Biopsy of Musculoskeletal Tumors
Editors: Tornetta, Paul; Einhorn, Thomas A.; Damron, Timothy A.
Title: Oncology and Basic Science, 7th Edition
Copyright ©2008 Lippincott Williams & Wilkins
> Table of Contents > Section I
– Evaluation and Management of Musculoskeletal Oncology Problems > 3
– Biopsy of Musculoskeletal Tumors
– Evaluation and Management of Musculoskeletal Oncology Problems > 3
– Biopsy of Musculoskeletal Tumors
3
Biopsy of Musculoskeletal Tumors
Edward A. Athanasian
Biopsy of musculoskeletal bone and soft tissue tumors
may appear to be a deceptively easy technical surgical procedure.
Patients may also perceive biopsy as a relatively straightforward,
low-risk procedure. While many aspects of the biopsy procedure require
only basic technical skills, the proper execution of biopsy of
extremity lesions requires careful preparation and planning, plus
exacting technique. Errors made at the time of biopsy may not become
apparent until the time of surgical resection or recurrence.
may appear to be a deceptively easy technical surgical procedure.
Patients may also perceive biopsy as a relatively straightforward,
low-risk procedure. While many aspects of the biopsy procedure require
only basic technical skills, the proper execution of biopsy of
extremity lesions requires careful preparation and planning, plus
exacting technique. Errors made at the time of biopsy may not become
apparent until the time of surgical resection or recurrence.
Risks of Improper Biopsy
Most extremity bone and soft tissue tumors are benign.
Fortunately, the risks of suboptimal biopsy when performed on benign
lesions are minimal. The risks of biopsy of malignant lesions are much
greater. Insufficient planning and poor execution have the potential to
complicate subsequent definitive treatment if the lesion under
consideration must be definitively treated surgically. Inadequate
biopsy has the potential to increase the risk to the patient’s life, as
demonstrated in studies performed by the American Musculoskeletal Tumor
Society. If the risks of biopsy are carefully considered, it becomes
very clear that fundamental understanding of planning and executing a
biopsy is imperative to maximize the ability to perform limb salvage
surgery and maximize patient survival.
Fortunately, the risks of suboptimal biopsy when performed on benign
lesions are minimal. The risks of biopsy of malignant lesions are much
greater. Insufficient planning and poor execution have the potential to
complicate subsequent definitive treatment if the lesion under
consideration must be definitively treated surgically. Inadequate
biopsy has the potential to increase the risk to the patient’s life, as
demonstrated in studies performed by the American Musculoskeletal Tumor
Society. If the risks of biopsy are carefully considered, it becomes
very clear that fundamental understanding of planning and executing a
biopsy is imperative to maximize the ability to perform limb salvage
surgery and maximize patient survival.
-
Improper incision placement or orientation
-
May compromise and complicate attempts at subsequent limb salvage
-
May result in need for performing amputation specifically as a result of the biopsy
-
In studies reported by the American
Musculoskeletal Tumor Society, amputation was required specifically
because of improper biopsy placement in as many as 18% of patients.
-
-
May result in need for more extensive amputation than otherwise might have been necessary
-
-
Improper excisional biopsy
-
Associated with greater risks than incision biopsy or needle biopsy
-
Specific anatomic regions such as the
axilla, antecubital fossa, carpal tunnel, groin, and popliteal fossa
are specifically at risk for contamination when excisional biopsy is
performed. -
Soft tissue contamination produced
following marginal excision of malignant lesions must be excised at the
definitive surgical excision.-
Extensive contamination increases the amount of soft tissue that needs to be resected.
-
Results in a greater need for soft tissue coverage to close wounds
-
-
Indications for Biopsy
Lesions That Do Not Require Biopsy
-
Many benign bone and soft tissue tumors
can be readily recognized clinically or radiographically and do not
require biopsy to establish a diagnosis (Table 3-1).
Lesions That Require Biopsy
-
Lesions that are not readily recognized
based on clinical examination or radiographic assessment should be
considered for biopsy. Even innocuous-appearing soft tissue lesions
that are painless and may have been present for a long period of time
without growth must be considered for biopsy if a differential
diagnosis limited to benign lesions only cannot be made. The rule of
thumb is that if the clinical and radiographic
diagnosis cannot be limited to benign lesions only, biopsy or referral
to a specialist is indicated. -
Soft tissue sarcomas frequently present
as painless masses that have been present for a long period of time
with recent change in size. Synovial sarcomas are notorious for this
sort of behavior.
