CHAPTER 72 –
Cole & Sekiya: Surgical Techniques of the Shoulder, Elbow and Knee in Sports Medicine, 1st ed.
Copyright ©
2008 Saunders, An Imprint of Elsevier
CHAPTER 72 – Medial Patellofemoral Ligament Reconstruction and Repair for Patellar Instability
Numerous surgical procedures have been described for treatment of patellar instability, most with generally favorable success rates. The medial patellofemoral ligament (MPFL) is the primary soft tissue passive restraint to pathologic lateral patellar displacement [4] [7] [17] and is torn when the patella dislocates. [15] [19] [23] There has been a great deal of interest recently in soft tissue procedures that address the MPFL. Techniques have been described to repair [5] [13] [15] [23] or to reconstruct [8] [18] [24] the MPFL in an attempt to restore its function as a checkrein. Regardless of which approach is taken, successful surgical treatment requires that the surgeon have a thorough understanding of the relevant anatomy and a working knowledge of patellofemoral biomechanics.
Patellofemoral complaints are among the most common problems encountered by physicians treating knee disorders, and instability represents a distinct subset that is usually amenable to surgical treatment. Instability represents a continuum ranging from minor incidental subluxation episodes to traumatic dislocation events. Patients with frequent subluxation episodes and patients who dislocate usually experience substantial knee pain, swelling, and stiffness, resulting in interruption of their normal occupational and recreational activities.
Patellar dislocations can occur from an indirect twisting mechanism as the upper body rotates while the foot remains planted on the ground. Less commonly, a direct blow to the medial aspect of the patella from contact sport or motor vehicle accident drives the patella laterally. The patella may be spontaneously reduced as the knee is extended, or a formal reduction maneuver may be necessary. With initial dislocation episodes, significant pain and swelling are caused by soft tissue and articular surface damage. The resulting hemarthrosis and quadriceps weakness may take several weeks to resolve. The degree of morbidity tends to decrease in patients who sustain multiple recurrent episodes.
Subluxation episodes are usually less dramatic and manifest as a feeling of instability and pain. Patients often describe the sense that the knee may “give out.” These episodes usually occur with trunk rotation during physical activity and result in a variable degree of pain and swelling. The pain is usually anterior and may be bilateral, especially in patients with malalignment or diffuse ligamentous laxity. The most important clinical determination to be made is whether the pain described by the patient is associated with patellar instability, as the common clinical entity of isolated anterior knee pain (patellofemoral pain syndrome) is nearly universally treated nonoperatively.
Tibiofemoral alignment is evaluated with the patient standing. The knee is then observed for intraarticular and extra-articular swelling, and the knee’s range of motion is formally measured and compared with the contralateral leg. The soft tissues are palpated for areas of tenderness. The examiner should try to identify the area of greatest tenderness along the course of the MPFL; this usually identifies the location of the tear.
A thorough ligamentous examination is necessary to rule out concomitant cruciate or collateral ligament tears. It is not uncommon to confuse the symptoms of a torn anterior cruciate ligament with patellar instability. Medial collateral ligament injuries occur commonly at the time of patellar dislocation. Measurement of the quadriceps angle (Q angle) can be used as a gross assessment of the lateral force vector. Patellar tracking is observed during active knee extension. The patella is observed for a tendency to slip laterally as the knee approaches the last 20 degrees of extension and the patella is no longer constrained by the lateral trochlear ridge (J sign). Patellar translation is estimated by applying a laterally directed force to the medial side of the patella with the knee in extension. The examiner attempts to quantify the amount of translation in quadrants and the consistency of the endpoint. An indistinct or “soft” endpoint suggests MPFL incompetence. A sense of apprehension with this maneuver (apprehension sign) supports the diagnosis of instability. Conversely, apprehension with medial translation may suggest medial instability. Lateral retinacular tightness is assessed by attempting to lift the lateral edge of the patella (tilt test). The retinaculum is considered tight if the patella will not correct to a neutral or horizontal position.
The standard radiographic series includes anteroposterior, lateral (30 degrees of flexion), tunnel, and sunrise (30 to 45 degrees of flexion) views. The tunnel view is useful to demonstrate osteochondral lesions or loose bodies in the notch. The lateral view gives information about patellar height and trochlear morphologic features. The tangential view shows the degree of subluxation and tilt as well as any osteochondral lesions.
Computed tomographic axial images (20 degrees of flexion) are helpful in some cases to quantify subluxation and tilt. Magnetic resonance imaging will demonstrate a characteristic bone bruise pattern affecting the medial facet of the patella and the lateral femoral condyle in patients who have recently sustained a traumatic dislocation. Chondral and osteochondral injuries are clearly localized by magnetic resonance imaging.
Indications and Contraindications
MPFL reconstruction is indicated in skeletally mature patients with symptomatic recurrent lateral subluxation or dislocation episodes. Attempts at nonoperative treatment, including activity modification, physical therapy, and bracing, have failed in most patients. Femoral fixation can be modified in skeletally immature patients so that the distal femoral growth plate is not at risk. Tibial tuberosity osteotomy procedures like the Elmslie-Trillat, which directly decrease the Q angle, have a theoretical advantage in patients with greater degrees of malalignment. The Fulkerson anteromedialization osteotomy is preferred in patients with malalignment and degenerative changes.[14] MPFL reconstruction or repair can be combined with distal osteotomy if neither alone is sufficient to provide adequate stability. Lateral retinacular release is reserved for patients with excessive lateral patellofemoral pressure and is ineffective as an isolated procedure in patients with instability.
