Myogenic, Lipogenic, and Epithelial Bone Sarcomas
Editors: Tornetta, Paul; Einhorn, Thomas A.; Damron, Timothy A.
Title: Oncology and Basic Science, 7th Edition
Copyright ©2008 Lippincott Williams & Wilkins
> Table of Contents > Section
II – Specific Bone Neoplasms and Simulators > 6 – Bone Sarcomas >
6.7 – Myogenic, Lipogenic, and Epithelial Bone Sarcomas
II – Specific Bone Neoplasms and Simulators > 6 – Bone Sarcomas >
6.7 – Myogenic, Lipogenic, and Epithelial Bone Sarcomas
6.7
Myogenic, Lipogenic, and Epithelial Bone Sarcomas
Hannah D. Morgan
Leiomyosarcoma and liposarcoma of bone and adamantinoma
are all rare osseous malignancies, each constituting less than 1% all
bone sarcomas. Patients with leiomyosarcoma and liposarcoma of bone
should be carefully examined to ensure that the bony lesion is a
primary tumor and not a metastasis from a soft tissue malignancy.
Adamantinoma should be always be considered in the differential
diagnosis of a tibial aggressive lesion.
are all rare osseous malignancies, each constituting less than 1% all
bone sarcomas. Patients with leiomyosarcoma and liposarcoma of bone
should be carefully examined to ensure that the bony lesion is a
primary tumor and not a metastasis from a soft tissue malignancy.
Adamantinoma should be always be considered in the differential
diagnosis of a tibial aggressive lesion.
Pathogenesis
Etiology
-
Leiomyosarcoma: arises from smooth muscle cells of intraosseous blood vessels
-
Liposarcoma: etiology unknown; immature adipose tissue
-
Adamantinoma: presumed ectopic epithelial cell residues
Epidemiology
-
Leiomyosarcoma
-
Extremely rare; <0.1% of all bone sarcomas
-
Male = female
-
First through ninth decades; mean age 45 to 50 years
-
Femur/tibia > humerus > ilium in extragnathic sites
-
-
Liposarcoma
-
One of the rarest primary bone tumors
-
Second through sixth decades; typically third and fourth decades
-
Major long tubular bones affected
-
-
Adamantinoma
-
~0.4% of bone sarcomas
-
Male:female 3:2
-
Young adults and children (mean age 30 years); typically older than osteofibrous dysplasia
-
85% involve tibia (especially anterior aspect); 10% tibia and fibula
-
Pathophysiology
-
Leiomyosarcoma
-
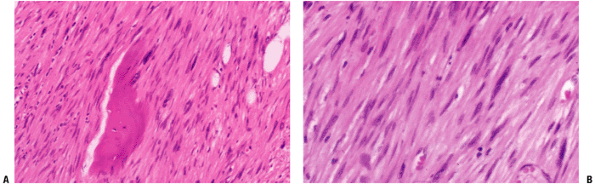
Spindle-shaped cells with eosinophilic cytoplasm and cigar-shaped nuclei arranged in bundles intersecting at right angles (Fig. 6.7-1)
-
Immunohistochemistry: positive vimentin (as for all sarcomas), positive smooth muscle actin
-
-
Liposarcoma
-
Either pleomorphic or round cell–rich high-grade lesion
-
Signet-ring lipoblasts may be seen.
-
-
Adamantinoma
-
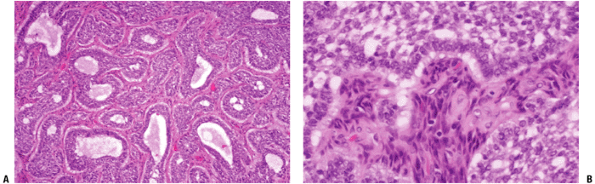
Epithelial cells surrounded by spindle-shaped fibrous tissue; little pleomorphism (Fig. 6.7-2)
-
P.210
 |
|
Figure 6.7-1 Leiomyosarcoma of bone. Note pleomorphic elongated cells with cigar-shaped nuclei.
|
Classification
-
Leiomyosarcoma: 80% of tumors are high-grade malignancies of smooth muscle origin
-
Liposarcoma: high-grade lesions of immature adipose tissue
-
Adamantinoma: low-grade malignant epithelial lesions with strong predilection for the tibia
Diagnosis
Physical Examination and History
Clinical Features
-
Leiomyosarcoma
-
Pain, palpable mass; pain usually present >6 months
-
20% of patients present with pathologic fracture.
