Nerve Decompression
surgery for ulnar nerve compression lesions at the elbow. Although
conservative management may be appropriate for spontaneous ulnar
neuritis, it is not indicated in certain instances associated with
fractures or dislocations. If the ulnar nerve is entrapped or impaled
by fracture fragments, or trapped in the joint following reduction of a
dislocation, early surgery is appropriate. If an open injury exists or
if open reduction of the fracture is necessary, then primary
concomitant surgical evaluation and management of the ulnar nerve
should be performed.
objective signs of neuropathy are not present. The conservative
management consists of instructing the patient to avoid leaning on the
elbow on the inner aspect and prolonged elbow flexion. A long arm
splint with the elbow at 70 degrees of flexion, the forearm in
midposition, and the wrist in neutral position is worn full-time for
approximately 4 to 6 weeks. With improvement, the splint is then used
only at night for an additional 2 to 3 months. If the patient improves
with this management, as happens in about 50% of cases, surgery can be
avoided.
subjective complaints. Often the numbness that is in the little finger
improves after 10 days; on occasion, symptoms are relieved within a day
or two. Weakness of the hand is a common finding in ulnar paresis. Even
without clawing of the fingers, there may be weakness of the intrinsic
muscles. In our experience, the pinch dynamometer mirrors the
electromyographic studies. If the strength improves, so does the
electrical conduction across the elbow.
continued, usually for 3 months. After removal of the night splint, the
patient is instructed not to sleep with the elbow
fully
flexed, but rather to put it out straight or on a pillow at night. If
necessary a 70-degree flexion splint can be worn. Certainly if the
condition recurs and there is no evidence of atrophy of the intrinsic
muscles, the splint can be worn for an additional 6- to 8-week period.
subside, and if there is progressive atrophy of the intrinsic muscles
and weakness as observed with the pinch dynamometer. Serial
electrodiagnostic studies in this instance, if performed, reveal
slowing of conduction of the ulnar nerve across the elbow, and
fibrillations and sharp waves in the ulnar nerve innervated intrinsic
muscles.
compensation or pending litigation, surgical intervention should be
considered very carefully and reluctantly.
patient can have a pure ulnar nerve lesion at the elbow or a
double-crush lesion, most frequently at the elbow and the neck.
Fibrillations on electromyographic studies in the cervical
paravertebral muscle region would suggest a more proximal lesion. A
conduction block across the elbow would confirm and localize the lesion
to the elbow. Nevertheless, one can have a lesion at both regions in a
double-crush lesion.
compression at the elbow is numbness in the dorsal and palmar aspects
of the little finger and half of the ring finger. For clinical
localization of the neural lesion to the elbow level, sensory loss on
the dorsoulnar aspect of the hand is a characteristic finding. With
more proximal neural involvement (i.e., in the chest, thoracic outlet,
or neck), a different pattern of sensory disturbance involving the
forearm or arm would be present. However, when the little finger is
numb only on the palmar side, the ulnar nerve compression is usually at
the level of the canal of Guyon.
result in paresis or paralysis of the flexor carpi ulnaris, flexor
digitorum profundus of the ring and little fingers, and the
ulnar-innervated hand intrinsics. A positive percussion sign helps
localize the problem to the elbow, but false-positives are not uncommon
findings in patients with “irritable” nerves. Dislocation of the ulnar
nerve, with or without snapping on elbow flexion, is often associated
with a positive percussion sign at the elbow level. It is also
important to be sure that the patient who has a symptomatic dislocating
and snapping ulnar nerve does not have a coexistent snapping medial
triceps. The dislocating ulnar nerve and snapping triceps can be
diagnosed clinically preoperatively (and typically intraoperatively);
it can be embarrassing to translocate the ulnar nerve anteriorly and
have persistence of the snapping postoperatively with elbow flexion due
to a triceps snap.
remain controversial. In our practice, we have attempted to correlate
electromyographic studies to the type of operative procedure. The
simple release of the arcade and the cubital tunnel retinaculum can be
utilized in those cases in which the compression is at that site and
the patient has persistent symptoms of mild ulnar neuropathy with
minimal or no electrical findings after appropriate conservative care
fails. It should not be utilized with a dislocating ulnar nerve.
ulnar nerve, it is best to perform anterior translocation, since the
soft-tissue restraints typically released in an in situ
decompression are not present. A completely dislocating ulnar nerve, in
contrast to a subluxating nerve, rarely causes problems. However, thin
patients with minimal subcutaneous fat may especially become
symptomatic. A dislocating nerve may be vulnerable to friction over the
medial epicondyle; a subluxating nerve may be susceptible to
compression against the epicondyle or by direct trauma.
practiced surgical procedure. We prefer the anterior translocation,
either subcutaneous or submuscular, over in situ
decompression, when there is electrical indication confirming the
patient’s more severe ulnar nerve involvement, especially when axonal
degeneration is present. Of subcutaneous and submuscular transposition,
we prefer the submuscular transposition.
authors. We have not been placing the nerve intramuscularly, as we have
often treated patients with recurrent neurologic symptoms. We do not
recommend the modification of the Learmonth procedure in which the
flexor pronator muscles are detached directly from the epicondyle and
reattached to it, or when the epicondyle is osteotomized. In addition,
we do not perform medial epicondylectomy.
The arm is externally rotated and a few folded sheets are placed under
the elbow to elevate it. The medial aspect of the elbow is in view,
with the medial epicondyle prominent. When the ulnar nerve is not to be
translocated, a longitudinal incision 6 to 8 cm long is made centered
at the region of the cubital tunnel retinaculum. The terminal branches
of the medial cutaneous nerve of the arm and the crossing branches of
the medial cutaneous nerve of the forearm in the region of the
olecranon should be preserved. They are found deep to the fat just
superficial to the fascia overlying the flexor carpi ulnaris (Fig. 16-2).
A rubber band is placed about each of them so they can be retracted.