Some authors recommend MPFL repair after first-time patellar dislocation.[23] I usually treat an initial subluxation or dislocation episode nonoperatively but will repair an acute MPFL tear when surgery is indicated for concomitant intraarticular disease (such as a large loose body or meniscus tear). MPFL repair may also be used to treat recurrent instability. With repeated instability episodes, the MPFL becomes attenuated and functionally incompetent. To re-establish the normal checkrein effect, the MPFL is tightened by cutting, shortening, and reattaching it at the patellar or femoral insertion or by midsubstance imbrication.
MPFL repair or reconstruction is contraindicated in patients with medial instability or isolated anterior knee pain. In patients with significant medial patellofemoral degenerative changes, great care should be taken not to overtighten the MPFL as this will result in excessive medial joint pressures and is likely to exacerbate patellofemoral pain.
Surgery is performed with the patient supine on a standard operating room table. A vertical thigh post is used to facilitate arthroscopic evaluation and is removed before the reconstruction is started. A tourniquet is placed around the proximal thigh on the operative side. A compressive thigh-high stocking is placed on the contralateral leg. The procedure is performed on an outpatient basis with use of general or regional anesthesia techniques. Prophylactic intravenous antibiotics are administered before incision.
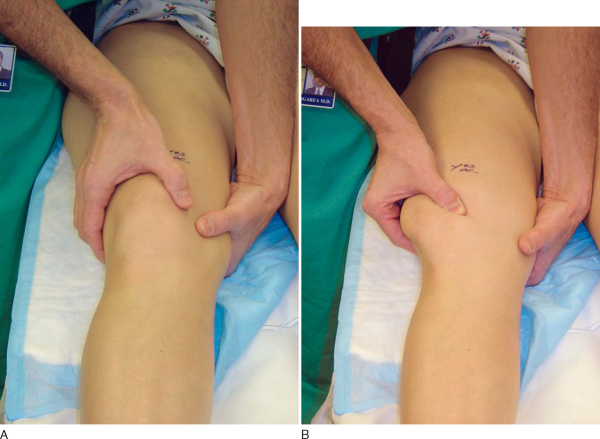
Once adequate anesthesia has been established, a comprehensive examination is performed. It is usually easier to characterize patellar stability, translation, and tilt when the patient is anesthetized. With the knee in extension, the position of the patella is determined at rest and with a lateral translation force applied (
Fig. 72-1
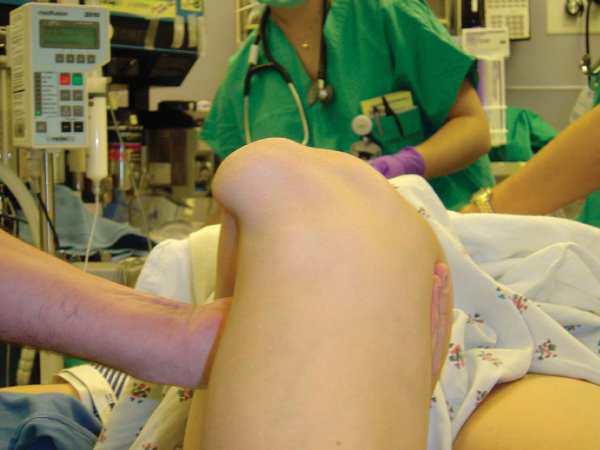
). Even an unstable patella will not stay dislocated unless the knee is maintained in a flexed position (
Fig. 72-2
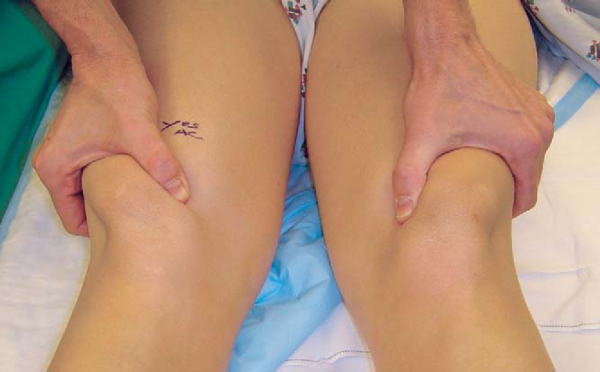
). It is important to compare the amount of translation on the symptomatic side with the normal lateral patellar translation in the contralateral knee. The examiner should use his or her thumb to push the patella laterally and assess the amount of translation as well as the consistency of the endpoint (
Fig. 72-3
). It is also important to assess the patient for lateral retinacular tightness. If the examiner is unable to evert the lateral edge of the patella to the neutral or horizontal position, and if the patient’s symptoms and preoperative radiographs are consistent with excessive lateral pressure, consideration should be given to a concomitant arthroscopic lateral retinacular release.
