BONE AND JOINT INFECTIONS IN CHILDREN
similar in their clinical presentation, diagnosis, and treatment. Both
are usually secondary to bacteremia, but they may occur simultaneously,
or the septic arthritis may be secondary to the osteomyelitis. When the
two are present, the problems of diagnosis, treatment, and sequelae are
greater than when either occurs alone. Although there are similarities
between them, subtle differences occur in the physical findings,
pathology, treatment, and prognosis. These differences make it
important that they be viewed as separate problems.
groups but is more common in children than in adults. The peak age for
occurrence is 18 to 24 months, and the mean age about 6 years. Since
the development of antibiotics, prognosis and results of treatment have
dramatically improved so that acute hematogenous osteomyelitis should
no longer be a life-threatening disease. Sequelae from the infection,
if diagnosed early and treated appropriately, are minimal and chronic
osteomyelitis is now rare. In older children, osteomyelitis is usually
monostotic, but in the neonate, it is not uncommon to find multiple
sites of infection.
infection that begins in the metaphysis of long bones and in the
metaphyseal equivalent areas of the remainder of the skeleton. In 1911,
Koch (25) demonstrated that the intravenous
injection of bacteria frequently caused infection that began in the
metaphyses of long bones near the physis. The metaphyseal location of
infection is related to the vascular anatomy in the region, where the
terminal nutrient vessels enter large sinusoidal vessels. The blood
flow velocity in these vessels is slow, which encourages the bacteria
to settle. In addition, the host defense mechanism is compromised
because of the paucity of monocytic cells in the sinusoidal spaces.
demonstrated in the rabbit model that the inflammatory response and
bone destruction are slightly more distal to the physis than the area
where the bacteria are found. It is in the area of this inflammatory
response that the destruction of the bone trabeculae occurs as a
response to the inflammatory process. By injuring the metaphyseal area
of the rabbit’s extremity just before intravenous injection of a bolus
of bacteria, Morrissy found that the development of osteomyelitis was
much more predictable than when there was no preceding injury. This
correlates with the clinical finding that there is frequently a history
of injury before the onset of osteomyelitis. Trauma may cause
thrombosis of the sinusoidal vessels, and the thrombus serves as a
culture medium for the bacterial growth.
cellular influx that will evolve into an abscess if the response is not
altered by appropriate treatment. The purulent exudate spreads through
the thin porous cortex of the metaphysis and elevates the periosteum,
which causes subperiosteal reactive bone formation.
is destroyed. Loss of the periosteum severely interferes with the
healing of a fracture should one occur in the course of the disease.
Before the development of antibiotics, the exudate, if it was not
surgically released, would eventually drain through the skin, as
described by Smith in 1874 (39). The elevation
of the periosteum interrupts the periosteal blood supply to the cortex.
Loss of the periosteal blood supply, along with vascular thrombosis
caused by the pus passing through the haversian system, renders the
cortex ischemic. The ischemic bone becomes a cortical sequestrum.
a cortical sequestrum is produced. The reason for this is not clear,
but it may be that the increased porosity of the metaphyseal cortex in
the young child allows the exudate to exit through the metaphyseal
cortex and decompresses the bone before it is forced through the
haversian system of the diaphysis. Where the metaphysis of the long
bone is partially intracapsular, as in the proximal femur, humerus, and
ankle, secondary septic arthritis can result from the spontaneous
decompression of the metaphysis.
demonstrated that metaphyseal vessels transverse the proximal femoral
physis. These vessels passing from the metaphysis to the epiphysis
apparently allow bacteria and the inflammatory process to cross the
physis into the epiphysis. In this age group, the physis and the
epiphysis can be severely damaged from the infection (Fig. 176.1). The vessels that cross the physis in the neonate progressively disappear beginning at 8 months of age (42).
By 18 months, the epiphyseal and metaphyseal circulations are
completely separate. Once the physis becomes a barrier to the
metaphyseal vessels, inflammatory destruction of the physis and
epiphysis is rare.
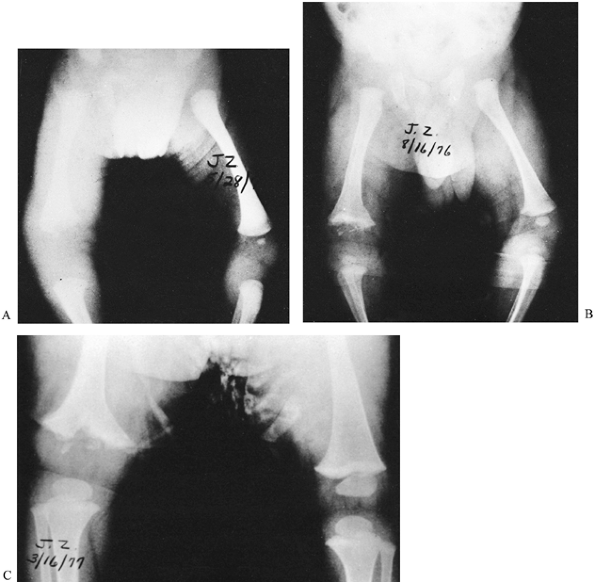 |
|
Figure 176.1. A: Two-month-old infant with osteomyelitis in the right femur. Note the soft-tissue swelling. B: Three months later, there is widening of the metaphysis with cupping as well as resorption of the ossified epiphysis. C: Seven months later, the femur was already shorter than the opposite femur.
|
severity of the infection, its location, and the age of the patient.
Approximately 30% of the patients with osteomyelitis are not ill and
have few systemic symptoms. In patients who are not ill, the initial
diagnosis is frequently in error, with the most common misdiagnosis
being some type of malignancy. Except for an elevated sedimentation
rate, which is almost always present, the laboratory data may be normal
in this subgroup of patients.
constant symptom is localized pain that causes the child to limp or not
use the extremity. Swelling is usually present, and its extent reflects
the severity of the infection. Local tenderness is always present. The
more superficial the bone, the more easily this can be demonstrated.
Why 30% of patients have a mild presentation of osteomyelitis is not
clear. In some instances, it reflects the use of antibiotics to treat
fever of undetermined etiology or other infections in other areas of
the body. It may also reflect the prevalence of the use of antibiotics
in the production of meat for consumption.
signs and symptoms are an elevated temperature in an anorexic,
irritable child with significant pain and limitation of function of an
extremity. Swelling, tenderness, and sometimes redness are common and
impressive. In the infant, swelling may extend throughout the entire
segment of the extremity (Fig. 176.2). Motion
in the adjacent joint is limited and causes protective muscle spasm.