The ulnar nerve, which passes longitudinally just deep to the
aponeurotic origin of the flexor carpi ulnaris, can be palpated.
which is about 4 mm wide, is released. Hemostasis is obtained with a
Malis hyfrecator; an epineurotomy of the ulnar nerve is usually not
necessary. If, after release of the soft-tissue restraints, the ulnar
nerve dislocates with elbow flexion, an anterior transposition should
be performed. The wound is closed.
 |
|
Figure 16-1. The arm is draped free and placed on an arm board. The proposed incision for an in situ decompression is demonstrated.
|
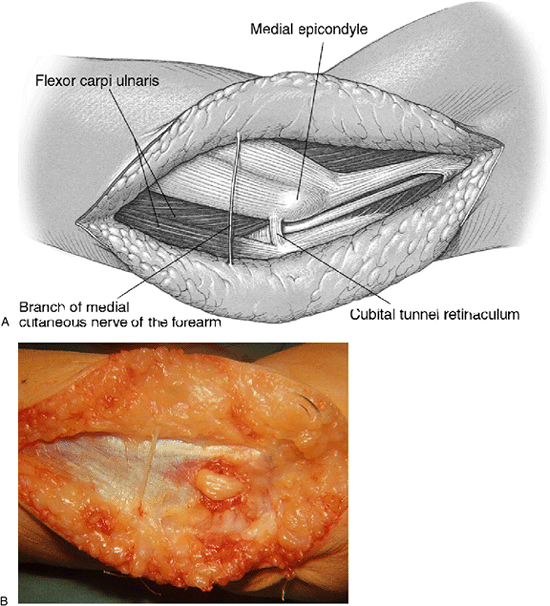 |
|
Figure 16-2. A,B: Typical appearance of the nerve at the cubital tunnel. Note the crossing branch of medial cutaneous nerve of forearm.
|
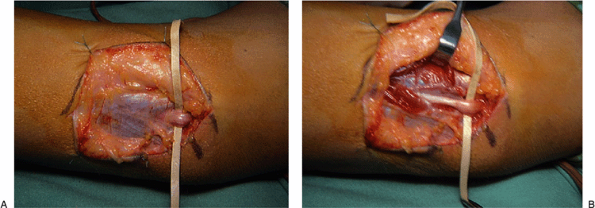 |
|
Figure 16-3. Compression of the nerve at this level (A) is seen clearly following release of the cubital tunnel retinaculum (B).
|
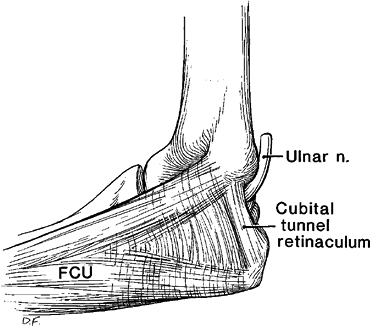 |
|
Figure 16-4. A schematic representation of the cubital tunnel.
|
In a primary case, an approximately 15-cm-long incision is made, curved
posterior to the medial epicondyle, centered on the epicondyle. The
medial cutaneous nerves of the arm and forearm are identified and
preserved. The ulnar nerve is identified in virgin tissue proximal to
the elbow and mobilized with its extrinsic vasculature. The arcade of
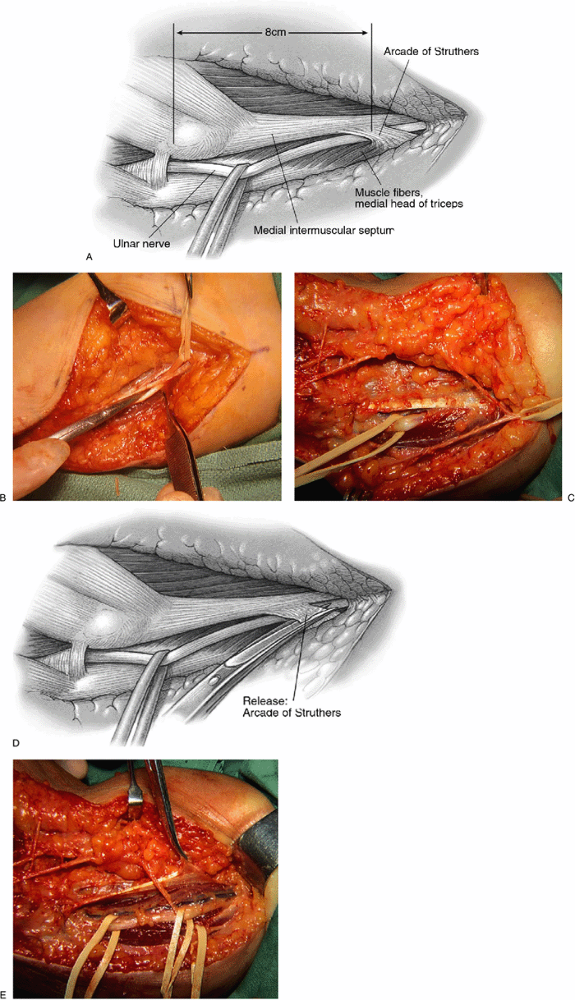
Struthers is released when it is found 8 cm proximal to the epicondyle (Fig. 16-5A,B).
An anatomic point for the presence of the arcade is in the appearance
of muscle fibers of the medial head of the triceps crossing superficial
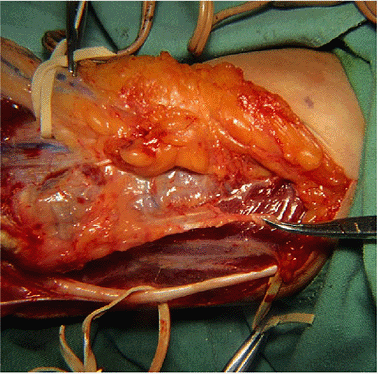
to the ulnar nerve (Fig. 16-5C). The arcade is released (Fig. 16-5D,E), and the medial intermuscular septum is freed and excised (Fig. 16-6).
retinaculum is released, as is the flexor carpi ulnaris aponeurosis.
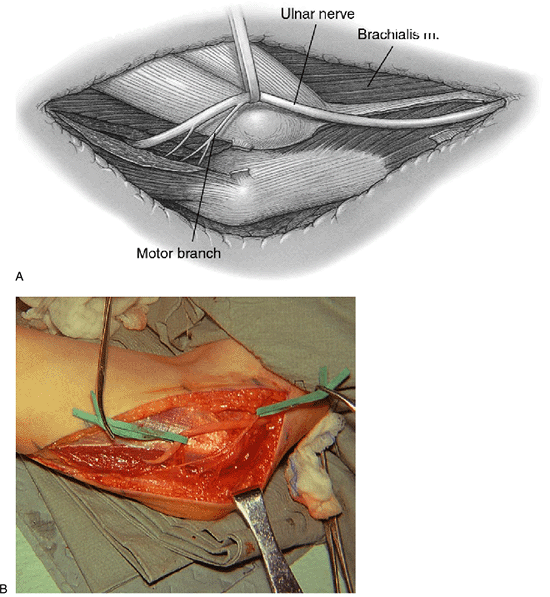
Just distal to the elbow joint, the articular branch is encountered and
should be released. The motor branches to the flexor carpi ulnaris and
the flexor digitorum profundus branch should be preserved. These
branches are mobilized microsurgically from the ulnar nerve as much as
necessary to permit the anterior neural transposition. These steps
permit the ulnar nerve to be easily brought forward over the epicondyle.