|
|
|
|
Figure 72-1 |
|
|
|
|
Figure 72-3 |
Diagnostic arthroscopy is performed by standard superolateral, inferomedial, and inferolateral portals. The suprapatellar pouch, the medial and lateral parapatellar gutters, and the posteromedial and posterolateral compartments are carefully assessed for loose bodies. The articular surfaces of the patella and trochlea are thoroughly visualized for chondral lesions. Most often, the medial facet of the patella and the proximal portion of the lateral femoral condyle are injured after traumatic patellar dislocation. Hemorrhage in the soft tissue and a capsular defect along the medial edge of the patella suggest a recent traumatic avulsion of the MPFL insertion. Patellar tracking in the trochlear groove is visualized as the knee is flexed and extended. The menisci and cruciate ligaments are assessed for concomitant pathologic changes. Any significant chondral lesions are addressed surgically with débridement, marrow stimulation, or repair techniques as indicated. Careful consideration should be given to whether large defects should be unloaded by tibial tuberosity osteotomy techniques, such as anteromedialization. If there is excessive tightness of the lateral retinaculum, an arthroscopic release may be performed at this point, although we have not found that to be routinely necessary.
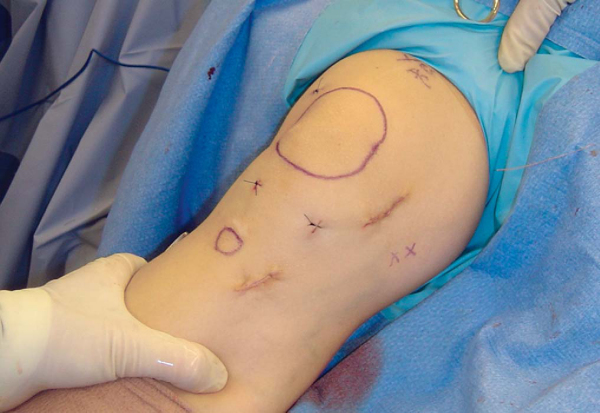
Surgical Landmarks and Incisions
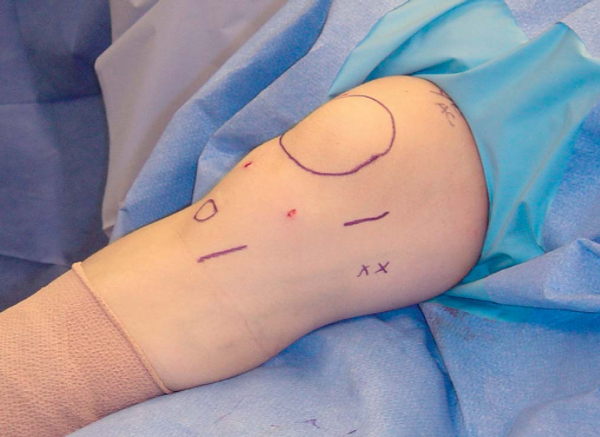
A marking pen is used to identify the location of the patella and tibial tuberosity (
Fig. 72-4
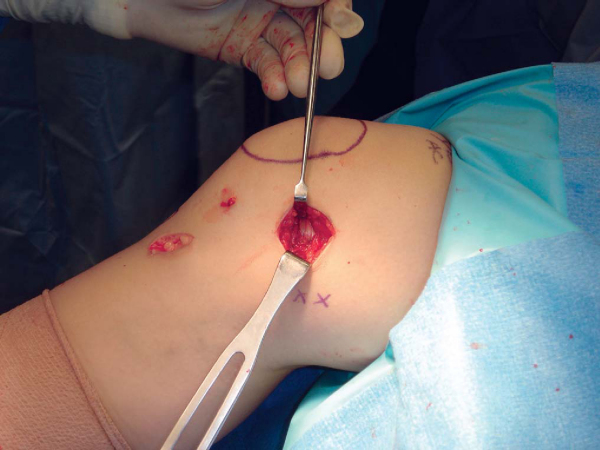
). Marks are also made over the adductor tubercle and just distally over the medial femoral epicondyle. The pes anserine tendon insertion site is localized by palpation. A 3- to 4-cm oblique mark is made for the harvest incision directly over the sartorial fascia insertion on the proximal medial tibia. A second 3- to 4-cm line is drawn directly over the midportion of the MPFL, halfway between the medial border of the patella and the medial femoral epicondyle. The position of this incision is modified if the location of the MPFL tear has been ascertained from the preoperative examination or magnetic resonance imaging and an MPFL repair rather than reconstruction is planned.
|
|
|
|
Figure 72-4 |
Specific Steps (
Box 72-1
)
The leg is exsanguinated with an Esmarch bandage, and the tourniquet is raised while the knee is in an extended position. A longitudinal incision is made directly over the MPFL midway between the medial edge of the patella and the medial epicondyle. Blunt dissection is used to expose the thick medial retinacular layer contiguous with the inferior border of the vastus medialis obliquus (
Fig. 72-5
). With gentle dissection, the MPFL is usually identifiable (
Fig. 72-6
). In the case of a recent dislocation, it may be possible to identify the site of failure at the medial border of the patella, at the medial femoral epicondyle, or less commonly in midsubstance.
| Surgical Steps | |||||||||||||||||||||
|
|
|
|
|
Figure 72-5 |
With a medially directed force applied to the lateral edge of the patella, the medial border of the patella is brought into the operative field. If the MPFL is avulsed at this location, a direct repair can be performed with bone tunnels or suture anchors. With retraction of the posterior edge of the wound, the adductor tubercle and medial femoral epicondyle can be exposed through the same incision. Some authors believe that the most common site of failure is off of the femur.[23] In this location, the torn MPFL can be repaired to a stump of remaining tissue with No. 2 nonabsorbable sutures or reattached directly to a freshened bone surface by two or three suture anchors. Repair of a midsubstance rupture is more challenging. It is not always possible to identify the location of tissue failure, and especially in chronic cases, it can be difficult to determine how much to shorten the attenuated ligament. Overtightening of the ligament will result in subsequent repair failure or overconstrain the patella. I generally reserve MPFL repair techniques for patients who have experienced recent instability episodes when acute surgical intervention is indicated for concomitant intraarticular disease.
