Staying Out of Trouble with Tumors
|
|
patient an enormous amount of trouble. Although detailing the
comprehensive management of musculoskeletal tumors is far beyond the
goals of this text, the principles of identifying and evaluating such
patients are fundamental to staying out of trouble in pediatric
orthopaedics. Regardless of the scope of your practice or special
interest, as an orthopaedist navigating through the care of thousands
of patients per year, you are bound to encounter someone with a tumor.
Often the presenting complaint is rather pedestrian: knee pain or a
backache, just like hundreds of such patients that will see you this
month (Fig. 17-1). Although few of us have the
training, experience, interest, or oncologic support team and services
to offer definitive treatment, we are the “net” that must capture these
patients immediately and get them to the right person, in the right
place, quickly and safely. The focus of this chapter is avoiding the
three major sources of trouble in managing tumors:
 |
|
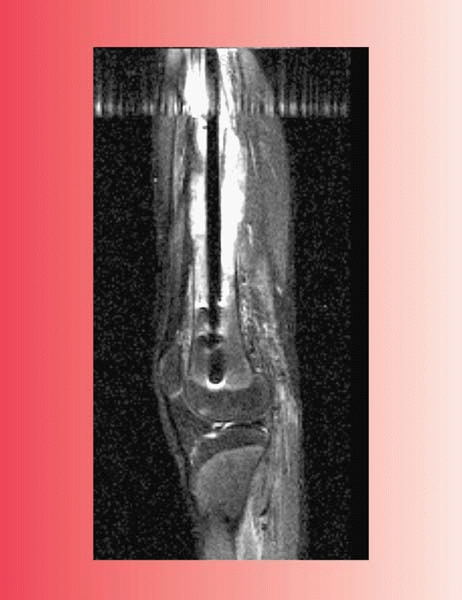
▪ FIGURE 17-1
Always get radiographs when children present with knee pain. Fortunately, this boy’s orthopaedist got radiographs when he presented with a month-long history of anterior knee pain. There is an aggressive, destructive lesion in the distal femoral metaphysis. A biopsy identified this as an osteosarcoma. |
-
Missing a malignancy
-
Overtreating a “no-touch” lesion or other imitator
-
Treatment problems and complications during the management of benign bone tumors and soft tissue lesions
the doctor are higher today than ever in history. A generation or two
ago, most children with osteosarcoma or Ewing sarcoma died despite
surgical treatment. If you were the doctor who missed one of these
diagnoses or botched the biopsy, odds were that it didn’t change the
mortality rate in most cases. Now, with the advent of effective
chemotherapy, the survival rates have improved dramatically, and thus
the consequences of missed diagnosis or delayed diagnosis are much
greater. The differential diagnoses for children who present with a
malignancy include infection and trauma.1
Pain from cancer and infections are often written off as resulting from
injury. Unfortunately, that causes trouble, because the life of a child
is filled with falls and bumps and bruises. Beware of attributing all pain to injury.
getting a good history from the patient and family. Pain is the typical
presenting complaint in a child with a musculoskeletal malignancy. It
is important to get the time course, location, severity and quality of
the pain. Night pain, for instance, has a notorious association with
tumors, infections, and other serious conditions. This should be
distinguished from pain that is related to activities, suggesting
musculoskeletal overuse. Most patients with malignancies have rapid
symptom development. Don’t be lulled into a false sense of security if
the pain has “only been there for a couple of weeks.”
to look sick. Like their peers, they are usually healthy and active at
presentation. Two exceptions to this are children with Ewing sarcoma or
leukemia. Children with Ewing sarcoma can have signs and symptoms that
look like an infection, as well as a fever and elevated C-reactive
protein (CRP) and erythrocyte sedimentation rate (ESR). If the history
and symptom complex suggest the possibility of a malignancy, the
physical examination should be more comprehensive than is typically
done by an orthopaedic surgeon focused on the back or limbs.
trouble with tumors. A child who presents with a new complaint of
extremity pain needs good quality plain radiographs of the painful
region on the first visit to your office. The orthopaedist should
resist the urge to dismiss periosteal reaction or other potentially
concerning radiographic findings as trauma if the trauma did not cause
a definitive fracture. Children who present with such radiographic
findings should be followed closely and should be considered for other
imaging studies. There are three pearls for staying out of trouble as
you evaluate radiographs for children in pain:
-
Look at all fracture radiographs closely to be certain that the fracture is not a pathologic
fracture. Perhaps the easiest place to miss a pathologic fracture is
with a displaced femur fracture, in which the overlapping bone and lack
of orthogonal views may allow a lesion at the fracture site to remain
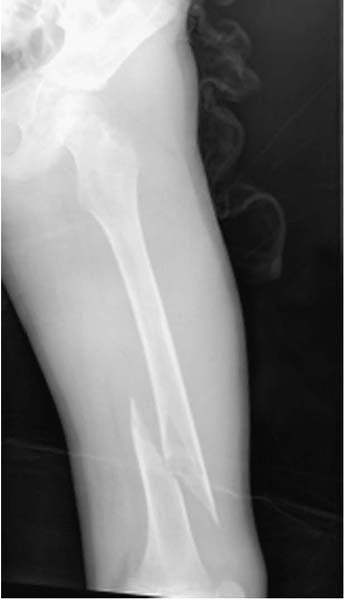
obscure (Fig. 17-2). -
Do what the radiologists teach their
residents: Look first at the areas away from the lesion or fracture.