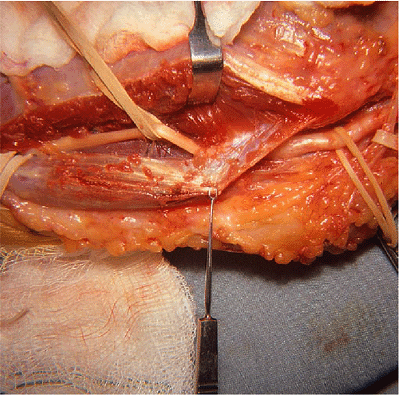
either plane to prevent kinking of the ulnar nerve distal to the
epicondyle by the common aponeurosis between the flexor digitorum
superficialis of the ring finger and the humeral head of the flexor
carpi ulnaris (Fig. 16-7). When the nerve is translocated anteriorly in the subcutaneous plane (Fig. 16-8), it therefore runs a straight course.
to the antebrachial fascia just lateral to the epicondyle. This
maintains the ulnar nerve anteriorly. A fascial sling can be utilized
alternatively to stabilize the nerve in an anterior position.
described in the subcutaneous technique. It differs in that the
incision is longer, being 8–10 cm proximal and distal to the
epicondyle. Furthermore, a sterile tourniquet is utilized.
it is technically demanding with two principles in mind. First, the
ulnar nerve must pass in a straight line. The longitudinal extrinsic
vascular supply should be maintained during mobilization of the ulnar
nerve. This should be preserved as far distally as possible by ligating
the muscular communicating branches, thus keeping the extraneural
vascular supply with the ulnar nerve. One can usually maintain the full
length of the venae commitantes to the elbow level. The vascular
variations about the elbow usually prevent full maintenance of the
extrinsic vascular supply. On rare occasion, the senior author has seen
an ulnar nerve that had only an intrinsic vascular supply and he
mobilized it as necessary to perform the procedure without difficulty.
Second, the ulnar nerve must run parallel to the median nerve in a good
soft tissue bed. If the deep bed is inadequate, as in severe arthrosis
or with bad articular fractures, especially
of the medial half of the elbow joint, a subcutaneous transposition of the ulnar nerve is a better choice.
 |
|
Figure 16-5.
The arcade of Struthers is demonstrated with the ulnar nerve passing through it. It arises about 8 cm proximal to the epicondyle (A,B). Muscular fibers of the medial head of the triceps are seen when an arcade is present (as in approximately 70% of limbs) (C). Release of the arcade of Struthers is necessary when translocating the ulnar nerve anteriorly (D,E). |
 |
|
Figure 16-6. The medial intermusuclar septum (in the hemostat) is being removed.
|
 |
|
Figure 16-7.
To prevent an entrapment of the ulnar nerve distal to the medial epicondyle, the common aponeurosis of the origin of the flexor digitorum superficialis and the humeral head of the flexor carpi ulnaris must be released. |
 |
|
Figure 16-8.
Subcutaneous anterior translocation of the ulnar nerve is seen with the nerve in a straight line. The articular branch has been released. The muscular branches are preserved (A,B). |
fibrosus is visualized by elevating the lateral skin flap. The
cutaneous nerves are preserved. The median nerve is identified deep to
the lacertus. A rubber band is placed about it and the overlying fascia
is released. The limits of the flexor pronator muscles are clearly in
view. The median nerve is lateral to it and the ulnar nerve is medial.
A large tonsillar hemostat is inserted just distal to the epicondyle
from the lateral side deep to the flexor pronator muscles, avoiding the
recurrent vessels. The flexor pronator group of muscles is severed 1.5
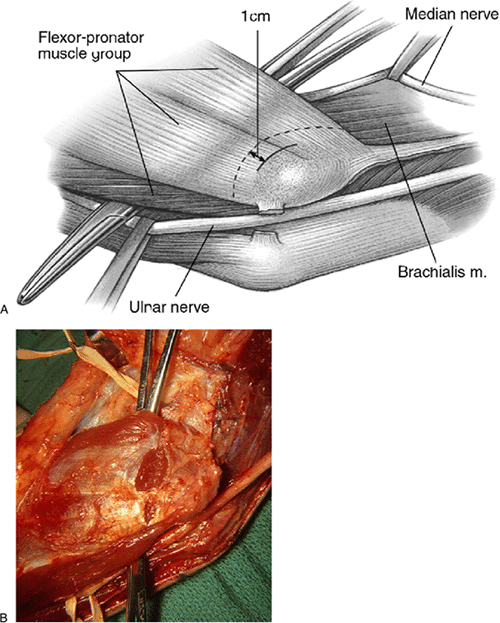
to 2 cm distal to the medial epicondyle (Fig. 16-9).
The exit of the hemostat on the medial side may be troublesome, and the
lateral two-thirds of the muscle is first incised and then the hemostat
is repassed. Muscle bleeders are clamped and tied. Using a periosteal
elevator, the flexor pronator muscles are stripped distally. Motor
branches to the flexor pronator mass may need to be neurolyzed to
prevent undue tension on them during the muscle elevation.
and the flexor pronator group of muscles is repaired either in its
original or in an elongated (e.g., Z-lengthened) position. Multiple 2-0
interrupted Maxon sutures are utilized. The fascia of the medial
longitudinal incision in the flexor pronator muscles is closed with 3-0
chromic sutures (Fig. 16-11). The skin is closed with interrupted nylon sutures or staples.
 |
|
Figure 16-9.
The tonsillar hemostat is deep to the flexor pronator group of muscles. It is usually inserted from the lateral side. Care is given to avoiding the collateral vessels on the anteromedial aspect of the elbow joint. It is superficial to the brachialis fascia. The adjacent rubber band is about the median nerve. The ulnar nerve is medial (A,B). |
elbow should be flexed and extended passively to ensure that any site
of compression has been eliminated and that any dislocation of the
nerve or triceps has been corrected. A dislocating medial triceps can
be treated by excision or lateral transposition of the offending
portion of triceps (Fig. 16-13).
nerve symptoms, often admixed with elbow pain, sometimes with snapping.