Types of Biopsies
Box 3-1 lists the types of biopsies used for musculoskeletal tumors, some of which are discussed in detail below.
|
Table 3.1 Soft Tissue and Bone Lesions That Can Usually be Recognized Clinically or Radiographically
|
||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Box 3-1 Biopsy Types Employed for Musculoskeletal Tumors
-
Fine-needle aspiration/biopsy
-
Core-needle biopsy
-
Open incisional biopsy
-
Open excisional biopsy
-
Primary wide excision
Fine Needle Aspiration (FNA)/Biopsy (Skinny Needle)
Setting
-
In the office under local anesthetic
-
In the radiology suite
-
Under ultrasound guidance for soft tissue biopsy
-
Under computed tomography (CT) guidance for bone lesion biopsy
-
Technique
-
Soft tissue
-
Small-gauge needle on syringe used to aspirate cells during multiple passes in several directions via single entry site
-
Cells collect in syringe or hub of needle and are examined on glass slides, often immediately after biopsy performed
-
-
Bone
-
Fine-needle aspiration biopsy of bone tumors may require additional anesthetic, depending upon site and need to enter bone.
-
If no soft tissue extension of process from bone, may require larger-gauge needle or drill to enter bone
-
Usually done under CT guidance
-
P.39
General Principles
-
Provides cells (cytology) but no true pattern of organization
-
Site and pathway for the needle need to
be planned such that they can be incorporated into a standard limb
salvage incision in the event wide excision of the tumor is
subsequently required. -
Needle placement must not compromise
amputation flaps in the event amputation is chosen as the most
appropriate treatment for the lesion in question.
Advantages
-
Principal advantage is the rapidity with which lesional tissue can be obtained and the histologic diagnostic process started.
-
Less costly than a hospital-based procedure
-
Limited discomfort for the patient
-
Limited soft tissue contamination
-
Limited risk as long as the biopsy site, needle entry point, and needle course are carefully planned
Disadvantages
-
Limited material to examine means lower likelihood of achieving specific diagnosis.
-
Usually performed by radiologist without detailed knowledge of need for appropriate placement of needle tract
-
Negative biopsy does not reliably exclude neoplasm.
-
Negative result often requires repeat aspiration or biopsy by another technique.
Indications
-
Relatively inaccessible lesions of bone and soft tissue
-
Confirmation of strong clinical suspicion of metastatic disease or sarcoma recurrence
Results
-
Diagnostic accuracy rate 64% to 88% for musculoskeletal tumors
-
Diagnostic accuracy is lower for benign tumors.
Needle Biopsy (Core-Needle Biopsy)
Setting
-
Same as for fine-needle biopsy
Technique
-
Soft tissue
-
A special hollow needle with a cutting
mechanism retrieves a cylindrical core of tissue approximately 2 mm in
diameter and several millimeters in length, depending upon the specific
type of needle used. -
Typically two or three passes of the needle are made.
-
-
Bone
-
Core-needle biopsy of bone tumors usually requires a heavier anesthetic.
-
Performed with a hollow trephine-type needle with much larger diameter (4 to 7 mm)
-
Usually done under fluoroscopic guidance, often with frozen section analysis done to confirm the presence of lesional tissue
-
General Principles
-
Provides for evaluation of pattern of tissue organization and cellular features
-
Placement of needle should follow same principles as for skinny needle.
Advantages
-
Provides more tissue for evaluation than fine-needle aspiration, with additional component of the pattern of organization
-
Otherwise same advantages as for
fine-needle aspiration/biopsy (rapidity of diagnosis; limited cost,
discomfort, contamination, risk)
Disadvantages
-
Principal disadvantage pertains to the
limited size of the tissue sample obtained, which has the potential to
reduce accuracy and result in sampling error. -
Molecular diagnostic testing, which can
be particularly useful in difficult cases, may not be able to be done
if inadequate tissue has been obtained to allow initial processing to
prepare for this potential need.
Indications
-
Core-needle biopsy is most often
indicated for superficial or accessible deep extremity lesions that are
of sufficient size to allow needle placement (>3 cm) and that do not
involve major neurovascular structures.
Results
-
Diagnostic accuracy rate 83% to 93% for musculoskeletal tumors
-
Diagnostic accuracy is lower if the lesion is thought to be benign.
Open Biopsy (Incisional or Excisional)
Setting
-
Typically in the operating room under
general anesthetic with pathologist standing by for frozen section
analysis to confirm adequacy of tissue
Advantages
-
Principal advantage relates to the larger
size of the tissue sample obtained, reducing the risk of sampling error
and allowing for more extensive histologic assessment as well as
molecular diagnostic assessment when needed. -
Gold standard in achieving high diagnostic accuracy
Disadvantages
-
Principal disadvantage of open biopsy is potential for improper execution by surgeon.
-
Potential adverse consequences of tissue contamination/exposure during open biopsyP.40
-
Any tissue exposed or manipulated at the time of biopsy is potentially contaminated with tumor cells.
-
Any hematoma or seroma that develops has the potential to further contaminate local soft tissues.