Study the full extent of every radiograph of a kid who presents to you
with pain. -
Certain areas are notoriously hard to see with standard radiographs. These four areas—the spine, sacrum, scapula (Fig. 17-3) and pelvis (Fig. 17-4)—can allow lesions to hide undetected for a long period of time.2
If you are suspicious of lesions in this area, do not hesitate to move
quickly to more sophisticated imaging (usually MRI) to allow better
visualization.
 |
|
▪ FIGURE 17-2
A femoral shaft fracture can often hide a pathologic lesion. This boy’s spiral distal third diaphyseal fracture was through a nonossifying fibroma. The lesion could not be well visualized until the fracture was reduced and studied in the operating room under fluoroscopy. |
-
Where is the lesion?
-
What is the lesion doing to bone?
-
How is the bone reacting to the lesion—e.g., is there a periosteal reaction?
-
What kind of tissue is being produced
that might suggest the histology of the lesion—e.g., is it calcified or
is it radiolucent, like cartilage cells or bone marrow elements?
after an abnormal plain radiograph is an MRI. It is important to work
with your radiologist, communicating specifically what you are looking
for and that a malignancy is in the differential. This is a
particularly important “staying out of trouble” point in the current
era of offsite imaging centers. If your patient must use one of these
centers for insurance purposes, write a very detailed prescription—or
better yet,
personally
phone the radiologist to say that you are concerned about a tumor. It
is important that the entire region is imaged (e.g., the entire femur)
and that the study technique is appropriate the first time. This
includes the use of contrast, when appropriate.
 |
|
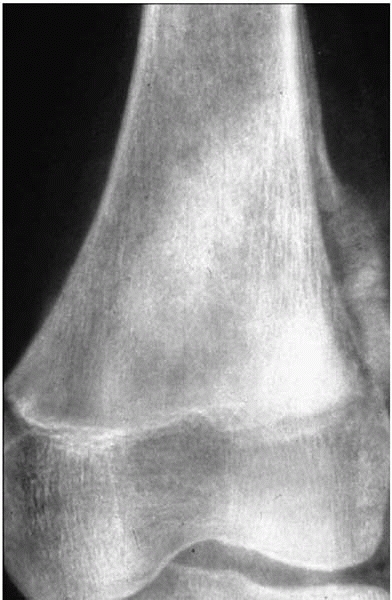
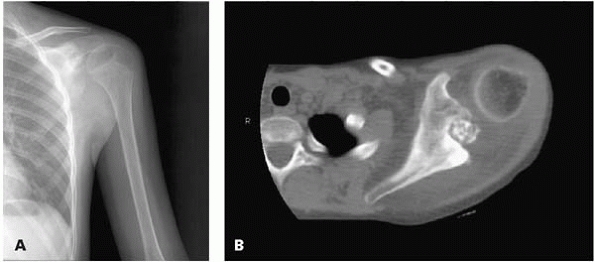
▪ FIGURE 17-3 This boy presented with shoulder pain. (A)
This AP radiograph of the shoulder was read as normal by the radiologist, who did not have the benefit of the history and physical exam. Fortunately, the orthopaedist reviewed the radiograph closely and recommended a CT scan. (B) This CT cut shows a large lesion in the scapula, which was later identified as an osteoblastoma. |
 |
|
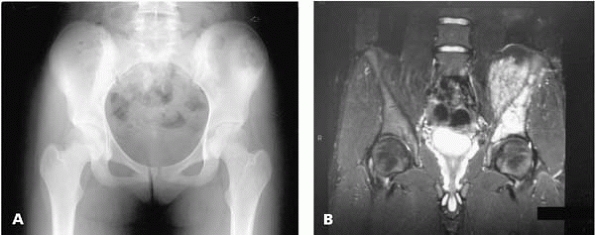
▪ FIGURE 17-4 The pelvis is a notorious area for hiding tumors. (A) This AP radiograph shows a slight abnormality in the left iliac wing. (B) An MRI visualizes the extent of this malignancy.
|
should never be done at the start of a workup. After a thorough
history, physical exam and imaging, a biopsy should be used to confirm what is suspected from a small list of possibilities.