Failed ulnar nerve surgery is typically due to either
misdiagnosis
or compression from incomplete decompression of the original pathology
or from secondary compression due to kinking at a nonreleased proximal
or distal structure, scarring within a surgical bed or constriction by
an overly tight sling. Neuromas from cutaneous nerves may result in new
pain or dysethesias. Recurrent ulnar nerve dislocation may result from
a ruptured sling or suture, and persistent snapping may become manifest
after transposition due to unrecognized dislocation of the medial
triceps.
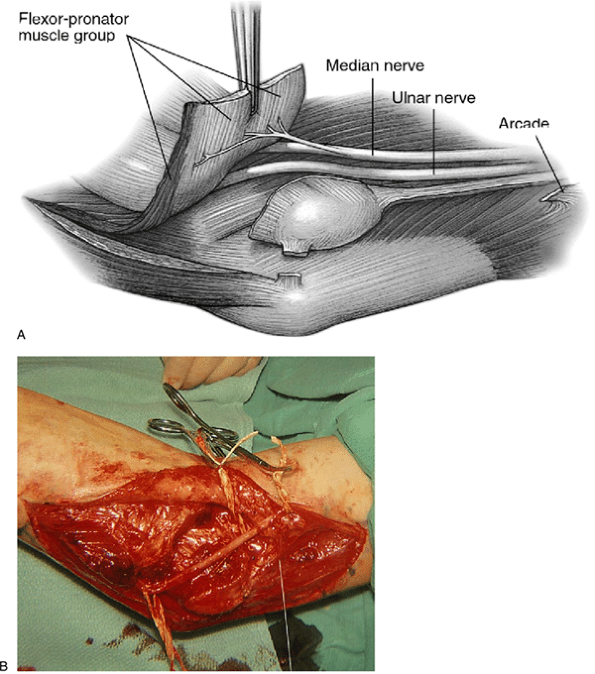 |
|
Figure 16-10. The tourniquet has been released. The ulnar nerve has been transferred deep to the flexor pronator group of muscles (A) adjacent to the median nerve (in rubber band) (B).
|
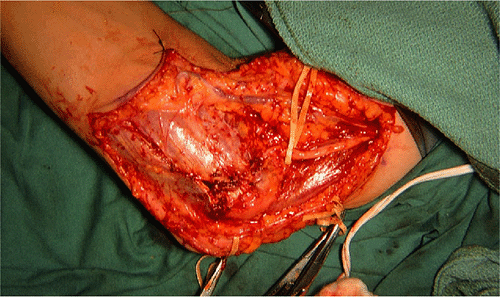 |
|
Figure 16-11. The flexor pronator group of muscle has been repaired. The ulnar nerve has been translocated submuscularly.
|
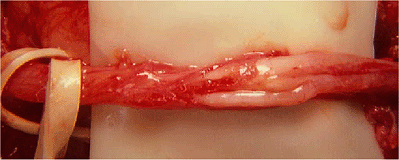 |
|
Figure 16-12. Epineurotomy of the nerve is necessary in some secondary cases.
|
revision surgery is extremely difficult. Based on our experiences, we
are more cognizant of the potential for kinking of the ulnar nerve when
just a small incision is made and the nerve is pulled anteriorly. It
can angulate very easily. Kinking of the ulnar nerve typically occurs
several centimeters proximal or distal to the medial epicondyle. For
example, the translocated nerve may be entrapped in a dense tendinous
path, especially if the distal aponeurotic arch has not been released (Fig. 16-14). Other factors that may sway us include patients who underwent previous in situ
decompression without relief, or intramuscular transposition with
transient relief. Conversely, factors that would discourage secondary
surgery include procedures performed by experienced surgeons or
operative notes that sounded technically sufficient, or patients with a
bizarre array of somatic complaints. In addition, patients presenting
with evidence of chronic, severe neurologic findings or muscle fibrosis
would not benefit from revision. In these patients, one can estimate
how many motor fibers are left in the hand with the pinch dynamometer.
If the strength of pinch on the dynamometer is less than 1 kg, motor
fibers
may
well be all out and the nerve can be almost nonfunctioning. Electrical
conductivity can be on the order of 0 to 17 m/sec across the elbow. If
the patient has had a prior anterior translocation and is in this poor
state, submuscular transposition may offer little if any chance of
success and could increase the symptomatology with an infarcted nerve.
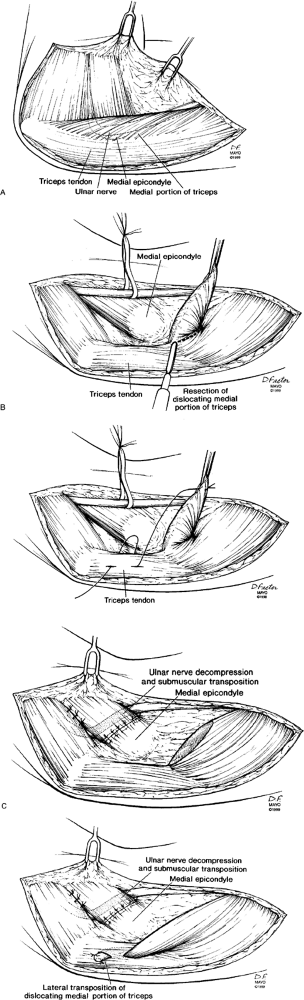 |
|
Figure 16-13. A:
Snapping of a portion of the medial triceps and dislocation of the ulnar nerve needs to be considered in patients undergoing ulnar nerve surgery. Patients should be examined for snapping triceps preoperatively and intraoperatively. B: The offending dislocating portion of the triceps can be excised or transposed laterally. C: Neurolysis and transposition of the ulnar nerve can then be performed. A submuscular transposition was performed in these examples. Snapping of the triceps was eliminated. (From Spinner
RJ, O’Driscoll SW, Jupiter JB, et al. Unrecognized dislocation of the medial portion of the triceps: another cause of failed ulnar nerve transposition. J Neurosurg 2000;92:52–57 , with permission.) |
Brunner zigzag type is frequently used in secondary procedures. The
incision often extends 10 to 12 cm proximal and distal to the
epicondyle. It is particularly important to define normal tissue
planes. To identify the ulnar nerve through the region of the earlier
surgery most easily, one should isolate the nerve proximally and
distally and then trace it through the elbow region. The multiple
muscular branches to the flexor carpi ulnaris and the one to the flexor
digitorum profundus
should
be preserved. Of these, the most important is the branch to the flexor
digitorum profundus; failure to maintain its continuity results in the
inability to flex the distal interphalangeal joint of the little
finger. On occasion, the ulnar nerve distally must be identified in the
midforearm. The plane between the flexor carpi ulnaris and flexor
digitorum superficialis is developed to identify and trace the ulnar
nerve proximally to the elbow. Crossing vessels are clamped and tied
with fine sutures. The deep fascia over the nerve is released to permit
the mobilization. The muscular branches are preserved. The Learmonth
procedure has proved to be the most reliable salvage operation.