-
Any tissue contaminated as the result of
biopsy of a malignant lesion should be carefully considered for
resection in continuity with the major tumor mass at that time of
definitive treatment if the lesion’s definitive treatment requires
surgery with widely negative margins. -
Need for greater soft tissue excision than would be required with needle biopsy
-
-
Potential adverse consequences of poorly placed open biopsy incision
-
If not placed in line with or immediately
parallel to a standard limb salvage incision, may result in need to
resect more soft tissue than might otherwise have been necessary had
the biopsy incision been ideally placed -
May result in need to sacrifice
functional tissues, including tendon, nerve, and vessels, in an effort
to achieve negative margins -
Commonly results in the need for soft tissue coverage in the form of skin graft, rotation flap, or free flap
-
-
Poorly performed open biopsy increases
the risk of positive margins at the time of definitive treatment and
increases risk of local recurrence.
Indications
-
Many bone and less accessible deep soft
tissue tumors are best approached with open biopsy (see details below
for incisional versus excisional biopsy).
Results
-
Accuracy rates of 96% for extremity bone
and soft tissue lesions following final analysis have been reported at
major cancer centers.
Box 3-2 Three Most Important Principles of Biopsy Incision Orientation
-
Longitudinal, not transverse, incisions
-
Longitudinal, not transverse, incisions
-
Longitudinal, not transverse, incisions
Biopsy Technique
Biopsy Incision Placement
-
Biopsy incisions must be carefully planned and placed.
-
As a general rule, biopsy incisions for extremity lesions should be longitudinal (Box 3-2 and Figs. 3-1 and 3-2)
-
Incisions that incorporate limited
oblique segments can be used at major flexion creases such as the
popliteal fossa, antecubital fossa, volar wrist flexion crease, and
palmar surface of the hand.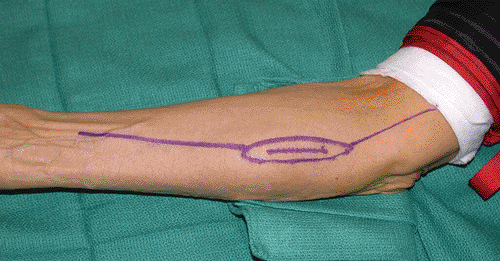 Figure 3-1
Figure 3-1
Longitudinal biopsy incision is drawn over the medial proximal forearm.
The potential limb salvage incision is drawn at the time of the biopsy
to ensure that it can be incorporated into a future planned wide
excision if this is subsequently needed. -
Transverse incisions and palmar oblique Z-Brunner incisions should specifically be avoided due to the extent of soft tissue contamination these will produce.
-
Oblique incisions may be used over the clavicle and ilium in line with planned line of resection of those bones.
-
-
Planning for biopsy incision placement requires a basic knowledge of limb salvage incisions and procedures.
-
Biopsy incisions should be placed in line
with or immediately parallel to the limb salvage incision that would be
required at definitive treatment. -
If the treating physician is not familiar
with the appropriate limb salvage incision or amputation flaps,
consideration should be given to referral to a musculoskeletal tumor
specialist.
-
-
Common incisions for bone tumor biopsy are demonstrated in Figure 3-3.
-
Incision placement for soft tissue
tumors is more dependent on the primary location of the tumor, the most
direct route to the tumor, which will limit contamination to a single
plane or compartment, and proximity of major neurovascular structures. -
Areas at high risk for contamination are depicted in Figure 3-4.
Incisional Biopsy
Incisional biopsy refers to the procedure where the
biopsy is performed by cutting directly into the tumor tissue and
removing a wedge of the tumor for analysis while leaving the majority
of the tumor intact and untouched. This is a relatively safe and
accurate form of biopsy; it carries relatively low risk when carefully
planned and properly executed. Any tissue that is touched or
manipulated at the time of biopsy will be contaminated with tumor
tissue and may have to be excised at the time of definitive tumor
treatment if the lesion is malignant. The lesion is approached by the
most direct means while limiting contamination to a planned region and
avoiding exposure and contamination of major neurovascular structures.
biopsy is performed by cutting directly into the tumor tissue and
removing a wedge of the tumor for analysis while leaving the majority
of the tumor intact and untouched. This is a relatively safe and
accurate form of biopsy; it carries relatively low risk when carefully
planned and properly executed. Any tissue that is touched or
manipulated at the time of biopsy will be contaminated with tumor
tissue and may have to be excised at the time of definitive tumor
treatment if the lesion is malignant. The lesion is approached by the
most direct means while limiting contamination to a planned region and
avoiding exposure and contamination of major neurovascular structures.