If at all possible, the biopsy should be done at the center where the
child will be treated, by the surgeon who is going to do the definite
cancer operation. Exceptions to this rule should be rare.
-
Use a tourniquet, but don’t exsanguinate with an Esmarch (gravity only).
-
Make longitudinal incisions in the extremities.
-
The biopsy should go directly through muscle, not in the planes between muscle groups.
-
The biopsy should stay out of joints and away from neurovascular bundles.
-
A large representative sample should be obtained.
-
Every biopsy should be cultured (every culture should be biopsied).
-
A frozen section should be carried by the
surgeon to the pathologist to assure that lesional tissue has been
obtained and to make a preliminary diagnosis if possible. -
Careful hemostasis is essential to avoid a large hematoma contaminated with malignant cells.
-
The surgeon should rarely leave the
operating room without a diagnosis, or at least a highly likely
differential diagnosis that is confirmed by the pathologist. -
If a drain is used, it should be inline with and close to the longitudinal incision.
radiographic features can lead to unnecessary biopsy, or worse:
amputations have been performed for some of these benign lesions when
the biopsy had malignant histologic features. The keys to staying out
of trouble with these “no touch lesions” are experience and pattern
recognition. If there is uncertainty, go over the radiographs with an
experienced bone radiologist who has an atlas of normal variants
available.
is well developed, there is circumferential calcification with a
radiolucent center. Biopsy of myositis ossificans can be dangerous,
because the pathology can look so aggressive that the lesion might be
mistaken for a malignancy. One key to staying out of trouble is to get
a CT or MRI if there is uncertainty.
 |
|
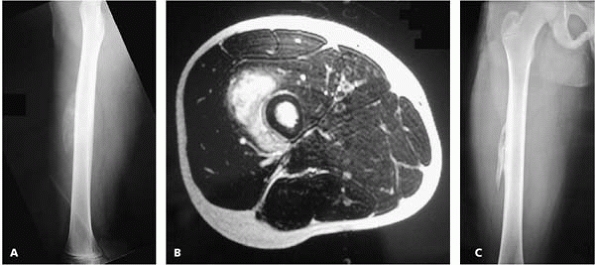
▪ FIGURE 17-5 Myositis ossificans can have a confusing or even scary appearance. (A)
This lateral radiograph of the femur of a boy presenting after trauma to the anterior thigh shows a calcified mass anterior to the midshaft of the femur. (B) This MR image shows that the calcified mass is in the vastus intermedius muscle and does not emanate from the femoral bone itself. This reassuring image clarified that this was a muscle process and not bone malignancy. Based on this evaluation, the surgeon avoided biopsy and just recommended radiographic followup over the next several months. (C) This AP femur radiograph, taken three months after initial presentation, shows myositis ossificans that has matured and no longer has a concerning appearance. |
but their anatomic location can be very helpful. An example is the
characteristic location of a rectus femorus avulsion at the anterior
inferior iliac spine. In pelvic avulsion fractures, the age of the
patient can also be helpful, because these fractures occur in a very
narrow age range early in adolescence. The history of injury also helps
a great deal. If there is any question about the identity of an
avulsion fracture versus a malignancy, it is valuable to repeat
radiographs a couple of weeks later.
 |
|
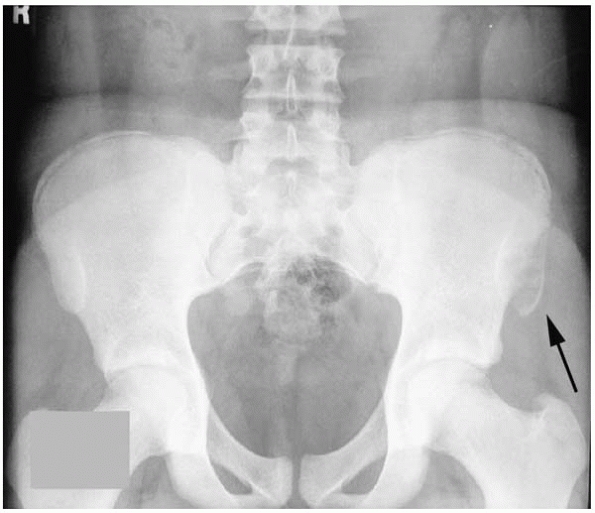
▪ FIGURE 17-6
Pelvic avulsion fractures can have a confusing appearance on initial presentation. This AP pelvis radiograph shows an anterior superior iliac spine avulsion fracture (arrow) that could easily be mistaken for an enlarging mass off the anterior iliac crest. |
appearance and should be identified primarily using pattern recognition
of an experienced orthopaedist or radiologist.
radiograph and MRI. Features of a detailed pain history (no night pain,
and pain only with activity and a probable history of overuse) can be
very reassuring. Concerning cases should be reviewed with the
radiologist, and repeat interval imaging a couple of weeks later may be
valuable.