Infants may present with pseudoparalysis, and older children will limp
or refuse to walk if the lower extremity is affected. Pain with
localized tenderness and swelling in the metaphysis of a long bone is
sufficient to make a presumptive diagnosis of osteomyelitis in an ill
child.
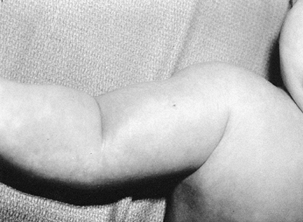 |
|
Figure 176.2. Osteomyelitis of the humerus. Swelling extends both proximal and distal to the humerus.
|
with a shift to the left. If infection has been present for several
days, the red blood cell count, hemoglobin, and hematocrit will be
lower than normal. Sedimentation rate is almost always elevated. Blood
cultures should always be taken when osteomyelitis is suspected; a
positive culture is usually found in 60% to 65% of patients.
the bone but in the deep soft tissues, where swelling is the initial
radiographic sign (Fig. 176.3). The swelling
occurs within the first 2 days of symptoms and obliterates the lucent
muscle planes progressively from the bone to the subcutaneous tissue.
With this pattern of soft-tissue swelling, the diagnosis of
osteomyelitis must be considered. When osteomyelitis is suspected, take
radiographs with the appropriate exposure and anatomic position. It is
important that both extremities have radiographs in the identical
position for comparison. Ask the radiographic technician to use
soft-tissue technique for the first radiograph
and
bone technique for a second. After 10 days of uncontrolled infection,
radiolucent areas can be seen in the metaphysis. New periosteal bone is
another late radiographic manifestation of osteomyelitis. This
periosteal bone is a result of the elevation of the periosteum by the
exudate that has passed through the cortex.
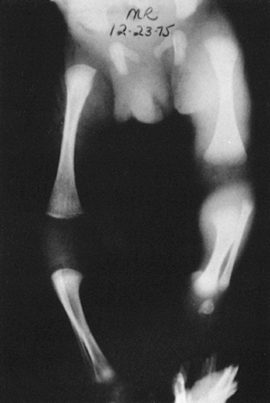 |
|
Figure 176.3. Soft-tissue swelling of the left thigh in a neonate with osteomyelitis.
|
possible early osteomyelitis includes the option of imaging with
radionuclides. An increase in uptake may be present in the area of
infection as early as 24 hours after onset, but an increase in uptake
is not diagnostic of osteomyelitis; it only reflects increased blood
flow and osteoblastic activity, which may also occur in the presence of
tumors and trauma. The value of the radionuclear studies is in
localization of the area of infection and in identifying multiple sites
of infection. The greatest help from a bone scan is in a patient who
has been partially treated; when the clinical signs are poorly defined,
as in infection of the pelvis or spine; and in managing a patient with
subacute osteomyelitis. False-negative bone scans and gallium scans do
occur (1). This is particularly true in
osteomyelitis of the pelvis and in the neonate. It is probably related
to the decreased blood flow from thrombosis. In the neonate, the bone
scan is accurate in only 30% to 40% of patients (2).
However, if both technetium and gallium scans are positive, the
diagnosis is almost always infection. This is particularly helpful in
patients with suspected osteomyelitis in the pelvis and will facilitate
an early diagnosis (12). Because gallium has more radioactivity than technetium, it should not be used except in very difficult cases (5).
Computed tomography (CT) scan and magnetic resonance imaging (MRI) can
be of benefit when the clinical facts are not sufficient to make the
diagnosis. Mazur et al. (27) reported a
sensitivity of 97% and a specificity of 92% for MRI. However, these
imaging modalities are seldom needed and should be used only in
difficult cases. Their routine use is not justified. A particularly
valuable area for the use of CT is in osteomyelitis of the pelvis, in
which early swelling can be identified as well as very early bone
changes that are seldom seen on routine radiographs made early in the
course of the infection.
made from the history, physical examination, and laboratory studies,
and is confirmed by radiographs, bone scan, MRI if needed, and
aspiration of the bone. Although 30% of the patients are not acutely
ill, the remainder are ill and give a history of the fairly rapid onset
of pain accompanied by swelling, localized tenderness, and impaired
function such as a limp, refusal to walk, or pseudoparalysis. In
those
who are ill, the temperature is elevated, and the young child is
irritable and anorexic. An exception is in the neonate who may not show
an elevated temperature and who may have minimal laboratory changes.
Neonates frequently present with failure to thrive. The neonate may be
moribund and respond poorly to physical stimulation. Because of these
differences, the diagnosis is frequently delayed until destruction of
the physis and epiphysis has occurred. In the neonate who shows failure
to thrive, osteomyelitis should be considered as a possible cause. The
most dramatic physical finding in the neonate usually is swelling of an
entire extremity (Fig. 176.2). Radiographs should show the characteristic soft-tissue swelling.
area of maximum tenderness with a large-bore needle. Use fluoroscopic
control to make certain that the needle enters the metaphysis near but
not in the physis. If pus is not obtained from beneath the periosteum,
push the needle through the cortex. Culture any aspirate, pus, or blood
obtained. In addition to culturing the aspirate, culture the blood,
nose, throat, and any skin lesions. Perform peripheral blood studies,
including a white blood cell count, hematocrit, and sedimentation rate.
Two significant sequelae arise with osteomyelitis. One is physeal and
epiphyseal destruction in the neonate and infant; the other is chronic
osteomyelitis with a cortical sequestrum in the older child. Prevention
of these two problems depends on early diagnosis and effective
treatment. When these two sequelae are prevented, the results from
treatment of osteomyelitis is uniformly good.
rest and protection of the involved limb is successful in most
patients. Surgical treatment is indicated if pus is obtained on bone
aspiration or if the response to conservative treatment is not
favorable within 48 hours. A favorable response is one in which the
temperature elevation rapidly diminishes, and the pain and swelling is
decreased. If no abscess is present and the correct antibiotic is
delivered in an adequate dose, pain, fever, and even swelling should be
significantly reduced by 48 hours.
choice of antibiotics is made from the statistics concerning the most
likely pathogen as influenced by age, presence of chronic disease, and
any organisms found on the Gram stain. After culture results and
sensitivities are known, change the antibiotic to a more appropriate
one, if necessary.