P.41
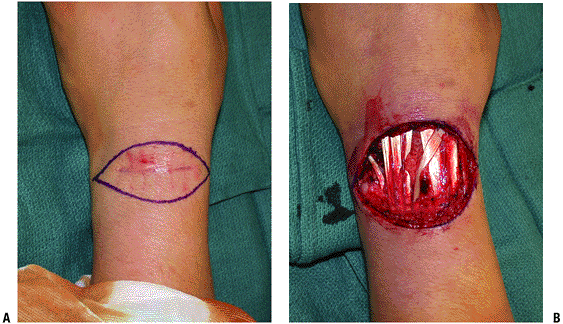 |
|
Figure 3-2 (A)
A malignant fibrous histiocytoma was approached with marginal excision biopsy prior to referral. Note the suture marks, which were placed far from the incision. (B) Wide excision of the tumor bed and all contaminated tissue from the biopsy results in a large soft tissue defect, which may commit the patient to a soft tissue coverage procedure that might not have been otherwise needed. |
Indications
-
Most commonly done for lesions >3 to 4 cm in maximum dimension
-
In certain situations, incisional biopsy may be the best approach for even smaller lesions, perhaps 1 to 2 cm.
-
Distal aspect of the arm or leg
-
Lesion in close proximity to a major neurovascular bundle
-
Lesion arises in a high-risk region such
as the axilla, antecubital fossa, carpal tunnel, groin, popliteal
fossa, medial malleolus, or plantar aspect of the foot.-
Alternative biopsy type in these
high-risk regions is primary wide excision (see below), which may also
carry significant morbidity due to the associated requirement for
sacrifice of additional soft tissue to act as margin at the time of
biopsy. The determination of which type of biopsy is best in a
high-risk region may be best done by a musculoskeletal oncologist.
-
-
-
Importance of frozen section during incisional biopsy
-
Main purpose is to confirm presence of
diagnostic tissue, not necessarily to establish definitive diagnosis on
which to base treatment. -
Potential for inaccuracy of frozen section analysis may lead to inappropriate initial surgical procedure.
-
No potential during frozen section for immunohistochemical or molecular analysis
-
Crucial for potentially malignant lesions
-
Less important for benign conditions
-
-
A second-stage procedure may be required
for definitive treatment after initial impression under frozen section
is confirmed on further review of permanently fixed tissue.-
Particularly true for suspected primary malignancy
-
-
Excisional Biopsy
Excisional biopsy refers to the procedure where the
entire lesion is excised in one piece while dissecting at the margin
through the reactive zone surrounding the lesion, accomplishing a
“marginal” excision. This type of biopsy is usually best done for small
soft tissue lesions that appear to be benign, such as lipomas.
entire lesion is excised in one piece while dissecting at the margin
through the reactive zone surrounding the lesion, accomplishing a
“marginal” excision. This type of biopsy is usually best done for small
soft tissue lesions that appear to be benign, such as lipomas.
Advantage
-
Lesion is removed in one stage and the
full lesion is available for pathologic analysis, thereby maximizing
accuracy and eliminating the potential for sampling error.
Disadvantages
-
Associated with significant risks, particularly when done for malignant lesions
-
Produces extensive contamination of all tissues exposed, retracted, or manipulated at the time of the procedure
-
If done for a soft tissue sarcoma,
residual microscopic disease will be left in the patient that will
require subsequent wide excision to minimize local recurrence risk. -
When done for large lesions, large
amounts of otherwise uninvolved tissues will need to be sacrificed to
achieve negative margins. Additional need to sacrifice uninvolved soft
tissues is greater than would be required in the setting of needle or
incision biopsy. -
If major nerves or vessels are exposed at
biopsy, definitive treatment may require excision of those vital
structures, thereby increasing the morbidity of definitive treatment. -
Suboptimal placement of biopsy incision
will increase the need for soft tissue coverage at definitive treatment
and might even preclude limb salvage procedures. -
The larger the size of the lesion and the
more distal in the extremity the tumor arises, the greater the risks
associated with excisional biopsy.
-
Indications
-
Best reserved for small lesions (<3 to 4 cm) arising within muscle of the proximal portions of the extremities.
P.42
P.43
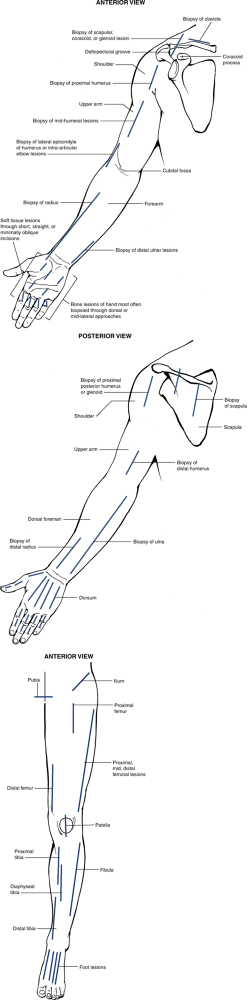 |
|
Figure 3-3 Common incisions for bone tumor biopsy in the upper right (A and B) and lower left (C) extremities.
|
P.44
Drain Usage and Placement
-
Meticulous hemostasis should be obtained prior to closure.