lytic/sclerotic pattern that is worrisome for malignancy. Fortunately,
in children, most of these occur in patients with sickle cell disease
and can be suspected from the medical history. Sometimes, doubt lingers
for days or weeks, and concern grows among multiple specialists; the
only way to be certain it is a bone infarct is to biopsy the lesion (Fig. 17-7).
 |
|
▪ FIGURE 17-7
This 3-year-old boy presented with a mixed lytic and sclerotic lesion in his proximal humerus. He had a history of sickle cell disease. For 3 weeks, the team caring for him could not determine if it was infarct, infection or tumor—or a combination. (A) This AP humerus was taken 1 week after biopsy was done to silence growing concern from the hematologists that this could be leukemia or some other malignant process. The biopsy revealed no sign of tumor or infection, just dead bone consistent with an infarct associated with his sickle cell disease. (B) Six months after biopsy. Amazing healing. It is great to be a kid. |
after a good history, physical exam, and plain radiographs, just get
the patient to the right place and right person as quickly as possible.
The final diagnosis, staging, and management are beyond the scope of
this text.
teenager with knee pain (or, less commonly, shoulder pain), and therein
lies a major cause of the diagnostic trouble: knee pain is ubiquitous
in teenagers, and may not even make it past a confident pediatrician.
For this reason, it is important not to let your guard down. Get
radiographs of the involved area whenever a patient presents with pain.
Typically, the osteosarcoma is metaphyseal, with a mixed lytic and
sclerotic pattern, and a soft-tissue mass. The famous Codman’s triangle
(Fig. 17-8) suggests a very aggressive lesion
such as osteosarcoma or Ewing sarcoma. Should you encounter a
pathologic fracture through a lesion suspicious for osteosarcoma,
closed management of the fracture is recommended. A splint or a cast
should be used and the child should be transferred to an expert
orthopaedic oncologist as soon as possible. For goodness sakes, don’t
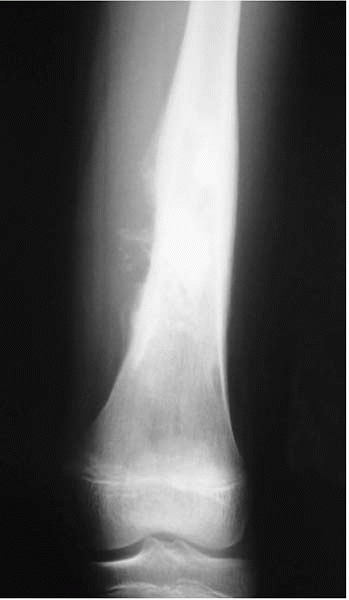
rod a femur with a suspicious looking lesion (Fig. 17-9).
 |
|
▪ FIGURE 17-8 Codman’s triangle associated with an osteosarcoma of the distal femur in a teenager.
|
 |
|
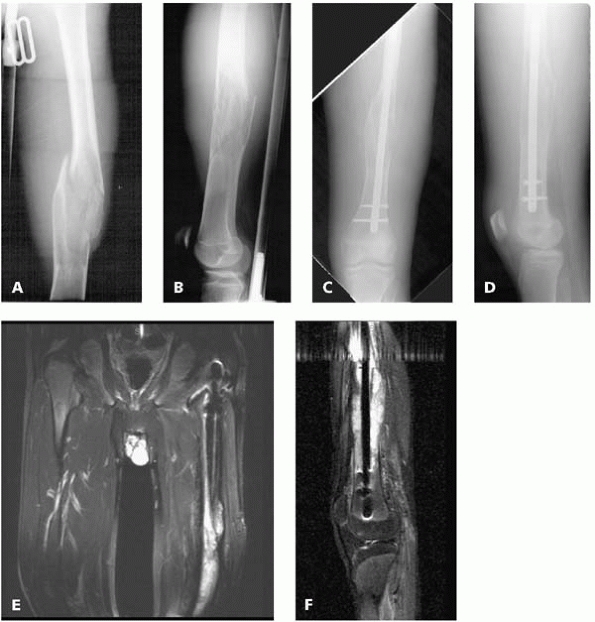
▪ FIGURE 17-9 The worst kind of trouble. This AP of the femur (A) and this lateral of the femur (B) show a pathologic fracture through a large destructive lesion. This AP (C) and lateral radiograph (D)
was taken after the surgeon treated the fracture with an intramedullary rod. The fracture did not heal and continued to show abnormal appearance. The patient was sent on for a consultation. (E,F) These MR images show the intramedullary rod passing through the malignancy in the distal femoral diaphysis. The entire shaft of the bone has been contaminated, compromising attempt at limb salvage surgery. |
because motion can be lost due to a blocking effect by the growing
lesion. It is important not to mistake a parosteal osteosarcoma for a
sessile osteochondroma.