from the primary surgery. These must be removed, and the ends of the
nerves should retract into normal fatty tissue. To prevent recurrent
neuromas, gently pull down the involved medial cutaneous nerve of the
arm or forearm and double-crush the nerve a centimeter apart. The site
of the distal crush is tied with 5-0 nonabsorbent suture and the nerve
is severed just distal to the suture. The nerve ends can also be
bipolared.
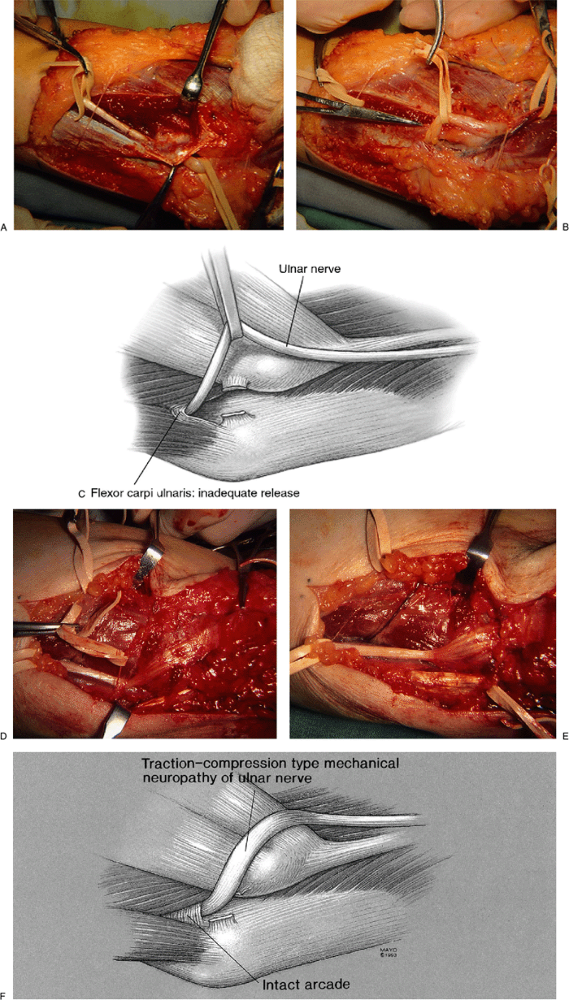 |
|
Figure 16-14. The ulnar nerve is seen passing below the arcade distal to the medial epicondyle (A).
If the arcade is not released, then kinking at the level distal to the elbow joint can be produced with a limited anterior ulnar nerve translocation (B,C). A case in which this was the cause of the resistant ulnar nerve neuritis: A traction-compression type mechanical neuropathy of the ulnar nerve is noted distal to the epicondyle (D–F). |
of a thin Silastic sheathing on one side of the nerve. Usually there is
good fat on the anterior side, while the Silastic membrane is placed on
the side that is adjacent to the dense fascial scar. The Silastic is
never put circumferentially about the nerve. This procedure is utilized
strictly to help with the traction neuritis.
decompression and transposition procedures, the extremity is
immobilized in a long-arm posterior splint for 5 to 7 days. Full motion
is encouraged after the splint is removed in patients who underwent in situ
decompression. In patients who had transposition and are relatively
pain-free, controlled movement is then initiated that balances the need
for gliding of the ulnar nerve in its new transposed bed, and adequate
time for healing
(e.g.,
submuscular repair or fascial sling). While anterior translocation
provides a more lax environment for the nerve (i.e., an approximately
2-cm-shorter course of the nerve), earlier movement is thought to
improve nerve gliding with filmier and longer adhesions that have
excursions to them. In this group of patients, elbow flexion is
permitted. Elbow extension is gradually increased during the subsequent
2 weeks. As numbness in the little finger resolves and the preoperative
pain decreases, movement is progressed once or twice a day for a few
minutes over the 3-week period. By starting the mobilization in this
manner, providing that there are no untoward symptoms, the patient
comes out of the dressing and immobilization at 3 weeks and elbow
movement is almost, if not fully, complete. One of the authors (RJS)
has extended this concept and starts movement immediately after
transposition. Physical therapy is required following the Learmonth to
help strengthen and mobilize the arm. In cases where there is digital
clawing, an ulnar metacarpophalangeal (MP) joint flexion cap orthosis
is often required to prevent hyperextension at the MP joint.
tunnel, the patient is frequently back to work by 3 to 4 weeks. With
the anterior subcutaneous transposition, 6 to 8 weeks are required, and
with the submuscular Learmonth procedure the disability is usually 3
months.
treatment for ulnar decompression that are not conclusive. Because of
variable degrees of compression and neural involvement preoperatively,
comparison of different studies and techniques has been difficult.
Typically, in addition, the results are strongly a function of the
surgeon rather than his or her technique. If the procedure is done
properly, approximately an 80–90% satisfactory result can be achieved
following ulnar nerve decompression.
nerve surgeries are instructed that we are looking for overall 50%
improvement. In them the goal of surgery is often to decrease the
severity of the pain. Some improvement in sensation and in strength of
the hand, as well as range of movement of the fingers, can be achieved.
The Learmonth procedure never restores a fully normal arm in secondary
or tertiary cases.
neuromas are not uncommon following nerve decompression. In our
experience, ulnar nerve symptoms develop from mechanical irritation
that persists or is brought about by an improper translocation, usually
with a kink or bend in the nerve or the nerve placed in a region that
allows intense scarring. The treatment for this is ideally to avoid the
circumstance. This can be done by placing the nerve in as straight a
line as possible, as has been emphasized in this surgical technique.
Cutaneous nerves must be identified and preserved.
motor dysfunction that required an extensive dissection. A submuscular
transposition was performed. Here the flexor pronator group was
elevated to a large degree to avoid kinking of the nerve (Fig. 16-15).
of the elbow are the posterior interosseous and the superficial radial
nerves. The posterior interosseous nerve basically is motor, whereas
the superficial radial is sensory.