-
The drain should not be a substitute for meticulous hemostasis.
-
If a drain is used, it should exit the patient immediately in line with the more distal end of the incision.
-
Lateral drain placement or placement at a
distance from the incision increases the need for soft tissue sacrifice
at the time of definitive treatment.
Primary Wide Excision
Primary wide excision refers to the procedure where the
tumor is definitively excised with a surrounding cuff or margin of
healthy, uninvolved, nonreactive tissue. This procedure should be
considered as a definitive cancer operation. This type of biopsy is
performed when the suspicion for malignancy is high and the risks of
contamination of biopsy outweigh the morbidity of the additional soft
tissue sacrifice that is required to achieve the desired negative
margins upon final analysis of the resected specimen. This type of
biopsy might be used for lesions arising in the axilla, antecubital
fossa, carpal tunnel, groin, popliteal fossa, or plantar aspect of the
foot. The decision of when to perform primary wide excision is complex
and requires considerable knowledge regarding the differential
diagnosis of the lesion and the morbidity of biopsy for the specific
anatomic region. The decision of when to perform this biopsy is often
best determined by an experienced musculoskeletal oncologist.
tumor is definitively excised with a surrounding cuff or margin of
healthy, uninvolved, nonreactive tissue. This procedure should be
considered as a definitive cancer operation. This type of biopsy is
performed when the suspicion for malignancy is high and the risks of
contamination of biopsy outweigh the morbidity of the additional soft
tissue sacrifice that is required to achieve the desired negative
margins upon final analysis of the resected specimen. This type of
biopsy might be used for lesions arising in the axilla, antecubital
fossa, carpal tunnel, groin, popliteal fossa, or plantar aspect of the
foot. The decision of when to perform primary wide excision is complex
and requires considerable knowledge regarding the differential
diagnosis of the lesion and the morbidity of biopsy for the specific
anatomic region. The decision of when to perform this biopsy is often
best determined by an experienced musculoskeletal oncologist.
Unique Considerations for Biopsy
Nerve Tumors (Box 3-3)
The presence of the peripheral nerve sheath tumor may be
suspected clinically based on differential mobility, as nerve lesions
tend to be more mobile in the medial lateral plane than the proximal
distal plane (being tethered by their neural attachment). Confirmation
of the presence of the lesion arising in a major nerve requires a
unique preparation and technical approach. There are greater risks for
permanent nerve injury following the biopsy of nerve-related lesions.
Marginal excision of schwannoma has been associated with a permanent
risk of injury of 4%, while the risk of injury following excision of
neurofibroma is significantly higher. The risk of injury following
biopsy of malignant nerve lesions has not been well characterized and
is superseded by the need to widely excise the lesion and nerve in an
effort to achieve local control of the tumor. It may be impossible to
determine whether a specific nerve lesion is benign or malignant based
on preoperative testing, including magnetic resonance imaging. Lesions
are often best approached through a direct approach to the nerve with
minimal dissection of the surrounding tissues. Dissection is done under
magnification when necessary while mobilizing healthy fascicles to
accomplish a marginal excision. If the lesion is not readily dissected
from the normal nerve fascicles, an incisional biopsy should be
performed, with subsequent treatment determined by frozen section or
permanent analysis.
suspected clinically based on differential mobility, as nerve lesions
tend to be more mobile in the medial lateral plane than the proximal
distal plane (being tethered by their neural attachment). Confirmation
of the presence of the lesion arising in a major nerve requires a
unique preparation and technical approach. There are greater risks for
permanent nerve injury following the biopsy of nerve-related lesions.
Marginal excision of schwannoma has been associated with a permanent
risk of injury of 4%, while the risk of injury following excision of
neurofibroma is significantly higher. The risk of injury following
biopsy of malignant nerve lesions has not been well characterized and
is superseded by the need to widely excise the lesion and nerve in an
effort to achieve local control of the tumor. It may be impossible to
determine whether a specific nerve lesion is benign or malignant based
on preoperative testing, including magnetic resonance imaging. Lesions
are often best approached through a direct approach to the nerve with
minimal dissection of the surrounding tissues. Dissection is done under
magnification when necessary while mobilizing healthy fascicles to
accomplish a marginal excision. If the lesion is not readily dissected
from the normal nerve fascicles, an incisional biopsy should be
performed, with subsequent treatment determined by frozen section or
permanent analysis.