3. Graft Harvest for MPFL Reconstruction
A variety of different autologous or allogeneic graft sources are available for reconstruction of the MPFL. I prefer to use the gracilis tendon, which is immediately adjacent to the reconstruction site and relatively easy to harvest. An incision is made directly over the pes anserine insertion, and blunt dissection is used to expose the sartorial fascia. The superior edge of the sartorial fascia is identified anterior to the superficial medial collateral ligament. The sartorial fascia is then incised and everted to expose the underlying gracilis and semitendinosus tendons. A combination of blunt and sharp dissection is used to separate the gracilis tendon from the sartorial fascia. The free end of the tendon is tagged, and a tendon stripper is then used to harvest the gracilis tendon. Although the gracilis is smaller and shorter than the semitendinosus tendon, it is still significantly stronger than the native MPFL [1] [3] [4] [16] and is almost always long enough to construct an adequate graft. After harvesting of the gracilis tendon, the sartorial fascia is sutured back to its insertion on the proximal medial tibia.
The gracilis tendon is then prepared for implantation by removal of muscle and other debris from the surface. The tendon is then folded over so that double-looped length can be measured. The diameter is also measured with a tunnel sizer. A double-looped graft length of 9 to 10 cm is sufficient and will be increased as necessary once the graft construct has been completed.
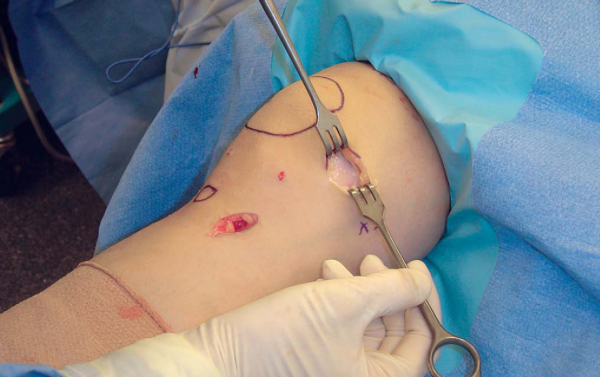
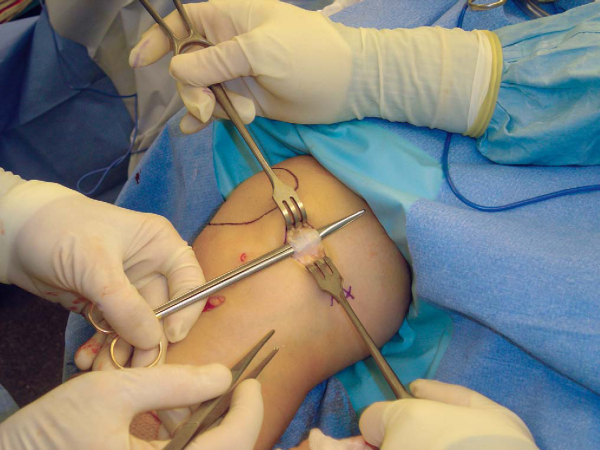
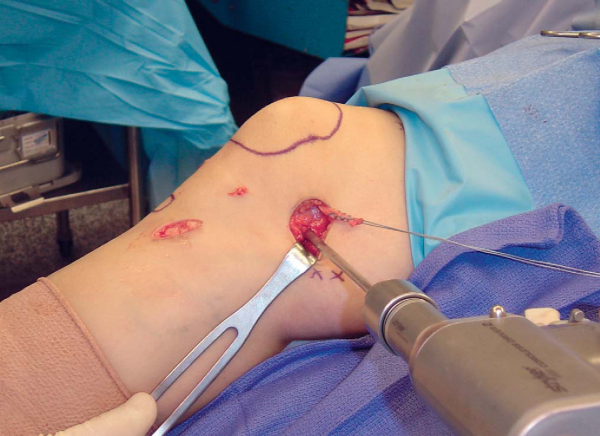
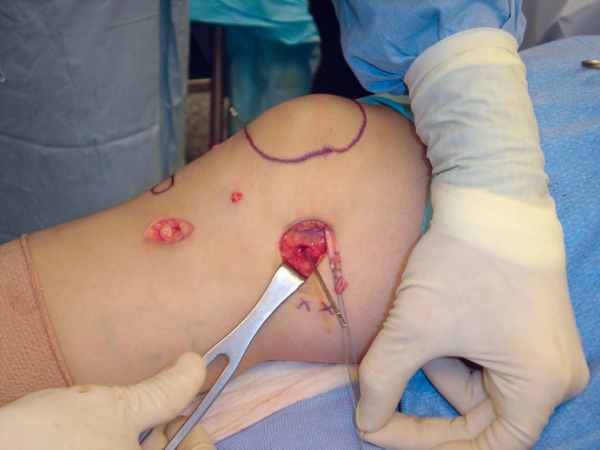
Soft tissue is cleared from the medial edge of the patella with a rongeur. The proximal and distal poles of the patella are localized digitally. A small transverse incision is made at the MPFL insertion on the medial edge of the patella (