group B Streptococcus, and gram-negative bacilli are frequent
pathogens. Therefore, in the neonate a semisynthetic penicillin will be
effective for the staphylococcal and streptococcal infections, but an
aminoglycoside should be added because of the possible presence of a
gram-negative organism.
this organism can be the pathogen in as many as 10% of the cases. For
these patients, cefuroxime may be the drug of choice because it is
effective against Staphylococcus, Streptococcus, and H. influenzae. If H. influenzae is the pathogen, the possibility of secondary meningitis is always present.
organisms are the most common pathogens causing osteomyelitis. In this
age group, a semisynthetic penicillin or a first-generation
cephalosporin is the antibiotic of choice. Both give adequate bone
levels and are effective. The advantage of the cephalosporins over
semisynthetic penicillins is that if the treatment is changed from
parenteral to oral antibiotics, the oral cephalosporins are more
palatable than either cloxacillin or dicloxacillin. If the pathogen
cultured is a methicillin-resistant S. aureus, vancomycin is the drug of choice.
or 3 days, if the response to treatment has been favorable with a
decrease in the fever, pain, and swelling, oral antibiotics may be the
preferred route of delivery in a selected group of patients. To use
oral antibiotics, there should be a positive culture to ensure that the
appropriate antibiotic is being administered. Compliance must be
ensured by knowing the dependability of the patient and family.
Tolerance of the oral antibiotic at the required dosage must be shown.
The dose of antibiotic must be sufficient to give a bactericidal level
as determined by measurement of the antibiotic level or by the dilution
technique. In the dilution method, a 1 to 8 dilution at the peak and a
1 to 2 in the trough should be bactericidal. The required dose of a
semisynthetic penicillin to obtain these bactericidal levels can be
reduced by the addition of probenecid in children of 2 years and older (33).
osteomyelitis, the duration of the antibiotic treatment is the
greatest. Dich et al. (10) reported data on a
series of patients with staphylococcal osteomyelitis treated for less
than 21 days compared with a group treated for more than 21 days. The
rate of recurrence of development of chronic osteomyelitis was 19% in
those treated for less than 21 days and only 2% in those treated for
more than 21 days. I prefer to judge the need for extended treatment on
the patient’s response and on the presence of bone changes on routine
radiographs. In all patients, antibiotics for 21 days should be the
rule, extending this to 6 weeks or more in selected cases. In the
patient who presents early and responds rapidly and in whom no abscess
is demonstrated by aspiration, the shorter period of antibiotic therapy
may
be adequate. When pus is present or radiographic evidence of bone
destruction or a sequestrum is present, give antibiotics for longer
periods of time. Before antibiotics are discontinued, the sedimentation
rate should be declining and near normal.
pus is obtained on the initial aspiration, if the clinical response is
not significant after 48 hours of antibiotics, or if there is
radiographic evidence of bone destruction requiring removal of a
metaphyseal sequestrum or granulation tissue. This latter indication is
controversial, and it may be preferable to depend on conservative
treatment before undertaking surgical curettage.
granulation tissue. Even though antibiotics may have been started, the
material removed at surgery should always be cultured. Make the opening
in the cortex small but large enough to insert a curet for removal of
granulation tissue and to irrigate the metaphysis thoroughly. Place the
hole near the physis in the area of maximum tenderness (Fig. 176.4),
taking care to protect the physis. This is best done under fluoroscopy
or by a marker placed at the level of the physis and confirmed by a
radiograph.
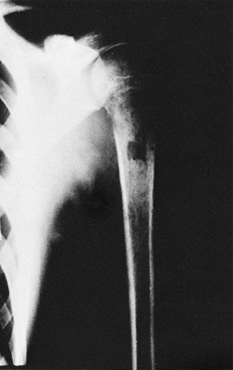 |
|
Figure 176.4.
The surgical opening in this proximal humerus is too distal to the epiphysis. The patient had a recurrence of symptoms 2 months later. |
Controversy exists as to whether irrigation with saline is helpful. If
it is done well, it should be of some benefit. Drains should be removed
after 48 hours.
position is important to prevent a fracture. It usually increases the
patient’s comfort level as well. Unprotected weight bearing after
surgical decompression or where there is extensive cortical destruction
with or without a sequestrum may result in a fracture.
results in a standoff, with local bone destruction that is limited
compared with that of acute osteomyelitis. This results in a subacute
infection. In 1969, King and Mayo (24) reported
data on a series of patients diagnosed as having subacute
osteomyelitis. In all of these patients, radiographic changes were seen
at the time the patients were first evaluated. They described eight
types of subacute osteomyelitis based on the radiographic appearance.
Others have added to this clinical description (14,15).
child is not ill, and little or no functional impairment is present.
The most constant complaint is a localized pain that may have periods
of exacerbation and remission. The pain frequently is exacerbated
following a period of unusual activity. If the involvement is in a
subcutaneous bone, local swelling is occasionally present. Like the
pain, the swelling seems to increase and subside with activity.
Symptoms may be present for weeks or months before the child is brought
to a physician for evaluation. Laboratory studies may be normal,
including the sedimentation rate, although it is elevated in some
patients.
the lesion. The most common type of subacute osteomyelitis is a
well-circumscribed lytic lesion with sclerotic borders, which is known
as Brodie’s abscess. Such lesions may be found in the metaphysis,
epiphysis, and rarely, in the diaphysis. Metaphyseal lesions frequently
extend across the physis and into the epiphysis (Fig. 176.5).
Fortunately, this appears to be a response that does not injure the
physis. A second type exhibits a lytic area in the cortex with little
or no bone response. In others, the cortex becomes very sclerotic but
without onion skin—like periosteal new bone. However, there is a
subperiosteal new bone type that has an onion skin appearance. Rarely,
the involvement
in the metaphysis may be diffuse without a clear border.
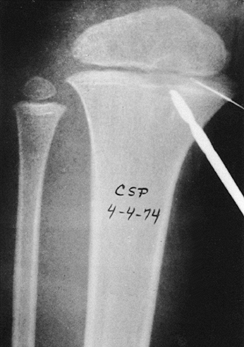 |
|
Figure 176.5. Brodie’s abscess extended from the metaphysis to the epiphysis. Normal growth continued after curettage of the lesion.
|
have an initial incorrect diagnosis. Subacute osteomyelitis should be
the diagnosis until proven otherwise if there is local swelling, with
local pain, and one of the classic radiographic appearances, along with
an elevated sedimentation rate. Bone scans are almost always positive,
although they are not diagnostic. MRI is frequently the most useful
imaging and should be used in those with a destructive lesion or in any
patient with radiographic changes suggestive of a malignancy.
surgery. If the diagnosis is established by the pathology, both gross
and microscopic, curet and culture the granulation tissue present in
the lesion. Start antibiotics immediately after surgery. In most of
these lesions, S. aureus is the pathogen;
therefore, a semisynthetic penicillin or a first-generation
cephalosporin is the drug of choice. If the diagnosis is made without
biopsy, some lesions will respond to antibiotics without surgery (35).