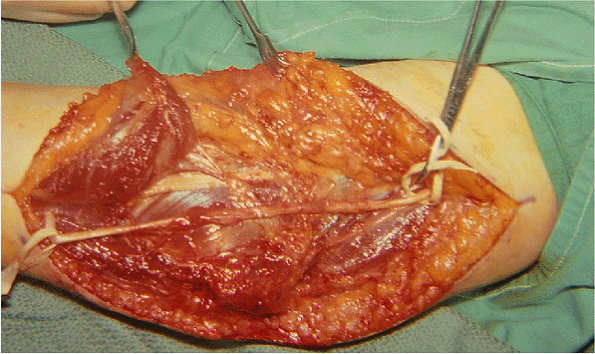 |
|
Figure 16-15. Adequate mobilization of the ulnar nerve permitted its straight course during this submuscular translocation.
|
brevis usually arises between these two nerve branches at the division
of the radial nerve.
type arborization pattern 1 cm distal to the distal edge of the
supinator muscle.
interosseous nerve pathology. The key to localizing the lesion is the
classic appearance of the hand. The wrist dorsiflexes in a radial
direction, and the fingers do not extend at the MP joints. There is a
dropped thumb (Fig. 16-16). Sensation is
intact. This is the presentation with a complete lesion. A partial
posterior interosseous nerve paralysis presents with weakness or
paralysis of some of the digits at MP joints. When the ring and little
fingers are initially involved, the term pseudo-ulnar claw
applies. If there is a dropped wrist, the localization is more proximal
in the arm. In contrast to posterior interosseous nerve compression,
superficial radial nerve compression would produce isolated sensory
disturbance in the dorsoradial aspect of the hand. More commonly, the
irritation occurs more distally at the radial styloid or where the
nerve passes from beneath the brachioradialis; however, it may occur in
the proximal forearm as well.
The site of percussion tenderness can help localize the level of the entrapment to the proximal or distal forearm.
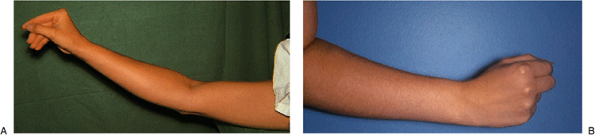 |
|
Figure 16-16.
Typical appearance of a hand with a complete posterior interosseous nerve paralysis. The fingers and thumb do not extend at the MCP joints (A). The wrist does extend but in a radial direction (B). |
the proximal end of the supinator muscle (i.e., at the arcade of
Frohse). The offending pathology is either the arcade alone or in
association with a systematic process like rheumatoid arthritis with
synovitis of the elbow joint, a lipoma, a radial bursa, a ganglion, or
other soft-tissue tumors. Routine radiographs are of help if they
reveal a soft-tissue shadow with the appearance of a lipoma in the
region. Changes in the joint suggestive of rheumatoid arthritis in
association with a synovitis are often diagnosed with articular changes
in the radial head and the adjacent elbow joint. The combination of a
posterior radial dislocation of the elbow in a Monteggia fracture with
posterior interosseous nerve paralysis is not an infrequent coexistent
condition. Similarly, comminuted fractures of the radial head,
especially with displacement, can be associated with this specific
motoneuron involvement. Fractures and bullet wounds of the proximal
third of the radius can be associated with this neural complication.
Furthermore, in preoperative planning, computed tomography (CT) and
magnetic resonance imaging (MRI) are occasionally helpful in
delineating further soft-tissue masses that can be palpable or
nonpalpable.
plating a fracture of the junction of the middle and proximal third of
the radius. A six-hole plate may well catch the posterior interosseous
nerve and compress it against the bone. The postoperative radiographs
reveal the most proximal screw at the level of the bicipital
tuberosity. The posterior interosseous nerve passes obliquely directly
opposite the bicipital tuberosity, where there is a bare area and
entrapment as a complication of plate fixation if diagnosed early can
be corrected. Exploration, neurolysis of the posterior interosseous
nerve, and replacement of the plate with one that is one hole shorter
proximally are usually successful. Similarly, if the posterior
interosseous nerve had been identified, a rubber band had been placed
about it for retraction at the time of the plating, and paralysis is
present after surgery, it is logical to observe this complication
without intervention. The lesion may well be neuropractic, because of
excess traction on the nerve at the time of the plating. The paralysis
may recover spontaneously. In a similar fashion, if one wants to remove
a long six-hole plate with three holes proximal to the fracture level,
it is wise to identify the posterior interosseous nerve proximally in
normal soft tissues. It is then traced through the plated area before
removal of the screws and plate. We have treated several patients who
have had a posterior interosseous nerve paralysis following plate
removal in this area, if the nerve had not been identified before
removal of the hardware.
spontaneous, the lesion most often is at the arcade of Frohse.
Electromyographic (EMG) studies will reveal fibrillations in the
supinator muscle. If, however, the pathology is at the level of the
distal end of the supinator, innervation of the supinator will not be
altered. There is a fibrotic band within the muscle that has been
described with spontaneous compression directly under the supinator
muscle. Here EMG changes within this muscle are most often found.
nerve decompression: the posterior (Thompson) and the anterior (Henry)
approaches. If the arcade of Frohse is to be exposed, this is done
anteriorly. If the distal third of the supinator is involved, then the
exposure can be done either from a posterior approach or with a
combination of the anterior and posterior approaches. If there has been
prior surgery in the region, then the combined approach must be
utilized to gain control of both the proximal and distal portions of
the posterior interosseous nerve.
posterior approach, the patient is supine. The arm is draped free and
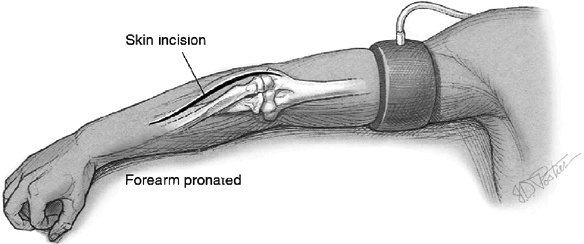
is placed on an arm board, and the forearm is pronated (Fig. 16-17).
 |
|
Figure 16-17.
To expose the posterior interosseous nerve the arm is draped free and placed on an arm board. The Thompson incision is used to expose the proximal forearm. The incision passes in a line from the wrist to the center of the lateral epicondyle. |
the arm. In this approach the skin incision is linear in line with the
midportion of the wrist and the lateral epicondyle (Fig. 16-17).