Fig. 72-7
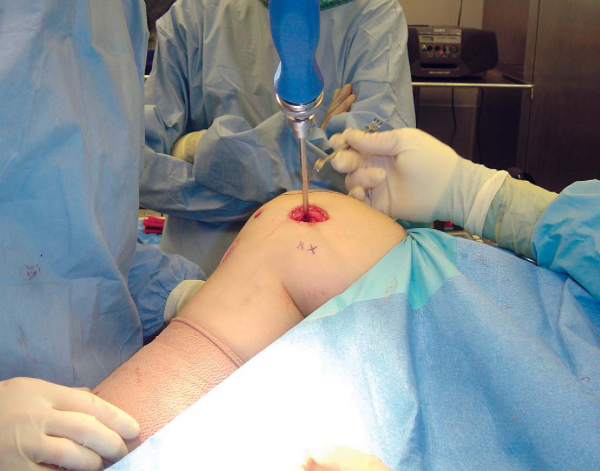
). A 2.5-mm drill bit is drilled from medial to lateral in the midportion of the MPFL insertion on the medial border of the patella (
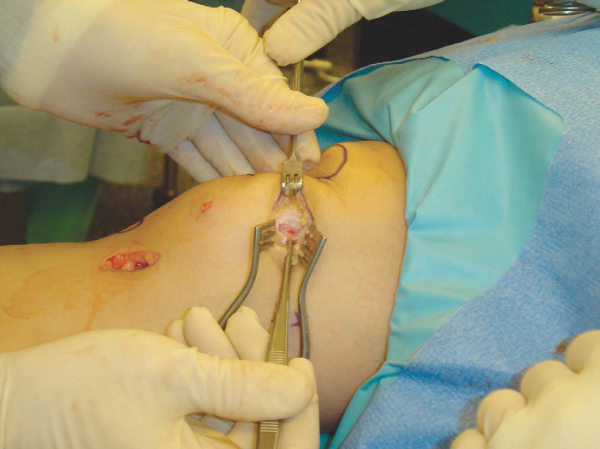
Fig. 72-8
), which is usually just above the equator of the patella. The surgeon must carefully advance the drill bit so that it exits the lateral edge of the patella without violating either the anterior bone cortex or posterior articular surface. The drill bit is then removed from the drill, and lateral fluoroscopy can confirm appropriate positioning (
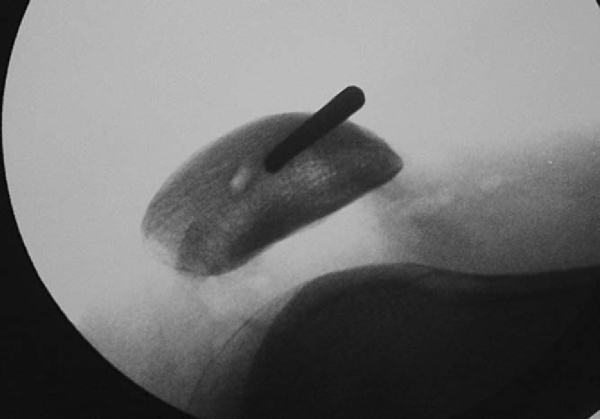
Fig. 72-9
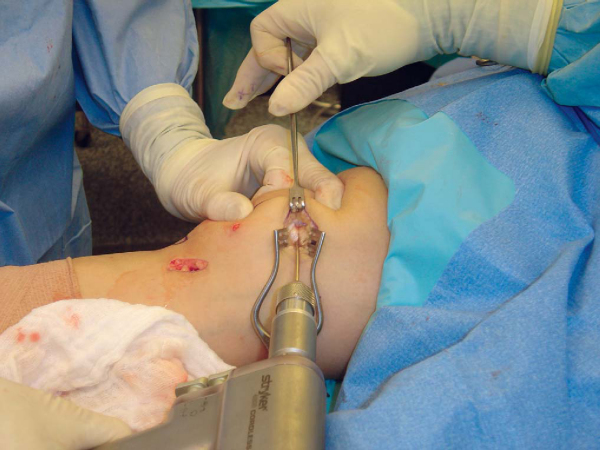
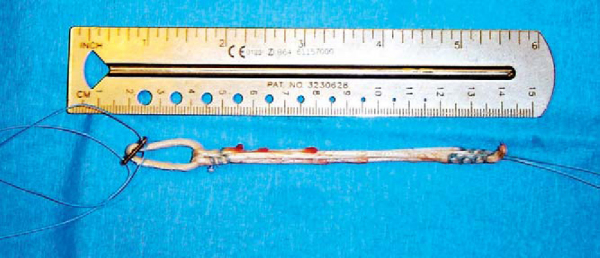
). The 2.5-mm drill bit is then replaced by a 2.0-mm eyelet K-wire, which is then overdrilled with a 4.5-mm cannulated drill bit. A depth gauge is then used to measure the length of the tunnel. On the basis of the length of the graft and the amount of graft that the surgeon wants in the patellar tunnel, the appropriate length Endobutton CL fixation device (Acufex; Smith & Nephew Endoscopy, Andover, Mass) is chosen. The graft construct is completed at the back table by passing the gracilis tendon tissue through the loop of the EndoButton and suturing it to itself with a No. 2 nonabsorbable woven suture. Sutures are also woven through the femoral end of the graft and will be used later to pull the graft into the femoral tunnel (
Fig. 72-10
). Two different-sized sutures are then passed through the two holes in the metal EndoButton. If the graft diameter is more than 4.5 mm, the patellar tunnel is enlarged with the appropriately sized cannulated drill bit to a depth of approximately 1 cm longer than the length of gracilis tissue that is intended to remain in the tunnel.