-
-
Liposarcoma
-
Patient typically presents with pain.
-
-
Adamantinoma
-
Pain, swelling, with or without pathologic fracture, with or without bowing
-
Symptoms are often present for months to years.
-
 |
|
Figure 6.7-2 Adamantinoma of bone. Note biphasic pattern of epithelioid component within fibrous background.
|
Radiologic Features
-
Leiomyosarcoma
-
Radiographs: aggressive osteolytic lesion with indistinct margins
-
Computed tomographic (CT) scan: bony destruction and extraosseous extension
-
Abdomen/pelvis scan to look for primary uterine/gastrointestinal tumor
-
-
Magnetic resonance imaging (MRI): typically low intensity on T1, high intensity on T2 images
-
Soft tissue mass often much larger than expected on x-ray
-
-
-
Liposarcoma
-
Radiographs show bone-destructive process.
-
MRI shows lesion bright on T2-weighted images.
-
-
Adamantinoma
-
CT scan: shows cortical involvement
-
MRI: high signal on T2-weighted images, possible soft tissue extension, may see multifocal disease and bone marrow extension
Treatment
Surgical and Nonoperative Options
All three of these tumors are usually amenable to limb
salvage and are treated most successfully with wide resection of the
involved bone followed by a reconstructive procedure. Leiomyosarcoma
and liposarcoma of bone, both of which tend to be high-grade,
aggressive tumors, may respond to adjuvant and/or neoadjuvant
chemotherapy and radiotherapy.
salvage and are treated most successfully with wide resection of the
involved bone followed by a reconstructive procedure. Leiomyosarcoma
and liposarcoma of bone, both of which tend to be high-grade,
aggressive tumors, may respond to adjuvant and/or neoadjuvant
chemotherapy and radiotherapy.
Preoperative Planning
-
Essentially the same as for all bone sarcomas (also see Chapter 4, Treatment Principles)
-
CT scans of chest/abdomen/pelvis
-
Determine whether bony lesion is truly primary tumor.
-
Rule out pulmonary or other metastatic disease.
-
Surgical Goals and Approaches (also see Chapter 4)
-
Remove all tumor with margin of surrounding normal tissue.
Techniques
-
Same as for all bone sarcomas (see Chapter 4)
Complications
-
Same as for all bone sarcomas (see Chapter 4)
Results and Outcome (Prognosis)
-
Leiomyosarcoma
-
Estimated 5-year survival 68% (100% if low grade)
-
Local recurrence 25%, lung metastases 25%
-
-
Liposarcoma
-
Difficult to determine due to small number of patients
-
Approximately 50% will develop metastases.
-
-
Adamantinoma
-
10-year survival approximately 85%
-
Time to local recurrence 5 to 15 years
-
Time to metastases up to 27 years
-
Patients need long-term follow-up.
-
-
Postoperative Management
-
Same as for all bone sarcomas (see Chapter 4).
Suggested Reading
Antonescu
CR, Erlandson RA, Huvos AG. Primary leiomyosarcoma of bone: a
clinicopathologic, immunohistochemical, and ultrastructural study of 33
patients and a literature review. Am J Surg Pathol 1997;21(11):1281–1294.
CR, Erlandson RA, Huvos AG. Primary leiomyosarcoma of bone: a
clinicopathologic, immunohistochemical, and ultrastructural study of 33
patients and a literature review. Am J Surg Pathol 1997;21(11):1281–1294.
Bouaziz MC, Chaabane S, Mrad K, et al. Primary leiomyosarcoma of bone: report of 4 cases. J Comput Assist Tomog 2005;29(2):254–259.
Goto T, Ishida T, Motoi N, et al. Primary leiomyosarcoma of the femur. J Orthop Sci 2002;7:267–273.
Kahn LB. Adamantinoma, osteofibrous dysplasia and differentiated adamantinoma. Skeletal Radiol 2003;32:245–258.
Qureshi AA, Shott S, Mallin BA, et al. Current trends in the management of adamantinoma of long bones: an international study. J Bone Joint Surg [Am] 2000;82(8):1122–1131.
