FRACTURES AND DISLOCATIONS OF THE PELVIC RING
II – FRACTURES, DISLOCATIONS, NONUNIONS, AND MALUNIONS > Pelvis and
Femur > CHAPTER 17 – FRACTURES AND DISLOCATIONS OF THE PELVIC RING
constitute a diverse group of skeletal injuries that usually result
from motor-vehicle accidents, industrial trauma, sporting events, or
falls from great heights. Apart from isolated fractures of individual
bones of the pelvic ring or avulsion fractures documented in athletes,
the typical pelvic ring disruption is notable for the immense amount of
force needed to provoke a displacement of the pelvic ring. A patient
who sustains such an injury is likely to present with other serious or
life-threatening injuries involving the musculoskeletal, respiratory,
central nervous, gastrointestinal, urologic, and cardiovascular systems
(14). The management of a pelvic ring fracture,
therefore, requires concomitant diagnosis and treatment of the other
systemic and musculoskeletal injuries.
pelvic ring fracture has focused on the emergency care of the
associated injuries with an initial neglect of the osseous disruption.
Usually, the immobilization of the pelvic fracture was accomplished by
bed rest and possibly the application of a pelvic sling, skeletal
traction, or a hip spica cast. Such marginally effective methods of
stabilization are inadequate for the control of profuse retroperitoneal
hemorrhage, which often complicates acute pelvic disruption.
Furthermore, bed rest and especially the use of a pelvic sling were
notable for extraordinary discomfort and general failure to achieve an
accurate reduction of a displaced fracture–dislocation.
complete immobilization of the patient, with the concomitant
complications of prolonged, enforced recumbency including urinary
retention, urinary tract infections, pulmonary emboli, infections,
decubitus ulcers, and sloughing of soft tissues under a sling. In the
past, most traumatologists tried to minimize complications by reacting
to problems that arose during or after a period of enforced recumbency
rather than anticipating the need for surgical intervention.
Historically, this reactionary format has characterized the whole
management protocol for pelvic trauma, including investigative
procedures. During the 1990s, McMurtry et al. (35) and Tile (70)
emphasized the need for a thorough diagnostic workup in virtually all
patients with pelvic and acetabular fractures so that the associated
injuries and the nature of the pelvic osseous disruption are accurately
documented.
traumatologists that most pelvic fracture victims who survive these
injuries have few major late problems. Retrospective analyses of pelvic
fracture victims are notable for the high incidence of late problems,
however, including pelvic pain, abnormalities of gait, limb-length
discrepancy, permanent nerve damage, and genitourinary tract problems.
During the 1990s, external fixation was recognized as an effective
method to control the profuse retroperitoneal hemorrhage associated
with major pelvic fractures and to lessen the typically severe pelvic
pain (57). When all the available series in
which this method has been used to manage unstable pelvic disruptions
are analyzed, about 40% of the patients complain of late pelvic pain
with or without a persistent deformity or a mobile nonunion (69,70). Early open reduction and internal fixation appears to reduce the incidence of these late problems (51).
Nevertheless, with the potential complications of the
surgery—particularly where an exposure of the pelvis necessitates
anatomic approaches that are not widely used for typical orthopaedic
procedures—other complications, including major neurovascular injuries
and visceral insults, are likely to occur unless the surgical team is
appropriately trained.
reconstruction are undergoing rapid change. This chapter is based on
our experience and the recommendations of other surgeons with an
interest in this field.
highly diverse group of injuries that are characterized by the force of
the provocative blow; the site(s), magnitude, and nature of adjacent
soft-tissue disruption; the quality of the involved tissues; and the
potential presence of a total hip
replacement (16,70). A pelvic disruption is the hallmark of violent or major trauma (Table 17.1).
 |
|
Table 17.1. Sources of Violent or Major Trauma to the Pelvis
|
blow, specific injury patterns are likely to ensue that involve diverse
supportive elements of the pelvic ring (Fig. 17.1). The injury patterns (69) are characterized by the sites of disruption and the magnitude and direction of displacement (Fig. 17.2).