or giant cell tumor (GCT). For this reason, careful evaluation of
imaging studies and a conscientious biopsy of every ABC and GCT will
help keep you out of trouble.
 |
|
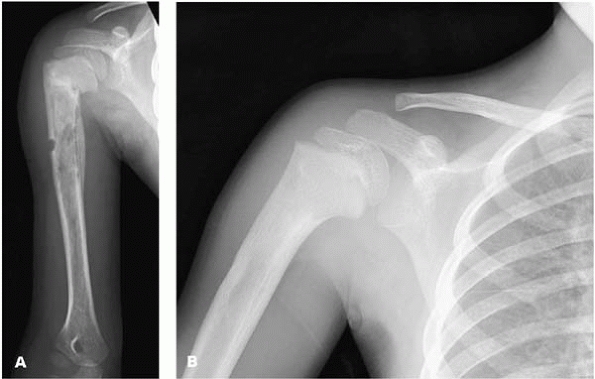
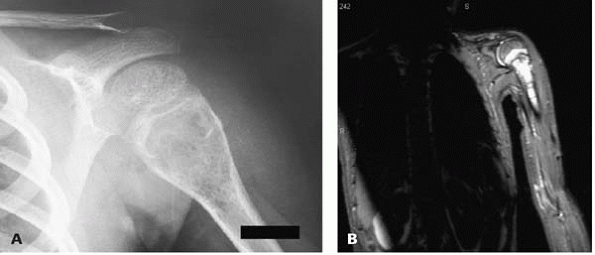
▪ FIGURE 17-10 Beware of dismissing bone cysts as benign, especially expansile lesions with an appearance of an aneurysmal bone cyst. (A)
This boy presented with shoulder pain. His AP shoulder radiograph seems to show a typical, classic aneurysmal bone cyst of his proximal humerus. (B) This MRI shows the lesion extending more distally into the humerus—an uncharacteristic picture. At the time of biopsy, telangiectatic osteosarcoma was identified. Always biopsy an ABC. |
but may have confusing constitutional symptoms such as fever and weight
loss. The femur, pelvis, and humerus are common sites. The appearance
on plain radiographs should alarm anyone familiar with the four
Enneking rules mentioned above. There will be a permeative pattern,
with little reaction by the bone and periosteal “onion-skinning.” Ewing
sarcoma is a very aggressive tumor. MRI will show a large soft-tissue
mass and tumor down the medullary cavity further than was thought from
the plain radiographs.
 |
|
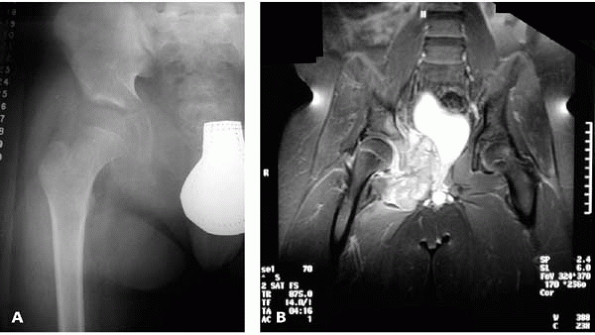
▪ FIGURE 17-11 One key to making the diagnosis of Ewing sarcoma is the large soft-tissue mass. (A)
What is causing the destruction of the superior and inferior pubic ramus and ischium seen on this AP pelvis radiograph of an 11-year-old boy? (B) The MRI shows the huge soft-tissue mass associated with this Ewing sarcoma. |
sarcoma if there are no metastases at presentation. Very few children
with metastases survive.
It is not unusual for a child to present to the orthopaedist with a
soft-tissue mass or lump on an extremity. The vast majority of these
growths are benign: ganglions, popliteal cysts, granuloma annulare,
etc. The key to staying out of trouble with lumps and bumps in children
is to recognize the signs that you may have encountered a malignant
soft-tissue mass:
-
The lesion is deep to the fascia
-
The lesion is bigger than 2 inches (about 5 cm) in diameter
-
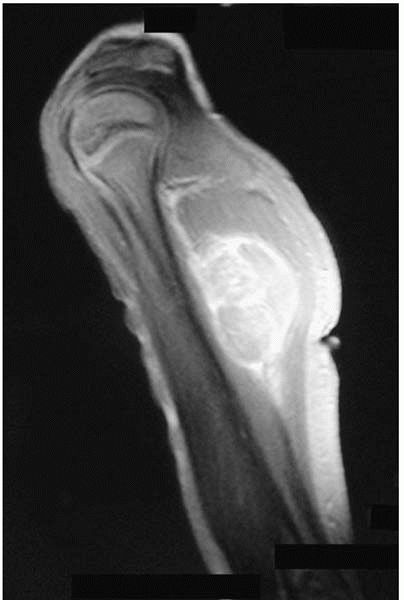
The lesion feels harder than muscle (this can be hard to detect, especially if the lesion is deep) (Fig. 17-12)
 |
|
▪ FIGURE 17-12
This 4-year-old presented with a mass on her upper arm. The lesion was large, deep and hard, all signs that it could be a worrisome process. This MR image shows a large deep lesion that proved to be a rhabdomyosarcoma on biopsy. |
family on to an orthopaedic oncologist for a biopsy. Reassure the
family that benign lesions are much more common than malignancies.