If the lesion is in the epiphysis and a surgical approach is
potentially harmful, the initial treatment should be antibiotics for
several weeks (Fig. 176.6). However, most
epiphyseal lesions can be drained without harm to the articular surface
or to the physis if the surgical approach is appropriately planned (15).
In the subperiosteal, cortical, sclerotic, and diffuse metaphyseal
types, surgical treatment has less to offer. In these patients, the
clinical picture is more likely to resemble a neoplasm, and a surgical
biopsy will be necessary for diagnosis. These types usually respond to
adequate antibiotic therapy.
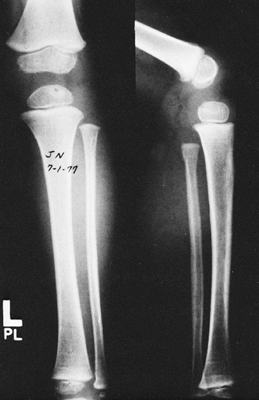 |
|
Figure 176.6. Lesion in the proximal epiphysis of the tibia.
|
than before the antibiotic era. It usually results from a delay in
diagnosis or inadequate treatment. It is seen more often today in North
America in immigrants from underdeveloped countries. The delay in
controlling the infection results in the formation of a cortical
sequestrum, which
is
due to ischemia from the cortical, intramedullary, and subperiosteal
spread of pus. The antibiotic cannot adequately reach the bacteria
located within dead bone, so surgical removal of the infected tissue
must be done.
after deep tissue cultures of the wound are made. Continue the
antibiotics for an extended period of time until the sedimentation rate
is normal and the wound is benign. The indication for surgical
treatment in chronic osteomyelitis is the presence of local pain and
swelling, with or without drainage, in a bone with an area of lysis or
a sequestrum, or both. Removal of the sequestrum and granulation tissue
is the goal of the surgical exploration. Care must be taken not to
jeopardize the integrity of the diaphysis by excessive removal of bone.
The sequestrum should be removed, preferably after the involucrum is
mature. Wide saucerization of the cortex should not be done because
bone regeneration in chronic osteomyelitis may be severely limited.
This is particularly true when there is no involucrum.
surgical treatment. If possible, the wound should be closed and drains
placed in the wound. Suction drainage and irrigation should be done for
several days postoperatively. An antibiotic irrigation solution may be
preferable to saline. The insertion of antibiotic-impregnated
methylmethacralate beads in the defect after debridement should be
considered in the patient who has had repeated surgical procedures. The
absence of an involucrum indicates an inadequate periosteum. Periosteal
healing is then unlikely. In such a patient, a cancellous bone graft
should be placed in the defect after there is healing from the initial
debridement and the infection is under control. If the bone cannot be
covered by local skin, a local muscle should be transferred to cover
the defect. In large uncovered areas, a myocutaneous flap may be
required to cover the bone and fill the defect adequately (41).
In some locations, a free microvascularized flap may be necessary. Good
full-thickness and soft-tissue skin coverage will accelerate and
improve the quality of healing (see Chapter 8 and Chapter 35). After surgical debridement, protect the extremity to prevent pathologic fracture.
Both produce fever, bone pain, tenderness, erythema, and swelling. An
increase in the white cell count and an elevated sedimentation rate are
also present. The presence of swelling and tenderness in the shaft of a
long bone is more typical of osteomyelitis in a patient with sickle
cell disease, whereas in a patient who does not have sickle cell
disease, osteomyelitis has its onset in the metaphysis.
astute evaluation of the degree of clinical signs and symptoms. The
bone should be aspirated and cultured, and blood cultures should made
if there is a strong possibility of infection. The pathogen in sickle
cell osteomyelitis may be S. aureus,
salmonella, or any other organism. Staphylococcus and salmonella are
the two most common organisms cultured in osteomyelitis in a patient
with sickle cell disease. In 15 patients with sickle cell disease with
osteomyelitis, Epps et al. (11) reported that S. aureus was culture in eight, salmonella in six, and Proteus mirabilis in one.
with the addition of antibiotics for 6 weeks. Chronic osteomyelitis in
the patient with sickle cell disease is more common than in those who
do not have sickle cell disease. Repeat surgical debridement may be
needed in those patients who are not free of symptoms by 6 weeks. As in
all patients with sickle cell disease, good hydration and transfusion
are preoperative requirements.
common in Europe than in the United States. In some reports, many
patients have pustulosis palmaris et plantaris as well as recurrent
multiple osteomyelitis (3,6,22). However, Yu and associates (45) reported on seven patients, none of whom had pustulosis palmaris et plantaris. Benhamou and associates (3)
found some patients who in addition to pustulosis palmaris et plantaris
also had Crohn’s disease and some others had polyarthritis. They
suggested that this condition was linked with seronegative
spondyloarthritis.
is gradual and usually without significant temperature elevation. Pain
is commonly reported. Lesions are most common in the femur, tibia, and
spine, but other bones may be involved. There may be swelling and
erythema over the lesions. Pressure over the involved part is painful,
and if the spine is affected, flexion and extension and pressure over
the involved vertebra causes pain.
sclerosis, hyperostosis, or lysis. The radiographic changes have the
appearance of bacterial osteomyelitis or of a sarcoma. Peripheral blood
changes are minimal. A technetium-99 scan may show increased uptake or
may be normal. Changes of inflammation suggestive of osteomyelitis
are seen on biopsy material. Cultures of the lesions are negative.
the patient has polyarthritis, symptoms generally last longer. Vertebra
plana may result if a vertebra is involved. The height of the vertebra
is not restored with time (45). Carr et al. (6)
reported one patient with progressive kyphosis that required spinal
fusion. Treatment is symptomatic with a nonsteroidal anti-inflammatory
drug. Carr et al. (6) found that antibiotics
may be helpful, and I have used cefalexin in one patient during
recurrent episodes with possible benefits; however, antibiotics are not
generally recommended.
gave credit to Eric Price for coining the word discitis for an
infection of the disc space with little or no bone involvement. For
many years, this remained a common concept of spinal infection in
children. However, with the use of tomography it became apparent that
osteomyelitis of the adjacent vertebrae always was present when the
disc was infection. Ring et al. (34) labeled
this condition pyogenic infectious spondylitis. The nomenclature for
discitis from a bacterial infection should no longer be confusing.
