Elbow Arthroplasty: Revision
IV – Elbow Reconstruction > Part C – Operative Treatment Methods
> 61 – Elbow Arthroplasty: Revision
elbow arthroplasties (TEAs) was published in 1996. With an average
follow-up of 5 years, the complication rate was 43% with a revision
rate of 18%. Main complications were the following: aseptic loosening
(radiographic 17.2% and clinical 6.4%), infection (8.1%), ulnar nerve
involvement (10.4%), instability (7% to 19%), and periprosthetic
fracture (3.2%). The French Orthopedic and Traumatology Society
reviewing 370 TEAs found a complication rate of 27% with a revision
rate of 17%. In this chapter, several specific features of the
presentation are discussed and surgical options available to deal with
failed TEAs are presented.
device failure, instability, periprosthetic fracture, and loosening.
Assessment to exclude the possibility of sepsis is the most important
consideration prior to any revision procedure, but especially in those
with early unexplained or unanticipated failure. Analysis of the
sedimentation rate and C-reactive protein are regularly performed along
with aspiration of the joint if there is any question of sepsis. Based
on the plain radiograph to analyze bone quality, the appropriate
preoperative plan is formulated. Surgery is not performed if
radiolucent lines are not painful. In such cases, the patient is
followed on a regular basis. Stiffness, scarring, and contracture of
the soft tissue and prior evidence of sepsis and status of the ulnar
nerve must also be noted.
mode of material failure. Although often contributing to component
loosening owing to polyethylene debris, mechanical symptoms may develop
because of metal-on-metal articulation or dislocation of the component.
Fracture of the metal components has been reported but is now less
common owing to improvements in implant design and materials. Isolated
bushing exchange is a successful procedure if there is no osteolysis
compromising component fixation.
types of TEA. Although usually occurring in the early postoperative
period as a frank dislocation, this also can present more insidiously
as weakness, giving way, clunking, or other mechanical symptoms.
Instability can be caused by ligament insufficiency, malpositioning of
the components, or uneven wear of the polyethylene. Examination under
fluoroscopy may show the cause of instability and allow the diagnosis
to be made. Splinting for a few weeks may help restore stability, but
if the elbow remains unstable after a period of splinting, a surgical
procedure may be indicated. Attempts to salvage an unlinked TEA that is
unstable can be unpredictable. One can attempt at least one soft tissue
procedure before undertaking removal of a well-cemented unlinked
prosthesis. Attention should be paid to maintaining or restoring an
adequate lateral collateral ligament complex by firmly reattaching it
to the lateral epicondyle with use of sutures through drill holes in
bone or by reconstructing the ligament with a tendon graft. Elbows with
malpositioned components do not respond to conservative treatment or
ligament reconstruction. Revision of the component positioning or
conversion to a semiconstrained implant is usually required.
implant insertion, as a consequence of neglected component loosening
with bone loss, or as a result of a traumatic
event.
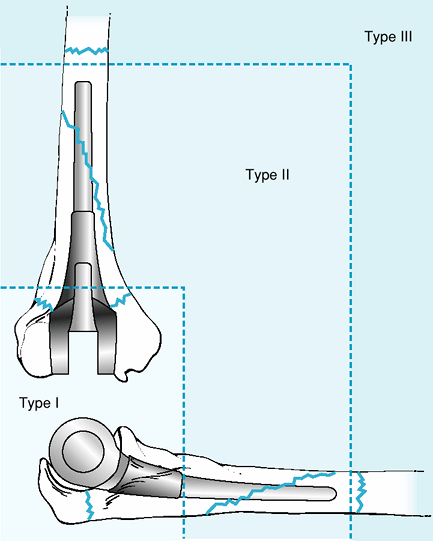
Mayo has proposed to classify these fractures as type I—metaphyseal;
type II—stem involvement; type III—proximal or distal to the stem tip (Fig. 61-1).
 |
|
Figure 61-1 Mayo Classification of periprosthetic elbow fractures. Type I: metaphyseal; type II: involves stem; type III: beyond stem.
|
periarticular segment (type I) usually do not require surgical
treatment if a linked prosthesis has been used. In unlinked prostheses,
fixation of the fragment is necessary to preserve implant stability.