P.45
P.46
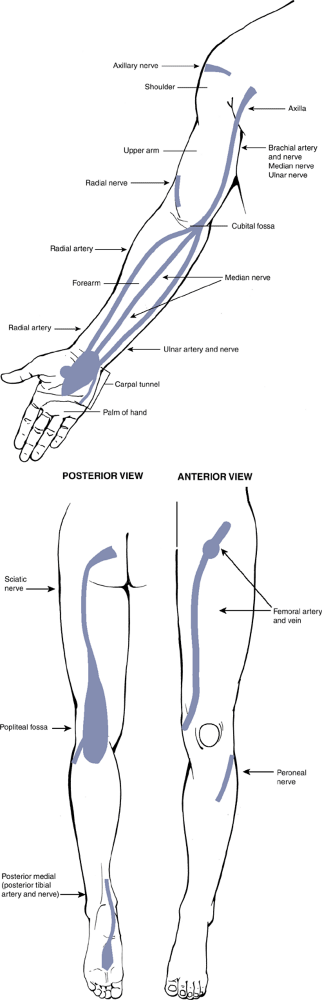 |
|
Figure 3-4 The areas shown in blue are at high risk for contamination during biopsy in the upper right (A) and lower left (B) extremities.
|
P.47
Box 3-3 Tumors Arising in Major Peripheral Nerves
-
Benign
-
Schwannoma (neurilemoma)
-
Neurofibroma
-
Malignant
-
Malignant peripheral nerve sheath tumor
Scapular Lesions with Anterior Extension
Scapular lesions with anterior extension present unique
risks for biopsy due to the proximity of the brachial plexus. The
approach for biopsy is dependent upon the extent of soft tissue
involvement and the primary location within the scapula.
risks for biopsy due to the proximity of the brachial plexus. The
approach for biopsy is dependent upon the extent of soft tissue
involvement and the primary location within the scapula.
-
Scapular body lesions: Best approached
through a posterior incision, particularly if scapular resection might
ultimately be required -
Lesions in the region of the glenoid neck
or base of the coracoid: Can be approached anteriorly by dissecting
along the superior aspect of the coracoid while meticulously
maintaining hemostasis to reduce the risks of soft tissue
contamination. The biopsy site can subsequently be incorporated into an
extended deltopectoral approach, which can readily be extended
posteriorly if needed.
Proximal Humerus Lesions
Most proximal humerus lesions can be biopsied through an
incision placed 1 to 2 cm lateral to the deltopectoral groove. If the
incision is extended distally beyond 5 cm from the acromion, the
terminal portion of the axillary nerve will be transected or injured.
This will result in denervation of only the most medial portion of the
deltoid without significant functional impairment. Placement of the
incision at this location reduces the risk of spread of hematoma
medially along the course of the cephalic vein and pectoralis major and
reduces the risk of contamination of the major neurovascular structures.
incision placed 1 to 2 cm lateral to the deltopectoral groove. If the
incision is extended distally beyond 5 cm from the acromion, the
terminal portion of the axillary nerve will be transected or injured.
This will result in denervation of only the most medial portion of the
deltoid without significant functional impairment. Placement of the
incision at this location reduces the risk of spread of hematoma
medially along the course of the cephalic vein and pectoralis major and
reduces the risk of contamination of the major neurovascular structures.
Intra-articular Lesions of the Elbow
Biopsy of lesions arising in or near the elbow is
particularly difficult. There are limited windows for biopsy that allow
access to the joint without contaminating major nerves. Posterior
biopsy carries little risk but might impair attempts at limb salvage if
anterior exposure is required to resect the lesion. There is a large
window of potential exposure near the lateral epicondyle, but this
approach might also impair limb salvage if mobilization of the median
and ulnar nerves is required at definitive treatment. At times,
separate biopsy and limb salvage incisions might be required where the
biopsy incision and contaminated tissues from biopsy are dissected
separately from the limb salvage incision and left attached to the
major tumor mass while the limb salvage approach is performed through a
separate incision on the opposite side of the elbow. Lesions in this
region require detailed planning and often unique approaches.
particularly difficult. There are limited windows for biopsy that allow
access to the joint without contaminating major nerves. Posterior
biopsy carries little risk but might impair attempts at limb salvage if
anterior exposure is required to resect the lesion. There is a large
window of potential exposure near the lateral epicondyle, but this
approach might also impair limb salvage if mobilization of the median
and ulnar nerves is required at definitive treatment. At times,
separate biopsy and limb salvage incisions might be required where the
biopsy incision and contaminated tissues from biopsy are dissected
separately from the limb salvage incision and left attached to the
major tumor mass while the limb salvage approach is performed through a
separate incision on the opposite side of the elbow. Lesions in this
region require detailed planning and often unique approaches.
Distal Radius Lesions
The approach to the distal radius is complicated by the
compact arrangement of functional tissues surrounding this structure at
the level of the distal forearm. The optimal site for biopsy placement
is often determined by the planned definitive treatment or limb salvage
incision. For example, lesions requiring wide excision of the distal
radius and carpus are best approached through a dorsal biopsy incision.