|
|
|
|
Figure 72-7 |
|
|
|
|
Figure 72-9 |
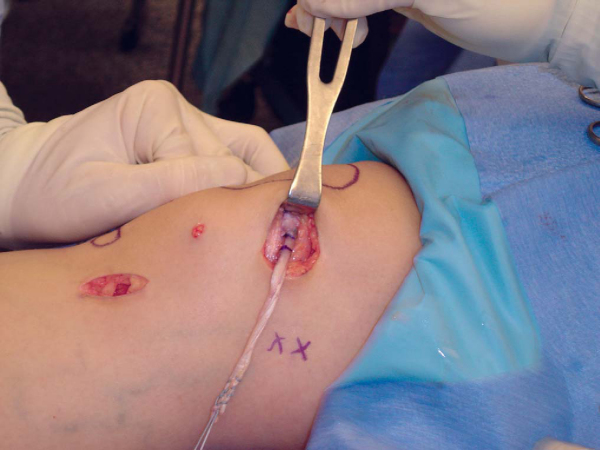
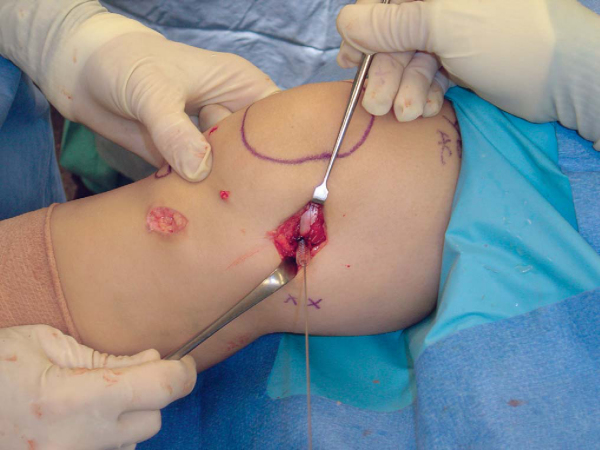
The 2.0-mm eyelet K-wire is passed from medial to lateral across the patellar tunnel and directed to exit the superolateral inflow portal. The four suture ends from the EndoButton are loaded through the eyelet of the K-wire, and the K-wire is pulled out through the superolateral portal site. With the EndoButton positioned lengthwise, the graft is pulled through the patellar tunnel until it clears the lateral edge of the patella (
Fig. 72-11
). Once it clears the patellar tunnel, tension is placed on the femoral end of the graft so that the EndoButton sits flush along the lateral edge of the patella. The position of the EndoButton can be confirmed fluoroscopically, and it can be manually manipulated so that it lies flush and lengthwise along the lateral edge of the patella.
|
|
|
|
Figure 72-11 |
A common technical error that occurs during MPFL reconstruction surgery is to place the femoral tunnel adjacent to the adductor tubercle rather than the medial femoral epicondyle; therefore, it is crucial to distinguish between these two bone prominences. Several anatomic studies have demonstrated that the femoral attachment site of the MPFL is at the medial epicondyle, which is approximately 1 cm distal to the adductor tubercle. [20] [21] [25] Others report the femoral attachment to be just anterior to the medial femoral epicondyle. [12] [26] A biomechanical study suggests that malpositioning of the femoral tunnel even 5 mm too far proximal can result in increased graft force and pressure applied to the cartilage of the medial facet of the patella.[11]
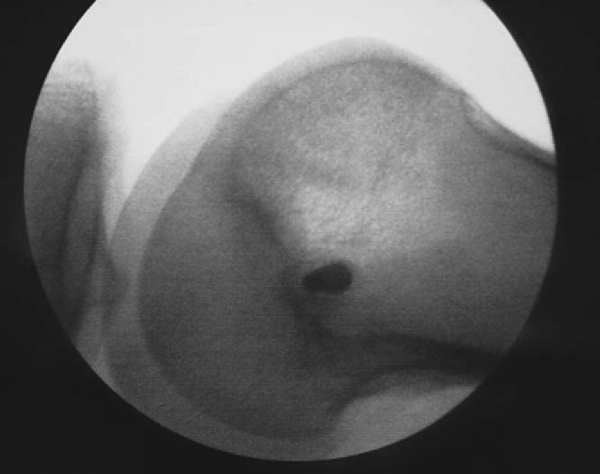
Once the anatomy is defined, a 2.5-mm drill bit is placed just anterior to the medial femoral epicondyle. This position can be confirmed fluoroscopically (
Fig. 72-12
). The graft is then passed through a soft tissue tunnel underneath the medial retinaculum and the remnant of the MPFL. The graft can then be wrapped around the drill bit, allowing assessment of graft isometry as the knee is flexed and extended. Minor changes in the position of the drill bit will allow fine-tuning of tunnel positioning. Once the optimal position for the femoral tunnel has been determined, the drill bit is replaced with a 2.0-mm eyelet K-wire. The K-wire is then overdrilled with a 6.0-mm cannulated drill bit to the appropriate depth based on the remaining length of the graft (
Fig. 72-13
). The suture ends are then passed through the eyelet of the K-wire, and the graft is pulled into the femoral tunnel as the K-wire is pulled out the lateral side of the knee (
Fig. 72-14
).