Pyogenic infectious spondylitis is not common but must be considered as
a possible cause of back pain in a child and particularly in the very
young child. The most frequent etiologic agent cultured is
staphylococcus; however, streptococcus and, to a lesser extent,
salmonella may be cultured (19,37).
symptom. Small children refuse to sit or walk. Leg pain and weakness
may cause the child to not walk or to limp. The classic picture is a
child sitting with the spine in extension and hands resting on the bed
behind the trunk for support. Percussion over the affected vertebrae is
painful even before changes on routine radiographs are present. When
the clinical picture of bacterial spondylitis is present, blood
cultures should be done. Hoffer et al. (19)
recommended computer-guided biopsy if cultures were negative. I believe
with the predominate bacteria known to cause this condition being
staphylococcus and streptococcus that it is reasonable to assume that
one of these organisms is present and to treat the child with the
appropriate antibiotic. A response should be noted within 3 days, and
if not, an additional antibiotic added or biopsy done. Patients who are
not treated with antibiotics do recover when the spine is immobilized;
however, this approach may prolong the time to recovery and can
increase the need for surgical drainage (34). I prefer to administer an antibiotic for the infection and to provide symptomatic treatment to relieve back pain.
The ilium is the most frequent pelvic bone affected, but the ischium
and pubis may be involved. Pain about the hip and a limp or refusal to
walk are commonly reported. Most patients have a fever. The white cell
count is elevated, as is the sedimentation rate.
buttocks or low back, with sciatica-like symptoms. The location of the
infection determines the location of pain. The hip is the most common
area for pain, and when this is the presentation, an infection has to
be considered. This pain occurs when the ilium is involved near the
innominate bone, and osteomyelitis near the acetabulum can decompress
in the hip joint and infect the joint. Buttock pain occurs if the ilium
outer table is eroded, usually near the sacroiliac joint. Decompression
through the inner wall may cause sciatica-like symptoms if the pus goes
into the true pelvis and irritates the sciatic nerve or the pus may
ascend and cause abdominal pain.
laboratory changes, the physical examination, and the use of the bone
scan, CT, and MRI (30,40).
Initial imaging with any of these modalities may be normal early in the
course of the disease, but all tests will become positive eventually.
The MRI will ordinarily be the first to show the swelling and bone
involvement. It can distinguish soft-tissue swelling and show whether
it is from bone or has a nonosseous origin. The MRI cannot
differentiate infection from a tumor or infarction (40).
patient can walk. Perform range-of-motion examination of the hip
gently. Passive motion of the infected hip is usually quite painful and
limited whereas in infection of the pelvis more motion is present and
less painful. Pressure on the pelvis is painful. Pressure over the
buttock is painful if the ilium has decompressed through the outer
table.
orally. If treatment is started before an extraosseous abscess
develops, antibiotic treatment is the only treatment required. When an
abscess is present, surgical drainage is appropriate. Drain gluteal
abscess posterior through the gluteus maximus, and a pelvic abscess
through the abdomen but stay retroperitoneally. Chronic osteomyelitis
of the pelvis is very rare, and most patients recover without residual
problems (30).
affects the very young child. The peak incidence is between the age of
1 and 2 years of age (16). In neonates and other infants, the hip is the joint most commonly affected, but
the knee is more commonly involved in older children. Infection in the
hip joint is frequently secondary to osteomyelitis of the proximal
femur, particularly in the infant. Septic arthritis of the shoulder and
ankle may be secondary to spontaneous decompression of pus from the
proximal humerus and distal fibula, respectively. Residual effects from
septic arthritis are related to a delay in diagnosis and treatment, and
to the presence of osteomyelitis.
was the organism most commonly cultured from septic joints. In a 1967
study of 116 infected joints in children with a mean age of 3 years, S. aureus was the most frequent organism cultured and 8% had H. influenzae (14). In the 1970s, the incidence of H. influenzae increased and was the most common organism cultured in children younger than 4 years of age (17). Jackson and Nelson (20) reported S. aureus
in 30%, group B streptococcus in 21%, and gram-negative organisms in
28% in children aged 1 month to 5 years. However, in the infant younger
than 3 months of age who acquires a joint infection while in the
hospital, staphylococcus is the most common organism cultured.
Approximately two thirds of hospital-acquired joint infections were
found by Dan (8) to have S. aureus and one fifth Candida species as the pathogen. Since the development of the vaccine for H. influenzae, this organism is rarely the pathogen in septic arthritis.
hematogenous route. This may be either direct inoculation into the
synovium or secondary to hematogenous osteomyelitis that decompresses
into the joint. Osteomyelitis with secondary septic arthritis of the
hip is common in the neonate but can occur in children of all ages. The
ankle and shoulder, where the metaphysis may be partially
intracapsular, occasionally is infected secondary to osteomyelitis. In
the infant, in whom there are vessels that transverse the physis from
the metaphysis to the epiphysis, the infection can spread from the
metaphysis to the epiphysis and the adjacent joint. Bacteria are
deposited in the synovium, and an inflammatory reaction develops. The
inflamed synovium allows blood products and bacteria to enter the
synovial fluid, including large numbers of leukocytes. The inflamed
synovium, the white blood cells, and the bacteria contribute to the
enzymatic destruction of the articular cartilage by the release of
collagenase and proteases. Even the chondrocyte may contribute to
cartilage destruction; Jasin (21) has shown
that the chondrocyte may be stimulated to release chondrolytic enzymes
by the action of either bacterial liposaccharide or interleukin 1
(IL-I). The depletion of glycoaminoglycans in the cartilage matrix
begins rapidly. Within 24 hours there is a significant loss. The loss
of collagen follows (38). It is because of the
multiple sources of these enzymes (bacteria, white blood cells, and
synovial cells) that antibiotics alone cannot prevent destruction of
the cartilage. Thorough cleansing of the joint is essential to
successful treatment.
rapid development of joint pain and a fever of 100° to 104°F (38° to
40°C). Although the rapid onset of pain is variable, it is usually
severe within 24 to 48 hours so that the child refuses to use the
extremity. Irritability and malaise may precede the onset of pain.