Fractures that involve the olecranon, however, should be fixed in all
patients since this will restore triceps function. Proximal or distal
to the stem (type III), cerclage wire or plate and screw fixation may
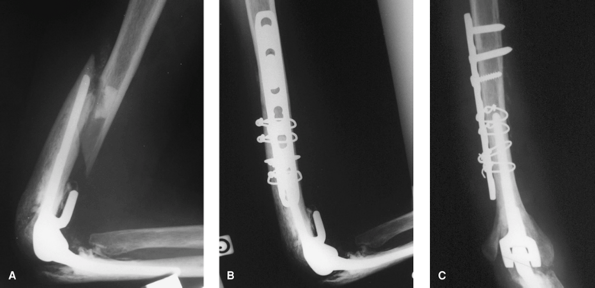
be required to stabilize the fracture (Fig. 61-2).
Fractures around the stem of the prosthesis (type II) almost always
require revision surgery because the implant is often loose. A longer
revision stem should be used to bypass the fracture, with cortical
strut allograft around the fracture.
 |
|
Figure 61-2 Periprosthetic type III fracture at the stem of the humeral component; the implant is well fixed (A). Osteosynthesis with plate and screw and cerclage wire (B, C).
|
long-term implant failure. A loose implant can be associated with bone
resorption, cortical thinning, and ballooning of the humerus or ulna.
Revision options are predicated on the quality of bone and presence of
a periprosthetic fracture. A salvage procedure such as arthrodesis is
rarely indicated. Arthrodesis requires a sufficient amount of bone
present. Resection arthroplasty is indicated in the presence of a
septic prosthesis. A stable resection arthroplasty can provide a
relatively comfortable joint. Not all patients after resection
arthroplasty will desire a reimplantation procedure. A revision
procedure with reimplantation can be performed when infection has been
eradicated.
fixation can be obtained with another stemmed implant given the amount
and quality of the intact cement mantle. If bone
stock
remains, a nonconstrained implant can be used. However, most of the
time a semiconstrained implant is preferred. Long-stem devices should
be available. For humeral revisions, a 15- or 20-cm-long humeral stem
is often needed. Long-stemmed ulnar components should also be available.
sterile tourniquet is often used. The lateral decubitus position is
preferred by some surgeons to allow wide exposure of the humerus and
radial nerve if necessary. The use of a posterior midline elbow
incision is preferred, but use of a previous skin incision is
recommended. The technical features of all revision options must
address the preservation of the triceps, identifying and protecting all
neurovascular structures, and protection of the cortical bone. If the
distal humeral columns are deficient, a triceps-preserving approach
should be considered. If the olecranon process is fractured, a
trans-olecranon approach is used for revision of the elbow arthroplasty
and repaired with tension band wire technique at the end of the
procedure. Proximally, the radial nerve is always identified, at least
by palpation. The ulna is extensively exposed in a subcutaneous fashion
as distally as necessary to have adequate exposure and to avoid
violation of the ulnar cortex. Extensive synovectomy is always
necessary; tissue samples are sent for pathology and culture.
of aseptic loosening but more problematic if they are firmly fixed. If
the implant design is tapered, the component sometimes can be extracted
by grasping the articulating surface and tapping on the prosthesis in a
retrograde direction. Removing as much cement as possible allows the
device to be removed more easily. Fracture or further bone loss from
aggressive attempts at cement removal should be avoided. A cortical
window around the stem or at the tip of the prosthesis should be
considered to avoid intraoperative fracture caused by more aggressive
attempts to remove the components. It should be fixed with cerclage
wire at the end of the procedure before cement injection. Once the
prosthesis is removed, cement that is firmly adherent should be left in
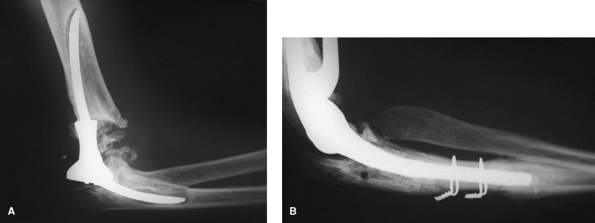
place unless it interferes with placement of the new components (Fig. 61-3). Powered cement removal instruments should be used with caution. Ultrasonic cement removal devices can be particularly useful.