The incision in the proximal half of the line is extended further
proximally onto the lateral ridge of the epicondyle. The posterior
cutaneous nerve of the forearm is identified and preserved. The plane
between the extensor carpi radialis brevis and extensor digitorum
communis is developed from the level of the outcropping muscles in the
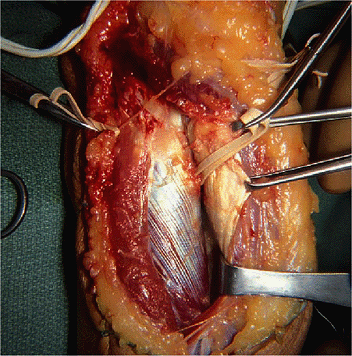
midforearm proximally (Fig. 16-18). The
supinator muscle is seen on the deep plane at the level of the
epicondyle. To expose the arcade of Frohse, the following variation in
the exposure can be applied: Detach the extensor carpi radialis from
its origin at the distal humerus and strip the distal half of the
extensor carpi radialis longus. By flexing the elbow and rotating the
forearm, the direction and course of the posterior interosseous nerve
deep to the superficial head of the supinator is determined by digital
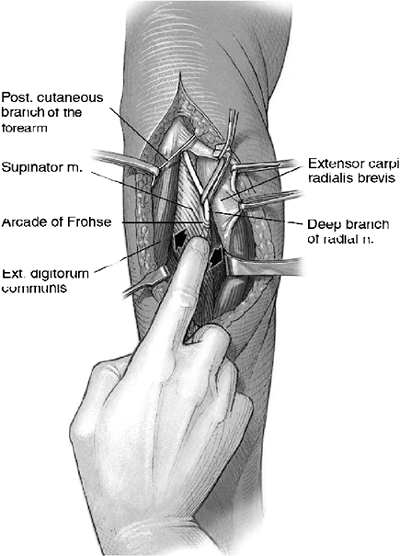
palpation. This is done by gently running the index finger over the
supinator in a radial-to-ulnar direction. The course of the
nerve is defined (Fig. 16-19).
It is followed to the proximal edge of the supinator, where the arcade
of Frohse is seen. The posterior interosseous nerve is in fat at this
level. At this region there are adjacent recurrent vessels; vessels
crossing the nerve are clamped and tied. The nerve can be exposed
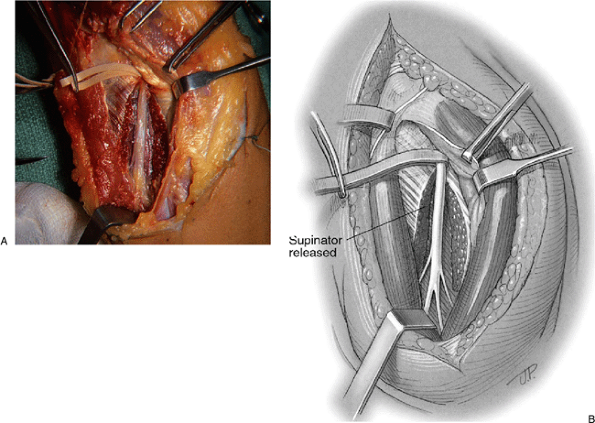
through its entire course through this approach. The arcade of Frohse
is released, as is the entire length of the supinator muscle to its
distal end (Fig. 16-20).
 |
|
Figure 16-18.
The plane between the extensor carpi radialis brevis and the extensor digitorum communis is entered and developed in a distal-to-proximal direction. |
position. For exposure of the posterior interosseous nerve, the arm is
draped free on an arm board and the forearm is supinated. A tourniquet
at 250 mm Hg is utilized.
the supinator muscle, a curvilinear incision or a chevron incision is
made; both of these come lateral to the antecubital crease,
then
down the anterior aspect of the proximal third of the forearm. The
lateral antebrachial cutaneous nerve and its branches will be found
just superficial to the antebrachial fascia at the elbow; rubber bands
are placed about it to maintain its identity and to retract a bit.
 |
|
Figure 16-19.
The posterior interosseous nerve is identified by palpation. Running the index finger over the supinator muscle in a radial to ulnar direction identifies the location and course of the nerve deep to the muscle. |
 |
|
Figure 16-20. The supinator muscle may be released, exposing the full course of the nerve (A,B).
|
the elbow in the interval between the brachioradialis and the
brachialis. A rubber band is placed about it (Fig. 16-21A).
The nerve is then traced distally to its major branches at the elbow
level. At the elbow level the posterior interosseous nerve and the
motor branches to the extensor carpi radialis brevis and the
superficial radial nerve are identified. There can be difficulty in
identifying the radial nerve proximally because on occasion there is a
fusion between the brachialis and the brachioradialis muscle. Another
key to this approach is to identify and protect the superficial branch
of the radial nerve, which is vulnerable to excessive traction. In this
instance the superficial radial nerve is identified in the fascia of
the brachioradialis on its posterior surface. It is traced proximally
to the main radial trunk and then the other two branches are identified
and the dissection is traced distally. Difficulty can occur if one does
not stay in the internervous plane or if there is partial or complete
fusion between the brachialis and brachioradialis. The appropriate
cleavage plane is identified by tracing the superficial radial nerve
proximally. There is an anterior and posterior leash of vessels about
the superficial radial nerve. These branches of the radial artery are
clamped and tied.
of the supinator muscle. There are numerous recurrent branches in this
area of the radial recurrent system. These are clamped and tied (Fig. 16-21B).
The posterior interosseous nerve is traced through the proximal half of
the supinator; a hemostat is passed deep to the arcade and the
supinator muscle, and superficial to the posterior interosseous nerve.
The arcade is cut by sharp dissection with scissors, and the supinator
muscle is split. It will be noted that there are numerous branches from
the posterior interosseous nerve to the supinator muscle (Fig. 16-21C).
With this approach good exposure of the posterior interosseous nerve in
this area is obtained. Once the nerve is freed and fully identified,
any compressive source such as a ganglion or tumor can be removed.
Similarly, if there has been a comminuted fracture of the radial head,
once the posterior interosseous nerve has been identified and
protected, the radial head can be removed from this anterior approach.
Likewise, in those patients with rheumatoid arthritis, a synovectomy
can be performed. It should be noted that the motor branch to the
extensor carpi radialis brevis is in the field and should be protected
with a rubber band as a gentle retractor.