Pseudoparalysis is common in the very young. The physical examination
is dramatic in the severity of the limitation of motion of the affected
joint. Even with the gentlest attempt to move the extremity passively,
there is extreme pain and spasm, and little motion is obtained. The
joint is swollen, hot, and globally tender. The affected joint assumes
a resting position that maximizes the capsular volume to reduce the
tension in the joint. The hip is held flexed abducted and externally
rotated. The most comfortable position for most other joints is some
degree of flexion. In the neonate, minimal spontaneous movement,
swelling of the joints, slight fever, and irritability may be the only
changes present.
usually, but not always, dramatic enough to exclude other diagnoses.
However, always aspirate a joint when infection is considered to be a
possibility, even when the child does not have the classic severe pain
of septic arthritis. Infected joint fluid is cloudy, and mucin is
diminished. A drop of fluid rubbed between the thumb and a finger will
feel watery and will not string as the opposed fingers are separated.
The most important studies of the fluid are a culture, Gram stain,
white blood cell count, and differential. If sufficient fluid is
available, other valuable measurements that should be obtained are
glucose and lactic acid levels. The white cell count is usually between
50,000 and 200,000. More important, the differential is greater than
90% polymorphonuclear leukocytes. A joint fluid sugar of 50 mg/dl less
than the blood sugar is common. In nongonococcal arthritis, the lactic
acid level is elevated. It is important that the specimen for cell
count is anticoagulated; if this is not done, the fluid will quickly
coagulate. This makes the cell count incorrect. Aspiration of the hip
and shoulder should be done under fluoroscopic control. If no fluid is
aspirated, injection of a radiopaque dye into the joint will confirm
that the needle is intra-articular.
studies should be done to include a complete blood count and
sedimentation rate. The white blood cell count is generally elevated
with an increase in polymorphonuclear leukocytes, and the sedimentation
rate is elevated. If the child is seriously ill and the infection
present for several days, the erythrocyte count and hematocrit will be
low.
be helpful when the cultures are negative; it is particularly helpful
when there has been partial treatment by antibiotics. This test can
identify the presence of H. influenzae, Streptococcus pneumoniae, and meningococcus (9). A gallium scan has some use when the diagnosis is difficult. Bowman et al. (5)
used gallium and reported accuracy in diagnosis of 91% in 34 patients
with septic arthritis. The radiation dose from gallium is higher than
from technetium, and it should be used only in difficult cases.
joint pain, and limited motion, there is little to confuse the
diagnosis. However, conditions such as toxic synovitis, monarticular
rheumatoid arthritis, and osteomyelitis can at times present a
diagnostic problem. The child with toxic synovitis can usually be
excluded by the clinical findings. The child is not ill, pain is not
severe, and motion is only slightly limited. Acute-onset monoarticular
rheumatoid arthritis may have many of the features of sepsis, and in
such a patient, joint fluid analysis may be the only way to
differentiate between these two diseases early in the course of the
disease. In rheumatoid arthritis, pain can be severe, motion very
limited, and the child febrile and ill. The synovial fluid may have as
many as 70,000 cells, but on the differential, there will be less than
80% polymorphonucleocytes, unlike the 90% to 100% found in septic
arthritis. In rheumatoid arthritis, the joint sugar level is similar to
that in the blood sugar. The mucin in rheumatoid synovial fluid is
diminished, as it is in septic fluid. The leukocyte differential and
the glucose levels may be the only differentiating feature between
these two diseases.
septic arthritis. In both conditions, the child may be febrile and have
the peripheral blood changes of infection. In both conditions, the
patient exhibits limited joint motion, but in osteomyelitis, the
adjacent joint will generally have a moderate range of motion if the
examiner is gentle and protects the extremity from sudden motion. By
careful palpation, tenderness will be found over the infected
metaphysis and not the joint. Swelling is also different. In
osteomyelitis, the swelling begins over the metaphysis and spreads to
include much of the extremity segment (Fig. 176.2).
The adjacent joint may be swollen from a sympathetic effusion, but it
is not particularly tender. The swelling in septic arthritis is
confined to the intracapsular space. If there is swelling over the bone
and also joint effusion, it is important to aspirate both the joint and
the bone, with aspiration first at the most unlikely site for the
infection, followed by aspiration of the suspected site. Radiographs
will show the typical deep soft-tissue swelling if osteomyelitis is
present. When soft-tissue swelling is present in the thigh and the hip
joint space is wide on the radiograph, the patient has osteomyelitis
plus a secondary septic arthritis until proven otherwise (Fig. 176.7).
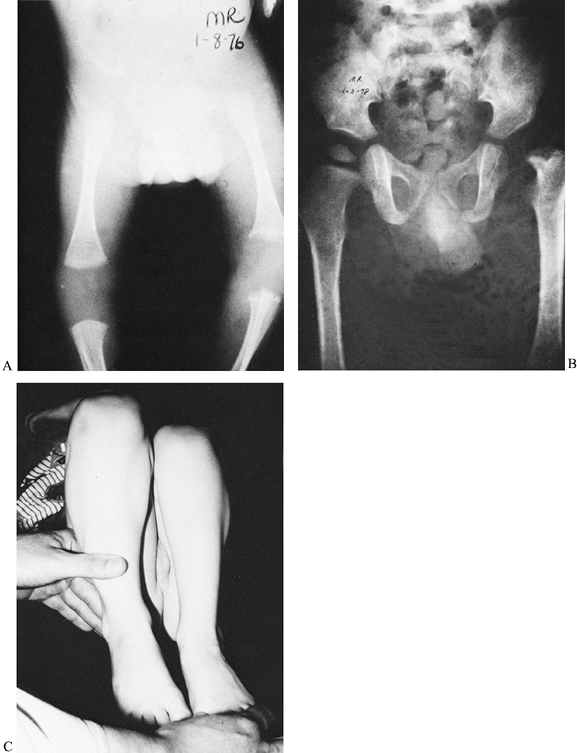 |
|
Figure 176.7. A: Soft-tissue swelling in a neonate. Notice the hip joint space. B: Two years later, the metaphysis is subluxated, the acetabulum is dysplastic, and there is no ossified femoral head. C: This image demonstrates the shortening of the left femur.
|
evacuate the debris associated with infection, relieve pain, and
prevent deformity. Since the discovery of antibiotics, controversy has
arisen as to the most effective way to cleanse the joint of the
products of infection. This has centered mainly on whether or not
aspiration and irrigation are as effective as arthrotomy and
irrigation. There are no adequate studies to evaluate the superiority
of one method over the other, and because of the many variables, it is
unlikely that a comparative study will ever be done.
with the appropriate antibiotic are the most important aspects of
treatment (17). There is little argument that
the hip is special and needs to be decompressed to prevent dislocation
as well as a tamponade of the circulation to the femoral head.