The window for biopsy at the level of Lister’s tubercle is 4 mm between
the third and fourth dorsal compartments. There is a substantial risk
of contamination of the extensor tendons at biopsy. Contamination of
these structures will require their subsequent excision in the event of
a malignant diagnosis. When intralesional treatment of a distal radius
tumor such as giant cell tumor of bone is anticipated, the volar radial
approach between the radial artery and the first dorsal compartment
allows excision of any volar soft tissue extension and excellent
exposure of the distal radius. This approach, however, is not easily
extended distally should additional exposure be required (Fig. 3-5).
compact arrangement of functional tissues surrounding this structure at
the level of the distal forearm. The optimal site for biopsy placement
is often determined by the planned definitive treatment or limb salvage
incision. For example, lesions requiring wide excision of the distal
radius and carpus are best approached through a dorsal biopsy incision.
The window for biopsy at the level of Lister’s tubercle is 4 mm between
the third and fourth dorsal compartments. There is a substantial risk
of contamination of the extensor tendons at biopsy. Contamination of
these structures will require their subsequent excision in the event of
a malignant diagnosis. When intralesional treatment of a distal radius
tumor such as giant cell tumor of bone is anticipated, the volar radial
approach between the radial artery and the first dorsal compartment
allows excision of any volar soft tissue extension and excellent
exposure of the distal radius. This approach, however, is not easily
extended distally should additional exposure be required (Fig. 3-5).
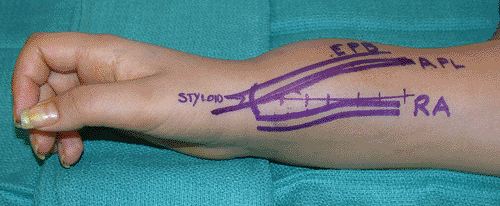 |
|
Figure 3-5 Biopsy incision for distal radius lesions with volar soft tissue extension.
|
Carpal Tunnel, Axilla, and Popliteal Fossa Lesions
Soft tissue contamination following excision biopsy or
marginal excision of malignant lesions in the carpal tunnel can produce
contamination that eliminates the possibility of limb salvage and
commits the patient to amputation of the hand. Even small lesions are
often best approached with either incisional biopsy or primary wide
excision. The risks of contamination of major nerves and blood vessels
are similar in the axilla and popliteal fossa, as well as other
extracompartmental sites.
marginal excision of malignant lesions in the carpal tunnel can produce
contamination that eliminates the possibility of limb salvage and
commits the patient to amputation of the hand. Even small lesions are
often best approached with either incisional biopsy or primary wide
excision. The risks of contamination of major nerves and blood vessels
are similar in the axilla and popliteal fossa, as well as other
extracompartmental sites.
Biopsy Planning and Procedure
Biopsy should be performed as the final step in the
analysis of a specific patient and lesion after complete history,
physical examination, laboratory and radiographic assessment have been
performed.
analysis of a specific patient and lesion after complete history,
physical examination, laboratory and radiographic assessment have been
performed.
Communication with the Pathologist
-
A differential diagnosis should be formulated in advance of the procedure and communicated to the pathologist.
-
Review of imaging studies will often assist the pathologist in generating a differential diagnosis.
-
Need for frozen section should be established in advance of the case.
Tourniquet Usage
-
When possible, the use of a tourniquet will minimize blood loss and assist in visualization at the time of biopsy.
-
Excellent hemostasis should be achieved
following release of the tourniquet prior to definitive closure to
limit the potential for hematoma and its attendant risks. -
Exsanguination of the limb with an
elastic bandage should be avoided owing to the risk of dislodging tumor
cells and causing venous embolization; gravity exsanguination is
preferred.
Planning the Incision
-
A final exercise that confirms appropriate preparation for biopsy is to draw the limb salvage incision or anticipated amputation flaps at the time of the biopsy prior to drawing the biopsy incision.
-
The biopsy incision should be placed in line with a portion of the limb salvage incision or away from the amputation flaps.
-
This approach minimizes the risks of
improper biopsy incision placement and assists in the attempt to
maximize limb salvage options in the event of a malignant final
diagnosis.
Dissection Technique
-
Following incision, dissection to the biopsy should be made using the most direct approach.
-
Dissection should be through tissues as opposed to usual surgical dissection, which is between tissue planes.
-
The dissection technique used should minimize the extent of contamination caused by the biopsy and limit the spread of hematoma.
-
All tissues contaminated at the time of
biopsy will need to be excised at the time of definitive excision if
the lesion is malignant on final analysis.
-
-
Biopsy is usually best performed at the periphery of the lesion.
-
Lesion is directly entered and a wedge of tissue excised.
-
Histologic Tissue and Culture Processing
-
If there is sufficient tissue available,
frozen section analysis is performed to determine a preliminary
diagnosis and to confirm the presence of lesional tissue. -
Additional tissue is sent for permanent analysis (1 cc minimum).
