting approach. At a minimum the nerve
needs to be identified in this technique and can be readily transposed
with little additional effort.
allow dislocation or subluxation of the elbow sufficient to allow for
implantation of any total elbow device. Releasing the medial collateral
ligament will allow for better correction of any pre-existing deformity
and give better access. If the device implanted is linked, neither
ligament needs to be repaired.
must be repaired or reconstructed. Although a medial collateral
ligament–deficient elbow may not dislocate in the native state, the
increased instability will jeopardize an unlinked implant, and
consideration should be given to repair or reconstruction.
cutting blocks and guides. However, there are some general principles
for humeral preparation. The posterior aspect of the capitellum is the
origin for the anconeus. The muscle should be dissected off of this
area to allow for complete exposure of the lateral column.
the humeral component. Although consisting of cortical bone, the
columns are narrow, and if notched or too generous a cut is made, the
column may fracture, separating the epicondyle from the humerus. If
this occurs, the most efficacious treatment is to excise the fractured
fragment if a linked implant is being placed. The loss of the
epicondyle and associated collateral will not have bearing on the
outcome. Attempts at fixation of these small fragments have little to
no net benefit. If the device is unlinked, this fracture will have
significant bearing on the potential stability. Either the fragment has
to be fixed or a linked device has to be used. If an unlinked device is
used, fixation can be done with a tension band and wires, or if the
fragment is large, a unicortical plate.
plane. Because of this, if the starting hole is not proximal enough, or
attention is not paid to the opening, there may be remnant cortical
bone tapering inward. This obscures the true diameter of the humeral
canal, and more important, can cause the humeral instrumentation or
implant to be pushed anterior or posterior. It is imperative to
recognize this, and if the canal is not accepting the rasps or trials,
to ensure that a sufficient amount of the distal humerus is removed to
allow a straight approach up the medullary canal. If it is apparent
that there is impeding bone, it is often easiest to remove with a
rongeur.
excised to allow for instrumentation of the medullary canal and
implantation of the device. The attachment site for the triceps must be
retained. Like the humerus, the opening
to
the medullary canal may not initially allow for correct broaching or
rasping. This is especially true in patients with osteoarthritis and/or
dense bone. When the subchondral bone is too dense, using a burr to
open the canal will save time and prevent potential catastrophic
fracture of the proximal ulna.
technique for implantation. However, some universal concepts are worthy
of consideration.
linked, or implanted all at once already linked. The advantage of
implanting each component separately is the ability to focus technique
on each side individually. The disadvantage is that of having to mix
two separate batches of cement and the extra time it takes for the two
sides to cure sequentially. The advantages of separate component
implantation far outweigh the disadvantages for the surgeon with
limited experience implanting total elbows. Linked implantation should
be done only by surgeons with high-volume experience.
for most systems, help with proper rotational alignment. The anterior
flange on many humeral components also aids in rotational alignment.
The flange’s main purpose is to resist posterior and rotationally
directed forces on the humeral component. These stresses are
significant and can lead to early failure if not neutralized. For this
reason, the flange should be grafted if the technique calls for it.
Evidence of the stress on the flange is noted postoperatively, when
most of these grafts heal and many hypertrophy.
 |
|
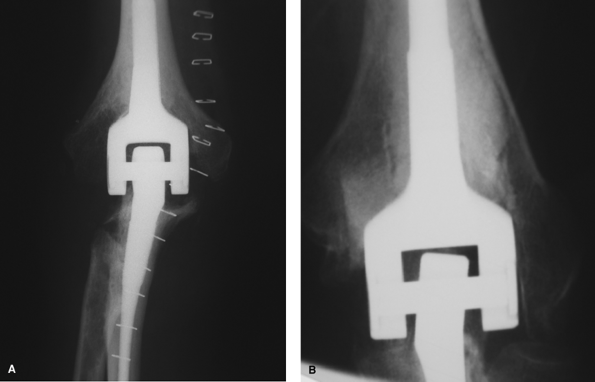
Figure 58-3 Plain radiographs of a balanced (A)and unbalanced (B) linked total elbow arthroplasty.
|
for in the ulnar component. Once the medullary canal is prepared, it is
cylindrical, allowing for rotation of the component. Some devices
account for this and have flanges or flat posterior aspects of the
component. Even with these additions, if one is not careful, the rasps
and trials (and eventually the final components) can be placed with
rotation. This will cause undue stress on the coupling in a linked
component and potential for dislocation in an unlinked device. There
are two reliable methods to ensure proper rotational alignment of the
ulnar component. As described by King, the flat spot on the dorsal ulna
is almost perfectly perpendicular to the plane of the greater sigmoid
notch. O’Driscoll has recently described the use of the radial head (or
shaft if the head has been resected) to align the ulnar component. The
ulnar component must be aligned so that the flexion axis of the device
passes through the radial head (or shaft) center when viewing from end
on.
using the trial implants. This is especially true in elbows with
significant pre-existing deformity. Unlinked devices have a high
dislocation rate. Unbalanced devices that are
not
linked are even more likely to dislocate. A linked device will not
dislocate, but if unbalanced, will load the bearings more than will a
balanced elbow (Fig. 58-3). This has been proposed as a cause of early failure.
approach that was used. Every effort should be made to reattach the
triceps to its native location. Marking the area of the Sharpey fibers
attachment with a suture can ensure later reattachment to the proper
location. If the triceps was detached only as soft tissue, drill holes
should be used for suture passage of no. 2 or no. 5 nonabsorbable
sutures to secure the repair. One can usually pass straight needles
directly through the ulna with a pin driver, obviating the need to
drill, and then pass the sutures with another device.
necessary for adequate exposure, they need not be repaired in the case
of a linked device. If the device implanted is unlinked, stability must
be restored or else the chance of dislocation is high. The ligaments
should be repaired, or if this is not possible, reconstructed.
to the skin flaps created during dissection. To decrease the likelihood
or severity of this, the subcutaneous tissue can be tacked down to the
fascia to close this potential space. When doing this, the cutaneous
nerves must be avoided to prevent neuromas.
postoperatively, splinting in full extension is helpful. This allows a
compressive bandage to be applied, and the elbow can then be elevated
or hung in a stockinette to decrease swelling.
N, Dunning CE, Johnson JA, et al. The flat spot of the proximal ulna: a
useful anatomic landmark in total elbow arthroplasty. J Shoulder Elbow Surg. 2004;13:206–207.
DR, Morrey BF. The Coonrad-Morrey total elbow arthroplasty in patients
who have rheumatoid arthritis. A ten to fifteen-year follow-up study. J Bone Joint Surg Am. 1988;80:1327–1335.
F, O’Driscoll S, Korinek S, et al. Loose-hinge total elbow
arthroplasty. An experimental study of the effects of implant alignment
on three-dimensional elbow kinematics. J Arthroplasty. 1995;10:670–678.