 |
|
Figure 61-3 Revision of a loose constrained implant (A). A long-stem semiconstrained implant has been used to bypass the area of loosening (B).
|
adequate length to bypass cortical defects or fractures and have
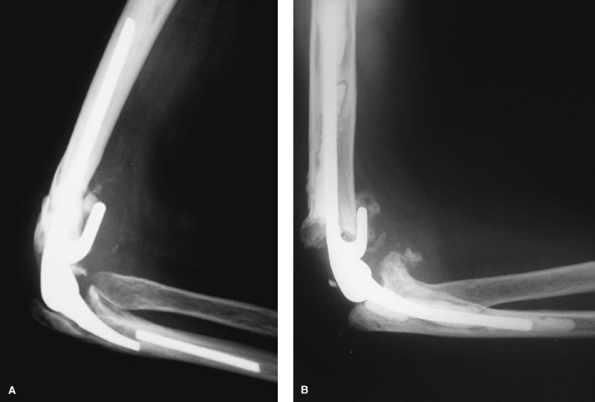
sufficient constraint to provide adequate joint stability (Fig. 61-4).
Modern cement technique is used with cement restrictors and pressurized
gun. Antibiotic cement should be routinely used. Caution should be
exercised in patients with cortical perforations or fractures to ensure
that cement does not damage adjacent neurovascular structures.
and the arm placed in extension with a splint to avoid tension or
pressure on the wound. The arm is elevated for 48 hours. Gentle active
motion can then be initiated. If the bone is fractured or if a bone
graft has been used, protection in a cast brace or splint for several
weeks may be undertaken. No formal therapy is prescribed, but
activities of daily living are encouraged.
osteolysis, or a periprosthetic fracture, an impaction or strut
grafting augmentation procedure is indicated.
purposes, to restore bone stock and to enhance the bone/cement
interface. At least 2 to 3 cm stem depth into intact bone in addition
to the augmentation fixators is needed. The
medullary
canal is plugged with a silastic device. A double-tube apparatus is
assembled. The nozzle of tubing used for femoral cementation is cut to
the length that corresponds to the extent of the lytic process. The
elbow cement injector tube is then inserted within the femoral tube,
extending distally into normal host bone. Cancellous bone graft or
graft substitute is tightly packed around the outer tube. The cement is
then mixed in the canister of the smaller elbow injector system and
inserted on the nozzle in situ. It is injected through the nozzle while
withdrawing to the level of the outer tube. At this point both inner
and outer tubes are simultaneously withdrawn while injecting cement
into the void created by the larger tube. The implant is carefully
inserted to the desired length (Fig. 61-5). This technique may be used for both the humerus and ulna, as an adjunct in the re-establishment of bone stock (Fig. 61-6).
 |
|
Figure 61-4 Ulna fenestration has been made to remove a fractured well-fixed ulnar component (A). Cerclage wire has been used to fix the cortical window before cementing a new ulnar component (B).