-
Tissue cultures are also taken and sent to the laboratory for analysis (“culture all tumors, biopsy all infections”).
Hemostasis
-
In most instances the tourniquet should be released prior to closure.
-
Means to achieve hemostasis:
electrocautery, gelatin sponge, methyl-methacrylate cement for bone
windows, or direct closure of the pseudocapsule or periosteum
Drain Usage and Placement
-
If a drain is required, it is placed with an exit point directly in line with the incision, usually at the distal aspect.
Wound Closure
-
Wound is closed in layers, with attention to suture placement.
-
Sutures should be placed close to the
wound margin to limit contamination and the extent of skin excision
that will be required at definitive treatment.
Instrument Handling
-
It is useful to use an instrument basin for contaminated instruments during the procedure.
-
Used operative instruments are placed in the basin with contaminated ends pointing in one direction.
-
Operative personnel limit the handling of instruments to the noncontaminated end.
-
If concurrent surgery is to be performed,
operative clothing and instruments should be changed for use in a
separately draped field to reduce the risk of cross-contamination.
Postoperative Care
If bone biopsy has been performed, measures should be taken to protect the limb from intraoperative and postoperative fracture.
Pathologic Assessment and Limitations
Most patients consider pathologic assessment to be a
definitive “black-or-white” process. Patients should be specifically
counseled prior to the biopsy procedure regarding accuracy rates for
frozen section analysis and permanent analysis of biopsy tissue.
definitive “black-or-white” process. Patients should be specifically
counseled prior to the biopsy procedure regarding accuracy rates for
frozen section analysis and permanent analysis of biopsy tissue.
-
Accuracy of frozen section analysis for
bone and soft tissue tumors is approximately 80%. If definitive
treatment is being considered based on frozen section analysis, the
surgeon must incorporate the possibility of an incorrect diagnosis into
the treatment plan. The potential for inaccuracy should be discussed
with the patient in advance. -
When is it reasonable to proceed with definitive treatment based upon frozen section?
-
When is it not reasonable to proceed based upon frozen section?
-
When malignancy is a major consideration
in the differential diagnosis, it is often best to defer treatment
until the final analysis is completed. The accuracy of a final
pathologic analysis at a tumor center is approximately 96%.
Preoperative discussion with the pathologist ensures the availability
of appropriate diagnostic testing, which can aid in maximizing the
effort to establish an accurate diagnosis and maximize patient care.
-
Suggested Reading
Domanski
HA, Akerman M, Carlen B, et al. Core-needle biopsy performed by the
cytopathologist: a technique to complement fine-needle aspiration of
soft tissue and bone lesions. Cancer 2005;105(4):229–239.
HA, Akerman M, Carlen B, et al. Core-needle biopsy performed by the
cytopathologist: a technique to complement fine-needle aspiration of
soft tissue and bone lesions. Cancer 2005;105(4):229–239.
Donner TR, Voorhies RM, Kline DG. Neural sheath tumors of major nerves. J Neurosurg 1994;81:362–373.
Heslin MJ, Lewis JJ, Woodruff JM, et al. Core needle biopsy for diagnosis of extremity soft tissue sarcoma. Ann Surg Oncol 1997;4:425–431.
Mankin HJ, Lange TA, Spanier SS. The hazards of biopsy in patients with malignant primary bone and soft tissue tumors. J Bone Joint Surg [Am] 1982;64:1121–1127.
Ogilvie CM, Torbert JT, Finstein JL, et al. Clinical utility of percutaneous biopsies of musculoskeletal tumors. Clin Orthop Relat Res 2006;450:95–100.
Scarborough MT. The biopsy. AAOS Instr Course Lect 2004;53:639–644.
Simon MA. Biopsy of musculoskeletal tumors. J Bone Joint Surg 1982;64:1253–1257.
Skrzynski
MC, Biernann JS, Montag A, et al. Diagnostic accuracy and charge
savings of outpatient core needle biopsy compared with open biopsy of
musculoskeletal tumors. J Bone Joint Surg [Am] 1996;78:644–649.
MC, Biernann JS, Montag A, et al. Diagnostic accuracy and charge
savings of outpatient core needle biopsy compared with open biopsy of
musculoskeletal tumors. J Bone Joint Surg [Am] 1996;78:644–649.
Trigg SD. Biopsy of hand, wrist, and forearm tumors. Hand Clin 2004;20(2):v, 131–135.
Yang
YJ, Damron TA. Comparison of needle core biopsy and fine-needle
aspiration for diagnostic accuracy in musculoskeletal lesions. Arch Pathol Lab Med 2004;128(7):759–764.
YJ, Damron TA. Comparison of needle core biopsy and fine-needle
aspiration for diagnostic accuracy in musculoskeletal lesions. Arch Pathol Lab Med 2004;128(7):759–764.