a few different types, but the alveolar is the most common type that
the pediatric orthopedist would encounter in the extremities. The usual
presentation of rhabdomyosarcoma is a painless mass deep within the muscle. Currently, when a child presents with an enlarging soft-tissue mass, an MRI is obtained.
that an orthopaedist will manage. The vast majority of these are in the
proximal humerus or proximal femur. The cyst will begin right below the
physis while it is active, and then the physis will grow away as the
cyst matures, usually during the teenage years. The goal of treatment
is to prevent a pathologic fracture and return the child to normal
activity as soon as possible. Because of the effect on weight bearing
and function, pathologic fracture through a UBC in the femur requires
more aggressive management than the same type of injury in the humerus.
A unicameral bone cyst usually has a very readily identifiable
appearance and location that practically all but assures the diagnosis.
Routine biopsy of every UBC seen on a radiograph is not necessary.
However, biopsy may be done at the time of surgical management to
confirm the diagnosis. Stanton et al.5
reported physeal arrest after treatment, although they had evidence of
early arrest even before treatment. It is possible that the lesion, or
a pathologic fracture through the lesion, led to the physeal arrest
seen in 10% of cases. To stay out of trouble, be certain families are
aware of the small risk of physeal arrest, and be careful with
curettage of lesions near the physis. An iatrogenic growth arrest would
be a high price to pay for a lesion that is known to resolve on its own
later in life.
been suggested for the treatment of UBC in children. This scenario
suggests that no single technique has been considered safe and
successful enough to gain universal adoption. The most successful
methods seem to have a few things in common: connecting the cyst with
the medullary cavity (intramedullary decompression that brings marrow
elements into the cyst) and minimizing surgery (don’t
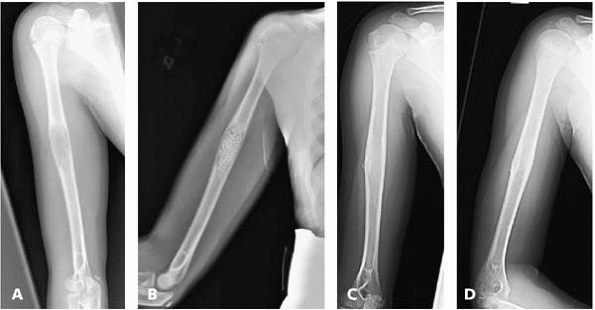
harvest huge amounts of iliac crest bone graft, use smaller incisions, or even just injection) (Fig. 17-13).
 |
|
▪ FIGURE 17-13 (A) This lateral radiograph of the entire humerus shows a lesion with a classic appearance of a UBC. (B) This radiograph was taken immediately after biopsy, curettage, and grafting with osteoset pellets. (C,D) This AP and lateral radiograph of the entire humerus just 6 months after treatment shows complete resolution of the cysts.
|
long bones in about 50% of cases and in the spine in about 30% of
cases. In the spine, they are usually in the posterior elements but can
extend into the body or even into the rib.1
One key to staying out of trouble with ABCs is being able to
distinguish them from their more benign cousin, the unicameral bone
cyst. ABCs are much more aggressive and warrant more aggressive
treatment. Usually ABCs are expansile (Fig. 17-14),
extending wider than the adjacent physis. UBCs, on the other hand, are
not expansile and do not extend beyond the width of the physis. Stay
out of trouble by biopsying every ABC that you encounter. Within the
lesion, there could be a telangiectatic osteosarcoma or giant cell
tumor lurking.
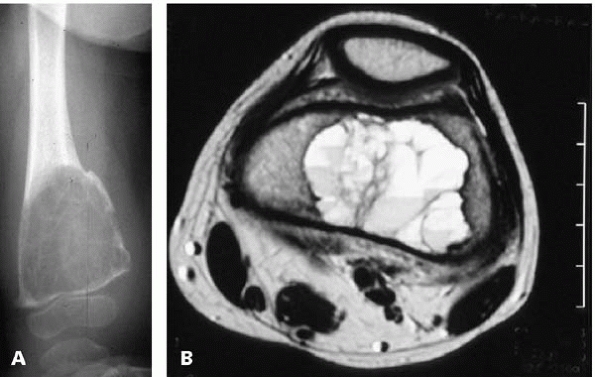 |
|
▪ FIGURE 17-14 (A)
This AP radiograph of the distal femur shows a huge aggressive expansive lesion with a radiographic appearance much more consistent with ABC than UBC. (B) This MRI cut through the distal femoral metaphysis shows the classic layering of blood and fluid in the multiloculated aneurysmal bone cyst. |
are often found on the small bones of the hands and feet. They do not
usually need to be excised, just properly diagnosed. To stay out of
trouble, be certain that you have the diagnosis correct: an enchondroma
should not be painful if it is not fractured. After diagnosing an
enchondroma, order a followup radiograph a couple of months later to
confirm that there is no growth, then counsel the patient about the
small risk of malignant transformation of these lesions in adults (this
is especially true in children with Ollier disease).