Arthrotomy is necessary for the hip to be cleansed adequately of the
debris and intra-articular pressure relieved. Intermittent or
continuous irrigation for 48 to 72 hours through catheters has been
used in the past (16). There is little proof
that it affects the outcome, but it is not to be condemned. However,
antibiotic solution should not be used for irrigation. Antibiotics
reach the synovium and synovial fluid sufficiently to deliver the
necessary bactericidal concentration of the antibiotic into the joint.
If postarthrotomy intermittent irrigation is used, extreme care to
maintain sterile technique is important to prevent a nosocomial
infection through the irrigation system. I no longer use postarthrotomy
irrigation.
aspiration of the joint to confirm the diagnosis. If pus is obtained,
the joint should be irrigated with saline until the irrigation fluid
returns clear. The hip and shoulder should be immediately opened and
irrigated after aspiration and joint fluid analysis confirms the
diagnosis. In other joints, if infection has been present for 4 or 5
days, the large amount of fibrin and debris that has accumulated may be
difficult to remove by needle. In these patients, arthrotomy or
irrigation by arthroscopy is the preferred method of joint debridement.
I do not recommend arthroscopic irrigation because I believe the
motions of the arthroscope necessary to remove the debris adequately
irritates the inflamed synovium. I have seen several failures when this
technique is used in a joint that by all rights should have had a successful outcome.
posterior approach. In the anterior approach the posterior superior
epiphyses vessels are less likely to be damaged, and the anterior
arthrotomy does not leave a posterior defect in the capsule where the
hip can dislocate.
-
Make an anterior approach (see Chapter 3).
-
Then make a 1 cm capsulotomy through which the joint is irrigated.
-
I do not drill the femoral neck. In the
infant, only a small portion of the neck within the capsule is
ossified, so drilling is likely to injure the physis. If the septic hip
is secondary to osteomyelitis, the femoral neck has already
decompressed itself. -
Although I no longer use irrigation
postoperatively, I do leave a drain near the opening in the capsule and
remove it at 48 hours. -
Take a synovial biopsy for culture and close the skin. Synovial tissue cultures may be positive when the aspiration is negative.
arthrotomy, I place the extremity in balance suspension traction for
several days in the older child. If properly applied, traction protects
the joint and relieves pain, separates the joint surfaces, and allows
motion. Protective use of the joint can begin shortly after the joint
is no longer painful. If the hip appears unstable or it is dislocated,
it should be held in the reduced position with a spica cast. Salter et
al. (36) has shown that in the rabbit,
continuous passive motion is beneficial to the articular cartilage. Its
value in septic arthritis in children has not been established, however.
systemic parenteral antibiotic should be started. It should be one that
is effective for the most likely pathogen. Age, environment, and the
Gram stain determine the initial choice of antibiotic. In the neonate,
multiple organisms are not uncommon. A semisynthetic penicillin for the
staphylococci and streptococci plus an aminoglycoside for the
gram-negative organisms should be used in the neonate. Between the age
of 1 month and 5 years, H. Influenzae is as common as staphylococcus and streptococcus infection if the child has not been vaccinated for H. influenzae.
In this age group, cefuroxime or ceftriaxone may be the drug of choice
because they are effective against both the gram-positive cocci and H. influenzae.
After age 5 years, a semisynthetic penicillin or a first-generation
cephalosporin is the drug of choice. Both are effective against
streptococcus and staphylococcus organisms. The oral cephalosporins
taste better than the oral synthetic penicillin, and for that reason, a
cephalosporin may be preferable.
intravenous antibiotics can be effective in selected patients. The
prerequisite for the use of oral treatment is a rapid clinical response
to treatment, a positive culture, an available laboratory to measure
serum levels, a reliable family, and proof that the child can tolerate
the oral antibiotic. A positive culture is beneficial for proving the
effectiveness of the blood level of the agent being used. The dosage
has to be sufficient to give a peak serum bactericidal level at a
dilution of between 1 to 8 and 1 to 16, and a dilution of 1 to 2 at the
trough or a bactericidal concentration present in the trough for those
antibiotics that have a bactericidal level. In a patient who does not
have a positive culture but who has shown a rapid response to the
parenteral antibiotic, it is not unreasonable to continue treatment
with an oral antibiotic. The duration for antibiotic treatment is not
absolute. In uncomplicated cases without osteomyelitis, 2 to 3 weeks
should be sufficient. Be sure the patient is symptomatically doing well
and that the sedimentation rate is reduced before discontinuing the
antibiotic.
treated within 2 days and in some cases even longer are minimal. In the
infant, a delay in diagnosis and treatment is more likely than in older
children. The hip is more commonly affected in the infant, and
osteomyelitis with secondary septic arthritis is more common in the
proximal femur. Until the physis becomes a barrier to the metaphyseal
vessels crossing into the epiphysis, the physis and epiphysis are in
jeopardy. This anatomic fact and the frequent delay in diagnosis in the
infant are the reasons that the poorest results from septic arthritis
are in infants and in the hip. The physis and the epiphysis may both be
completely destroyed by osteomyelitis with associated septic arthritis.
Spontaneous decompression of the pus into the hip joint may cause
damage to the epiphyseal blood supply by tamponade, and the
intracapsular pressure may cause a dislocation of the hip. In my report
in 1967, 15% of infected hips had a poor result and 75% of poor results
were patients with septic arthritis of the hip secondary to
osteomyelitis (16).
in the hip, where delay in treatment of the infant has allowed the
partial or complete destruction of the physis and epiphysis. If the hip
is dislocated, closed reduction is superior to open reduction (44).
When the residual femoral head is inadequate to be effective as a
functioning hip; attempts at surgical reconstruction should be avoided.