|
 |
|
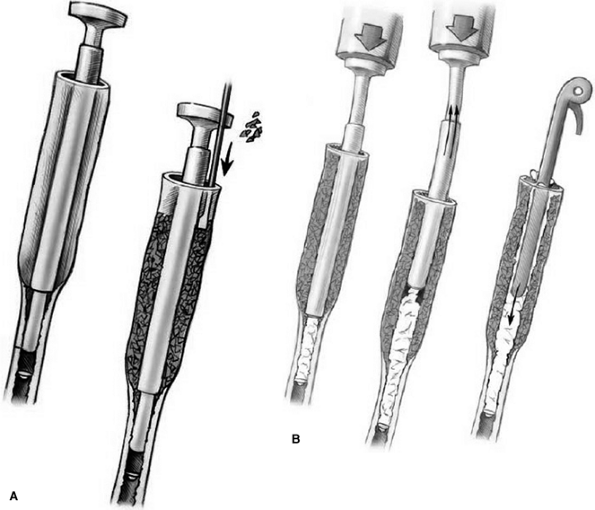
Figure 61-5 A double tube is used and allograft bone is packed around the outer tube (A);
The cement is injected through the smaller tube, which is slowly withdrawn to the level of the outer tube; then both tubes are withdrawn simultaneously while injecting cement into the void created by the larger tube (B). |
 |
|
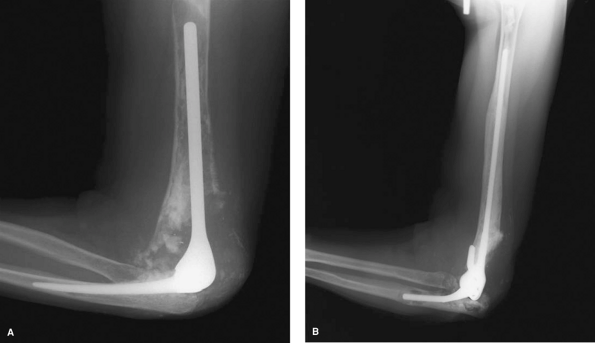
Figure 61-6 Marked osteolysis (A) effectively treated with impaction grafting at 6 years (B).
|
 |
|
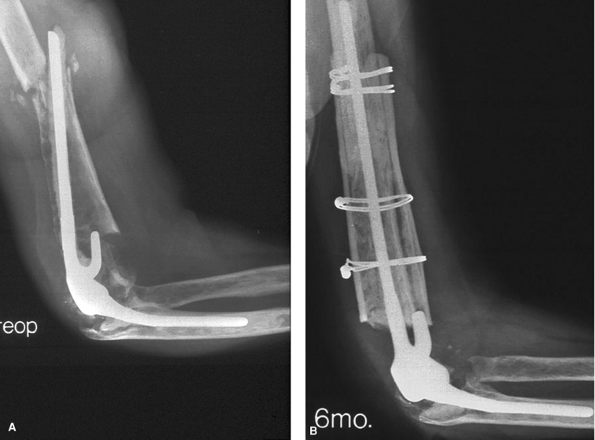
Figure 61-7 Patient with severe bone loss and fracture after failed Coonrad/Morrey implant (A). At 2 years, the struts have incorporated with a successful clinical and radiographic outcome (B).
|
periprosthetic fractures and for distal humeral or proximal ulnar bone
loss. The most effective application to the humerus is that of an
anterior strut that transverses the osteolysis or fracture and captures
the flange anteriorly. A posterior strut is used to enhance stability
and to prevent the wire cutting through the host bone. The use of an
extended flange, anterior strut graft, and 2 cm of shortening allows
management of distal humeral deficiencies of ≤7 cm (Fig. 61-7).
a component reconstruction with strut graft, or an allograft-prosthesis
composite (APC) are options. In this last option, the prosthesis is
fitted and cemented to the allograft. When that has hardened, the
prosthesis is cemented in the host. The difficulty of obtaining
interface union between the host and allograft bones has limited the
use of this strategy.
An evolution in prosthetic components and varied surgical options has
allowed the surgeon to deal with different presentations of TEA
failure. Although the complication rate is higher than in primary TEA,
the frequency of problems continues to decrease as experience with
revision surgery increases.
J, O’Driscoll SW, Morrey BF. Periprosthetic humeral fracture after
total elbow arthroplasty treatment with implant revision and strut
allograft augmentation. J Bone Joint Surg Am. 2002;8:1642–1650.