|
|
|
|
Figure 72-12 |
|
|
|
|
Figure 72-14 |
6. Graft Tensioning and Fixation
Tension is placed on the free suture ends on the lateral aspect of the knee as the surgeon moves the knee several times through a full range of motion. As the knee flexes and extends, the amount of tension in the graft can be ascertained by the amount of traction felt on the free suture ends (
Fig. 72-15
). In addition, the graft tension is directly palpated and visualized through the medial incision. It is extremely important not to overconstrain the graft. The knee is placed in full extension, and the patella is translated laterally. An attempt is made to identify the graft length that reproduces the same amount of normal lateral translation noted on the contralateral side. Once the appropriate graft length is identified, graft fixation is obtained by a 7.0-mm bioabsorbable cannulated femoral interference screw (
Fig. 72-16
). The screw is countersunk below the surface of the bone, and fixation can be augmented by sewing the graft to the adjacent soft tissue at the tunnel opening (
Fig. 72-17
). It should then be confirmed that the knee has full range of motion, the patella is no longer dislocatable, and the graft provides a firm checkrein preventing pathologic lateral translation.
|
|
|
|
Figure 72-15 |
|
|
|
|
Figure 72-16 |
|
|
|
|
Figure 72-17 |
Subcuticular wound closure offers excellent cosmesis (
Fig. 72-18
). Cryotherapy devices are routinely used to help control pain and swelling. A compressive dressing is then applied, followed by a thigh-high compression stocking and a postoperative brace locked in full extension.
Rehabilitation (
Box 72-2
)
Immediately postoperatively, patients are instructed in quadriceps sets and straight-leg raises. Patients are instructed in crutch ambulation and are allowed touch-down weight bearing. The brace remains locked in full extension for 1 week, at which point patients are encouraged to begin knee range of motion and to progress as tolerated. Weight bearing is also allowed to progress as tolerated. Patients are encouraged to attend formal physical therapy three times per week, where knee range of motion and quadriceps-strengthening exercises are emphasized. The brace is unlocked for ambulation as soon as quadriceps strength is sufficient. Patients are given the goal of achieving 120 degrees of knee flexion by 4 weeks postoperatively, and the brace is generally discontinued by 6 weeks. Patients are encouraged to achieve full knee range of motion by 8 weeks and allowed to progress to jogging and sports-specific drills by 12 weeks. Most patients are able to return to sports by 4 to 5 months.
As with many knee reconstruction procedures, the most common postoperative complication is loss of motion. Flexion and extension deficits may be secondary to inadequate postoperative rehabilitation. This risk can be minimized by early weight bearing and motion exercises. Flexion deficits may also be secondary to intraoperative technical errors (malpositioning or overtensioning of the graft). These errors could also overload the medial patellofemoral joint, resulting in pain and arthrosis, especially in the setting of a traumatic medial patellar chondral lesion. Other complications include recurrent instability, painful hardware, and patellar fracture.
| PEARLS AND PITFALLS | ||||||||||||||||||||||||||||||||||||||||||
|
Published studies reporting outcome after MPFL repair or reconstruction have been limited by small numbers of patients, retrospective design, and lack of control groups but have been generally favorable. [2] [6] [8] [9] [10] [15] [22] [23] Most authors report satisfactory outcomes (good and excellent) in the range of 83% to 96% (
Table 72-1
). [1] [8] [9] [10] [15] [22] Because a variety of different outcome parameters have been used, it is difficult to compare results, indicating the need for larger, multicenter controlled studies.
| Author | No. of Patients | Followup | Results |
|---|---|---|---|
| MPFL Reconstruction | |||
| Deie et al[6] (2003) | 4 | 7.4 years | Dislocation, 0; Kujala score: 96.3 |
| Drez et al[8] (2001) | 15 | 2.6 years | Dislocation, 0; Kujala score: 88 |
| Ellera Gomes[9] (1992) | 30 | 3.25 years | 83% good-excellent |
| Ellera Gomes et al[10] (2004) | 15 | 5.0 years | 87% satisfied |
| MPFL Repair | |||
| Avikainen et al[2] (1993) | 14 | 6.9 years | Dislocation, 1; 86% good-excellent |
| Garth et al[15] (2003) | 20 | 2.7 years | 95% good-excellent |
| Nomura et al[22] (2004) | 5 | 5.9 years | Kujala score: 97.6; 80% good-excellent |
| Sallay et al[23] (1996) | 12 | 3 years | Dislocation, 0; 58% good-excellent |
1.