Following total destruction of the head and neck, trochanteric
arthroplasty has been done in an attempt to improve function and to
maintain length (7). This procedure has many
problems. With time, dislocation gradually occurs. A varus osteotomy
delays the subluxation, but with time,
the varus gradually straightens and subluxation occurs (13). Betz et al. (4)
reported that the long-term functional results in the patient in whom
the head is destroyed is better if reconstruction is not attempted.
sequela to hip infection, it can be successfully treated by the
appropriate osteotomy. The prerequisite to an osteotomy about the hip
is a femoral head that is stable and sufficient to function
effectively. If this is not possible, reduction of the head should not
be attempted.
countries of the developed world, particularly in North America. But it
remains a common scourge in underdeveloped countries. Over the past
decade, there has been a substantial increase in migration to developed
countries from underdeveloped areas, such that tuberculosis is now more
common and must always be suspected in children who present with
chronic infections, particularly if their families have recently
immigrated from underdeveloped countries. Tuberculosis is a chronic
granulomatous infection caused by Mycobacterium tuberculosis. In countries where raw milk is consumed, bovine transmission can cause infection by Mycobacterium bovi.
Tuberculosis is a localized destructive disease that spreads by the
hematogenous route from a primary focus, most commonly located in the
lungs and infected mediastinal lymph nodes.
joints can occur by direct hematogenous infection of the synovium or by
invasion of the joint from an adjacent osteomyelitis involving the
epiphysis or metaphysis. A tuberculous focus in bone spreads by
centrifugal destruction of bone, producing increasing amounts of
exudate and caseous necrotic material. Increasing pressure and bone
destruction results in perforation of the bony cortex, forming a
soft-tissue “cold abscess” so named because of the absence of acute
inflammation. The infection spreads along tissue planes and may present
as a subcutaneous abscess or fistula.
adjacent bone involvement results in the proliferation of tuberculous
granulation tissue in the joint, which produces a pannus that rapidly
covers articular cartilage, destroying the cartilage and underlying
subchondral bone. Destruction is most extensive around the periphery of
the joint at the attachments of the synovial membrane.
with a history of easy fatigability and weight loss. The evolution of
the disease is insidious, and involvement is usually monoarticular or
in a single site. It is important to seek a family history of
tuberculosis. With lower extremity involvement, the patients limp and
the affected joint is stiff. Crying at night is typical, because the
pain seems to increase because protective muscle spasm relaxes at
night. Physical findings depend on the anatomic area involved.
Infection is most frequent in the spine, followed in order of frequency
by the hip, knee, ankle, sacroiliac joint, shoulder, and wrist.
Tuberculous spondylitis in the child is characterized by a painful,
stiff back and a protective gait in which the child keeps the back
hyperextended. Infection in the thoracic spine and the thoracolumbar
junction is common. Kyphosis develops as bone destruction progresses.
In the extremities, muscular atrophy is usually marked.
anemia, normal or only a slight increase in the peripheral white blood
cell count, a modestly elevated erythrocyte sedimentation rate, and
positive tubercular skin test. Synovial fluid analysis shows a white
blood cell count averaging 20,000 cells/mm3 (range 3,000 to
100,000 cells), with 40% lymphocytes and monocytes, which is much more
than that seen in pyogenic infections. Cultures are usually positive,
but diagnosis can be quickly confirmed by histologic examination of
tissue obtained by biopsy of the synovium of infected joints or sites
of bone involvement.
demonstrate the changes from tuberculous arthritis, although it is
often indistinguishable from monoarticular rheumatoid arthritis.
Characteristic findings are the triad of Phemister, which consists of
periarticular osteoporosis, gradual narrowing of the joint space, and
erosions of the bone peripherally at the synovial attachments. In the
late stage of tuberculosis, there may be complete destruction of
joints, with dense sclerotic changes in adjacent bone. The disease is
typically monoarticular, as opposed to juvenile arthritis, which is
usually polyarticular. In the spine, the initial presentation shows
disk space narrowing and destruction of the adjacent endplates of the
vertebrae. With progression of the disease, a paraspinal mass is
common. Subsequent collapse of involved vertebra and extension of the
infection to adjacent levels leads to kyphosis and formation of a
gibbous. Infection often extends along the psoas muscle sheaths and can
present as abscesses in the flanks or groin. Paraplegia can occur due
to tuberculous involvement of the meninges or due to mechanical
pressure from the infection and collapse of the vertebral elements.
This is known as Pott’s disease.
drugs. General medicine measures to treat other focuses of the disease
and to ensure good health habits and adequate nutrition are important.
Orthopaedic care consists of conservative measures to preserve motion
and strength, and to prevent deformity. Surgery is performed when
necessary to debride necrotic bone and soft tissue, and to eliminate
abscess. Today, surgery is most commonly required to correct spinal
deformity and treat paraparesis. For
treatment,
three drugs are preferred, and because of the large percentage of
drug-resistant infections in certain areas, four drugs may be
advisable. Drugs include isoniazid, rifampin, streptomycin, ethambutol,
and pyrazinamide, as well as others. Pyridoxine supplementation may be
necessary when treating with isoniazid. Monitor patients for
hepatotoxicity, impaired renal infection, eighth cranial nerve
toxicity, serum sickness–like syndromes, and thrombocytopenia.
scheme: *, classic article; #, review article; !, basic research
article; and +, clinical results/outcome study.
L, Chamot AM, Kahn MF. Synovitis-Acne-Pustulosis
Hyperostosis-Osteomyelitis Syndrome (SAPHO) A new Syndrome Among the
Spondyloarthritis. Clin Exp Rheumatol 1988;6:109.
J. Undersuchangen uber die Lokalisation der Bakteries das Verhalten des
Knochen Markes und die Verenderungen der Knochen, inc besondere der
Epiphysen bei Infekuouskrankheiten. Z Hyg Infectionskr 1911;69:436.
JH, Ross G, Cumming J, et al. Usefulness of Magnetic Resonance Imaging
for the Diagnosis of Acute Musculoskeletal Infections in Children. J Pediatr Orthop 1995;15:144.
CG, Yeager AS. Use of the Serum Bactericidal Titer to Assess the
Adequacy of Oral Antibiotic Therapy in the Treatment of Acute
Hematogenous Osteomyelitis. J Pedaitr 1979;96:131.
RB, Bell RS, Keeley FM. The Protective Effect of Continuous Passive
Motion on Living Articular Cartilage in Acute Septic Arthritis: An
Experimental Investigation in the Rabbit. Clin Orthop 1981;159:223.
RL, Schurman DJ, Kajiyania G, et al. The Effect of Antibiotics on the
Desctruction of Cartilage in Experimental Infectious Arthritis. J Bone Joint Surg 1987;69-A:1063.