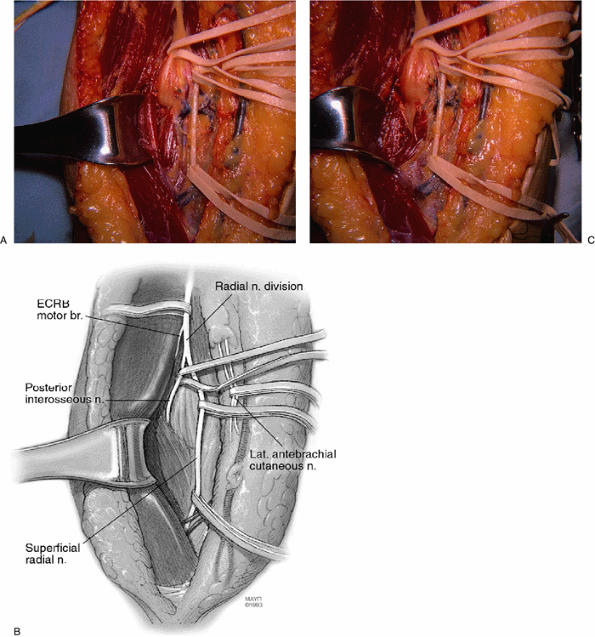 |
|
Figure 16-21.
In the anterior approach, the radial nerve is identified between the brachialis and brachioradialis muscles. The region of the arcade of Frohse is exposed (A). The rubber bands are about the lateral antebrachial cutaneous and superficial radial nerves. The posterior interosseous nerve and the motor branch to the extensor carpi radialis brevis are seen. Note the radial recurrent vessels. Some of the key crossing vessels are seen tied (B). The arcade of Frohse has been released. See how flattened the nerve is under the arcade (C). |
the elbow at 90 degrees, the forearm at midposition, and the wrist in
neutral. The plaster extends to the proximal interphalangeal (PIP)
joints and allows for stabilization of the MP joints in extension for a
short period. Postoperatively, active exercises of the PIP and distal
interphalangeal (DIP) joints are encouraged. A protective dynamic
splint can be utilized afterward in the recovery phase, if necessary.
The thumb is sometimes splinted in extension in a static splint at
night until there is restoration of full extension of the thumb, as
flexion contracture of the DIP joint can develop.
stimulation to the paralyzed muscles is utilized, and the paralysis
usually will subside or show improvement gradually over a 3-month
period. The last muscle to recover most commonly is the extensor
pollicis longus. When this nerve has been compressed or irritated
because of a discrete mechanism, decompression is virtually 100%
successful. Less predictable are those instances in which the
exploratory decompression was performed in association with symptoms of
lateral epicondylitis. In general, as the indications for the
exploration are less discrete, so also is the likelihood of realizing a
satisfactory result.
from failure to obtain adequate hemostasis. The tourniquet should be
released before closing the skin. It is conceivable to have a
Volkmann’s type process develop if good hemostasis has not been
achieved. This is an important concept, as hematomas cause more
inflammation. The fibrosis that can occur in this instance can
jeopardize the neural recovery.
or closure are a distinct possibility with posterior interosseous nerve
decompression, and great attention to detail is necessary to avoid this
complication.
postoperative splinting program is due to extension contractures of the
MP and interphalangeal joints. This is especially true if outriggers
are utilized and the outrigggers extend to the tips of the fingers. If
such splints are used, the loop should extend just under the proximal
phalanx, leaving the interphalangeal joints free. It is extension of
the MP joint that is lost in a posterior interosseous nerve para-lysis
in the digits. The patient should be allowed to flex his fingers a few
times a day with the outriggers completely removed.
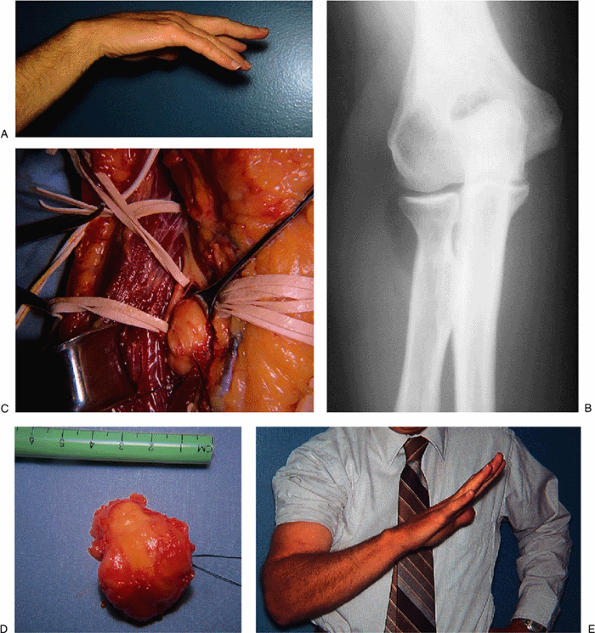
palsy presented with a “pseudo-ulnar claw” hand of several months’
duration (Fig. 16-22A). Radiographs revealed a lucency consistent with a lipoma present in this vicinity of the radial head (Fig. 16-22B).
Exploration was performed through an anterior approach and confirmed
compression of the posterior interosseous nerve at the arcade of Frohse
by a lipoma (Fig. 16-22C). The tumor was resected (Fig. 16-22D). The patient obtained complete recovery in 2 months (Fig. 16-22E).
 |
|
Figure 16-22. A:
The “pseudo-ulnar claw” hand representative of a partial posterior interosseous nerve paralysis. It is not a true claw hand seen with low ulnar nerve palsy because there is no hyperextension at the metacarpophalangeal joint of the ring and little fingers; rather, there is a drop in these digits at this joint. B: Radiographs revealed a soft-tissue lucency adjacent to the radial head typical of a lipoma. C: The lipoma was exposed through the anterior approach. Traction sutures were placed in it to facilitate its delivery of the tumor. The epineurium of the radial nerve was incised longitudinally. This allowed for more room to extricate the tumor safely. D: Specimen lipoma. E: Recovery occurred gradually over a 6-week period. |
M, Kaplan EB. The relationship of the ulnar nerve to the medial
intermuscular septum in the arm and its clinical significance. Hand 1976;8:238–242.
RJ, O’Driscoll SW, Jupiter JB, et al. Unrecognized dislocation of the
medial portion of the triceps: another cause of failed ulnar nerve
transposition. J Neurosurg 2000;92:52–57.