Amis AA, Firer P, Mountney J, et al: Anatomy and biomechanics of the medial patellofemoral ligament.
Knee 2003; 10:215-220.
2.
Avikainen VJ, Nikku RK, Seppanen-Lehmonen TK: Adductor magnus tenodesis for patellar dislocation. Technique and preliminary results.
Clin Orthop Relat Res 1993; 297:12-16.
3.
Ciccone WJ, Bratton DR, Weinstein DM, Elias JJ: Viscoelasticity and temperature variations decrease tension and stiffness of hamstring tendon grafts following anterior cruciate ligament reconstruction.
J Bone Joint Surg Am 2006; 88:1071-1078.
4.
Conlan T, Garth Jr WP, Lemons JE: Evaluation of the medial soft-tissue restraints of the extensor mechanism of the knee.
J Bone Joint Surg Am 1993; 75:682-693.
5.
Davis DK, Fithian DC: Techniques of medial retinacular repair and reconstruction.
Clin Orthop Relat Res 2002; 402:38-52.
6.
Deie M, Ochi M, Sumen Y, et al: Reconstruction of the medial patellofemoral ligament for the treatment of habitual or recurrent dislocation of the patella in children.
J Bone Joint Surg Br 2003; 85:887-890.
7.
Desio SM, Burks RT, Bachus KN: Soft tissue restraints to lateral patellar translation in the human knee.
Am J Sports Med 1998; 26:59-65.
8.
Drez Jr D, Edwards TB, Williams CS: Results of medial patellofemoral ligament reconstruction in the treatment of patellar dislocation.
Arthroscopy 2001; 17:298-306.
9.
Ellera Gomes JL: Medial patellofemoral ligament reconstruction for recurrent dislocation of the patella: a preliminary report.
Arthroscopy 1992; 8:335-340.
10.
Ellera Gomes JL, Stigler Marczyk LR, Cesar de Cesar P, Jungblut CF: Medial patellofemoral ligament reconstruction with semitendinosus autograft for chronic patellar instability: a followup study.
Arthroscopy 2004; 20:147-151.
11.
Elias JJ, Cosgarea AJ: Technical errors during medial patellofemoral ligament reconstruction could overload medial patellofemoral cartilage: a computational analysis.
Am J Sports Med 2006; 34:1478-1485.
12.
Feller JA, Feagin Jr JA, Garrett Jr WE: The medial patellofemoral ligament revisited: an anatomical study.
Knee Surg Sports Traumatol Arthrosc 1993; 1:184-186.
13.
Fithian DC, Meier SW: The case for advancement and repair of the medial patellofemoral ligament in patients with recurrent patellar instability.
Oper Tech Sports Med 1999; 7:81-89.
14.
Fulkerson JP, Becker GJ, Meaney JA, et al: Anteromedial tibial tubercle transfer without bone graft.
Am J Sports Med 1990; 18:490-496.
15.
Garth Jr WP, DiChristina DG, Holt G: Delayed proximal repair and distal realignment after patellar dislocation.
Clin Orthop Relat Res 2000; 377:132-144.
16.
Hamner DL, Brown Jr CH, Steiner ME, et al: Hamstring tendon grafts for reconstruction of the anterior cruciate ligament: biomechanical evaluation of the use of multiple strands and tensioning techniques.
J Bone Joint Surg Am 1999; 81:549-557.
17.
Huberti HH, Hayes WC, Stone JL, Shybut GT: Force ratios in the quadriceps tendon and ligamentum patellae.
J Orthop Res 1984; 2:49-54.
18.
Muneta T, Sekiya I, Tsuchiya M, Shinomiya K: A technique for reconstruction of the medial patellofemoral ligament.
Clin Orthop Relat Res 1999; 359:151-155.
19.
Nomura E: Classification of lesions of the medial patellofemoral ligament in patellar dislocation.
Int Orthop 1999; 23:260-263.
20.
Nomura E, Horiuchi Y, Kihara M: Medial patellofemoral ligament restraint in lateral patellar translation and reconstruction.
Knee 2000; 7:121-127.
21.
Nomura E, Inoue M: Surgical technique and rationale for medial patellofemoral ligament reconstruction for recurrent patellar dislocation.
Arthroscopy 2003; 19:E47.
22.
Nomura E, Inoue M, Osada N: Augmented repair of avulsion-tear type medial patellofemoral ligament injury in acute patellar dislocation.
Knee Surg Sports Traumatol Arthrosc 2005; 13:346-351.Epub Nov 27, 2004.
23.
Sallay PI, Poggi J, Speer KP, Garrett WE: Acute dislocation of the patella. A correlative pathoanatomic study.
Am J Sports Med 1996; 24:52-60.
24.
Schock E, Burks R: Medial patellofemoral ligament reconstruction using a hamstring graft.
Oper Tech Sports Med 2001; 9:169-175.
25.
Smirk C, Morris H: The anatomy and reconstruction of the medial patellofemoral ligament.
Knee 2003; 10:221-227.
26.
Steensen RN, Dopirak RM, McDonald 3rd WG: The anatomy and isometry of the medial patellofemoral ligament: implications for reconstruction.
Am J Sports Med 2004; 32:1509-1513.