that will most often present to the orthopaedist in a school-age child
with a radiolucent lesion of the vertebral body, ilium, or a long bone.
Initially on radiographs, there is usually little reaction around the
lesion. Punched out lesions in the skull is one common manifestation.
Initially, LCH lesions are usually cold on bone scan. To stay out of
trouble, refer the child for evaluation by a pediatric hematologist
once the diagnosis is suspected. Some believe chemotherapy may be
beneficial for the most fulminant forms of the condition.
 |
|
▪ FIGURE 17-15 (A)
This AP radiograph of the forearm shows a destructive lesion in the proximal radial diaphysis. Further workup of the patient revealed multiple lesions and a diagnosis of Langerhan’s cell histiocytosis. Therefore, the lesion did not need to be biopsied or treated. (B) This radiograph of the forearm taken only 7 weeks later shows progressive resolution of the lesion. |
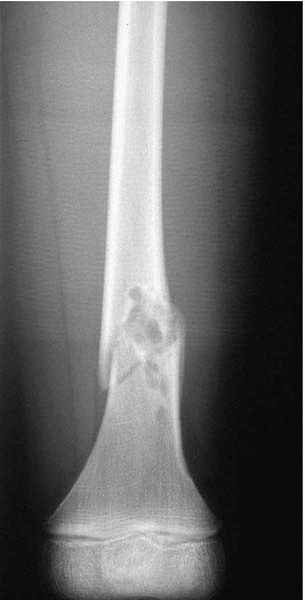 |
|
▪ FIGURE 17-16 A pathologic fracture through a nonossifying fibroma at the junction of the distal diaphysis and metaphysis.
|
are extremely common bone lesions in children. One author estimated
that up to 40% of young children have an FCD/NOF lesion.6
There are two classic appearances. They can be small, cortically-based
radiolucencies but are rarely mistaken for another lesion; these are
usually termed fibrous cortical defect. On the other hand, they can be
eccentric, metaphyseal radiolucencies; these are usually termed
nonossifying fibroma. These are the lesions that are associated with a
pathologic fracture (Fig. 17-16). There should be no periosteal reaction present when the
lesion is initially discovered (unless there has been a fracture in the
past). Fibrous cortical defects are classic “no touch” lesions: most
should be left alone and not biopsied. Treatment of larger lesions is
controversial. Arata7
recommended that lesions greater than 50% of the bone diameter should
be treated with curettage and grafting. More recently, Easley and Kneisl8 recommended that even large lesions can be observed rather than treated with curettage and bone grafting.
orthopaedist as either a teen or preteen with a single lump around the
knee, shoulder or ankle (isolated osteochondroma) or a school-age child
with multiple bumps, possibly limited motion of the forearm or hip, and
a parent who has the same kind of condition (multiple hereditary
exostosis).
lesions will grow as the child grows, but at an unpredictable rate.
Proportional growth of the lesion is not a sign of malignancy in
children, but it may be in adults. The pelvis, spine, scapula, and
proximal femur are sites where there is a higher risk of malignant
degeneration later in life. Recommended management is observation with
an examination and radiograph each year. Some lesions may cause
problems as they enlarge. Such problems are usually, but not always, in
children with multiple hereditary osteochondromatosis. The enlarging
lesion can lead to hip subluxation.9
is similar to staying out of trouble with UBCs: avoid overtreatment
(and collateral damage during treatment). One study10
reported on 80 patients who had excision of 285 osteochondromas. There
were 10 complications in the group (12.5% per patient population); the
most common were peroneal neurapraxias. Other complications included an
arterial laceration, a compartment syndrome, and a fibula fracture. The
authors concluded that the surgical risk for the management of
osteochondromas is low and is comparable to the risk of other related
elective procedures. However, osteochondromas should not be routinely
excised unless proper indications exist for their removal. A sessile
osteochondroma is rarely symptomatic. If you choose to remove an
osteochondroma because of symptoms, consider the risk to adjacent
neurovascular structures (especially in the proximal medial humeral
area) or the adjacent physis (be especially aware of physeal damage
when excising osteochondromas in very young children). An
osteochondroma can split a nerve, putting it at great risk at the time
of resection.11
medical history is night pain that is relieved by either aspirin or
nonsteroidal antiinflammatory drugs. It can be very hard to diagnose an
osteoid osteoma on plain radiographs alone, especially if the osteoid
osteoma is in the spine or the pelvis. Stay out of trouble by getting a
fine-cut CT to confirm the diagnosis (Fig. 17-17).
Beware of getting an MRI: an osteoid osteoma on MRI can be a very scary
looking lesion with adjacent bone edema. To avoid getting an MRI
reading with a differential diagnosis that includes malignancies and
infections, be sure your radiologist knows that you suspect an osteoid
osteoma. If you are going to take the child to the operating room to do
an excision, be certain that you can find the lesion when you get
there! Helpful suggestions include measuring the distance
between the lesion and bone landmarks, or using intraoperative radionuclide or tetracycline-fluorescent labeling.
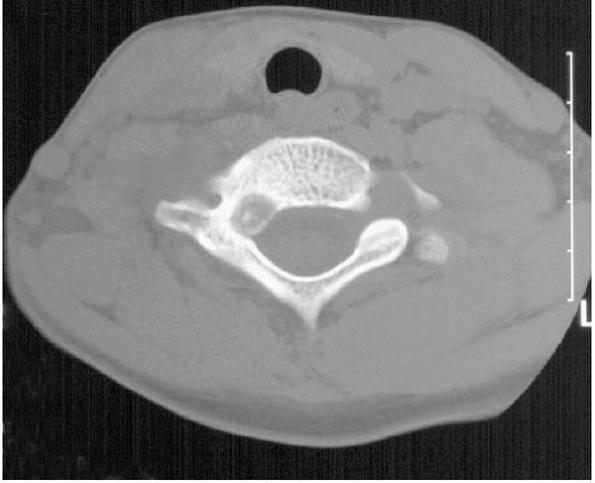 |
|
▪ FIGURE 17-17
Fine cut CT is extremely valuable to identify an osteoid osteoma of the spine, and clarify its location. This child presented with neck pain and a classic osteoid osteoma history. The lesion, however, was very difficult to visualize on plain films. The nidus is seen beautifully on this fine cut CT section. |
is to be certain that all of nidus is excised. Depending on the
location lesion and surgeon preference, this can be accomplished with
either curettage or en block resection.
extremity, it is important to instruct protective weight bearing
afterwards to avoid pathologic fracture.
-
Don’t be lulled into a false sense of security if the pain has “only been there for a couple of weeks.”
-
Pain from cancer and
infections are often written off as resulting from injury. Because the
life of a child is filled with falls and bumps and bruises, beware of
attributing all pain to injury. -
Children with Ewing
sarcoma can have signs and symptoms that look like an infection, as
well as a fever and elevated CRP and ESR. -
Look at all fracture radiographs closely to be certain that the fracture is not a pathologic fracture.
-
Study the full extent of every radiograph of a kid who presents to you with pain.
-
When ordering the
MRI for a child with a potential malignancy, it is important to work
with your radiologist, communicating specifically what you are looking
for and that a malignancy is in the differential. A biopsy should be
used to confirm what is suspected from a small list of possibilities. -
If at all possible,
the biopsy should be done at the center where the child will be
treated, by the surgeon who is going to do the definite cancer
operation.
-
If there is
uncertainty, go over the radiographs with an experienced bone
radiologist who has an atlas of normal variants available. -
Biopsy of myositis
ossificans can be dangerous, because the pathology can look so
aggressive that the lesion might be mistaken for a malignancy.
-
Should you encounter
a pathologic fracture through a lesion suspicious for osteosarcoma,
closed management of the fracture is recommended. -
Careful evaluation of imaging studies and a conscientious biopsy of every ABC and GCT will help keep you out of trouble.
-
The large soft-tissue mass on MRI is the key to making a radiographic diagnosis of Ewing sarcoma.
-
Recognize the signs that you may have encountered a malignant soft-tissue mass:
-
The lesion is deep to the fascia.
-
The lesion is bigger than 2 inches (about 5 cm) in diameter.
-
The lesion feels harder than muscle (this can be hard to detect, especially if the lesion is deep).
-
-
Avoid overtreatment of UBC and osteochondroma.
-
When managing a
child with a UBC, be certain families are aware of the small risk of
physeal arrest, and be careful with curettage of lesions near the
physis. -
Aneurysmal bone cysts are much more aggressive and warrant more aggressive treatment.
-
ABCs can be
secondary to, or associated with, other lesions. These include not only
telangiectatic osteosarcoma and giant cell tumors, but also
chondroblastoma and nonossifying fibroma. -
Fibrous cortical defects are classic “no touch” lesions: most should be left alone and not biopsied.
-
After excising a large osteochondroma, consider providing the child with appropriate immobilization.