The external rotation injuries are further subdivided by the magnitude
of the diastasis of the pubic symphysis and the potential presence of a
sagittal malrotation or posterior displacement of the involved
hemipelvis (Fig. 17.3). A crushing injury is
typified by a logger who sustains a blow by an errant tree that strikes
his pelvis. The force of the blow is likely to displace the involved
portion of the pelvis, especially the iliac wing, into the
intraabdominal cavity to provoke a frank or occult injury to the bowel
or bladder (Fig. 17.4). Upon elastic recoil of
the fracture fragments, a seemingly benign radiographic picture can
mask a serious visceral impairment.
 |
|
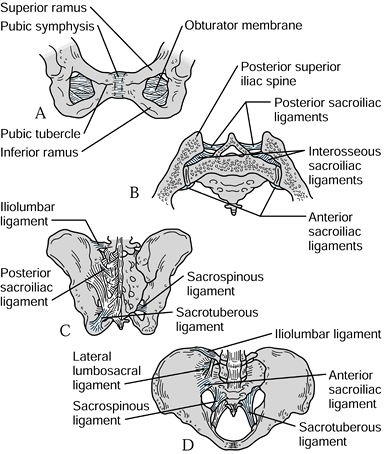
Figure 17.1. Schematic views of the pelvis with the principal ligamentous supports. A: Symphysis pubis fibrocartilage. B: Posterior sacroiliac ligaments. C: Posterior view. D: Anterior view.
|
 |
|
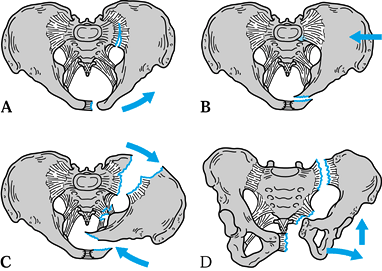
Figure 17.2. Schematic view of the principal pelvis injury patterns as determined by the vector of the provocative blow. A: Anteroposterior compression or external rotation injury. B: Stable lateral compression or internal rotation injury. C: Unstable lateral compression or internal rotation injury. D: Unstable vertical shear disruption.
|
 |
|
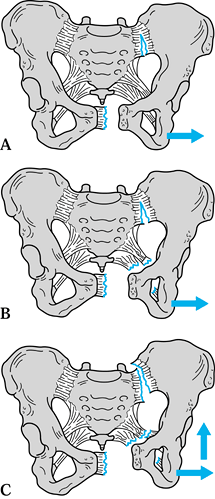
Figure 17.3. Subclassification of a diastasis of the symphysis by the vector and magnitude of the displacement of the involved hemipelvis. A: External rotation. B: External and sagittal malrotation. C: Posterior displacement with sagittal and external malrotation.
|
 |
|
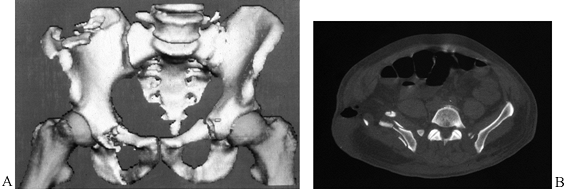
Figure 17.4.
The three- (3D) and two-dimensional (2D) CT views display a stable iliac fracture in a logger of 42 years who was struck by an errant tree. In the 2D CT, the highly displaced loop of small bowel superficial to the lateral ilium accompanied the elastic recoil of the iliac fragments, producing a small bowel obstruction. |
increase in pelvic trauma secondary to moderate or minor trauma
involving osteopenic bone is being encountered (46,55). Osteopenia varies widely in etiology and severity (Table 17.2),
and in the most florid cases it accounts for pathologic and
insufficiency fractures in which no relevant traumatic insult can be
identified. While the healing potential of the bone varies greatly
across this spectrum of clinical problems, overall the postirradiation
group displays the poorest healing potential. A representative example
is shown in Fig. 17.5.
 |
|
Table 17.2. Moderate and Minor Trauma Involving Osteopenic Bone
|
 |
|
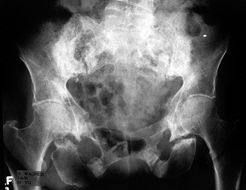
Figure 17.5.
Outlet view of a woman of 64 years with eight radiation-induced pathologic fractures and nonunions of bilateral superior and inferior rami, lateral ilia, and sacral ala. |
of the pelvis that acutely or belatedly follows the insertion of a
total hip arthroplasty. The injury patterns are markedly
heterogeneous,
depending on the force of the injury and the presence of osteopenia,
typically secondary to steroid ingestion or irradiation. In such a
clinical case, the fracture may extend from the sacral ala into the
adjacent posterior ilium and across the quadrilateral surface, with
loosening of the cup (Fig. 17.6).
 |
|
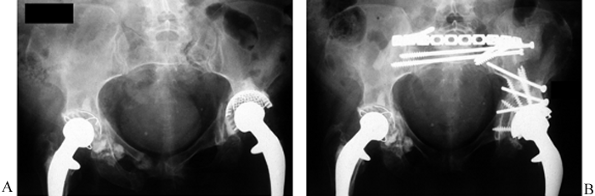
Figure 17.6.
Pre- and postoperative AP pelvic views of a steroid-dependent lupus patient of 35 years who developed an insufficiency fracture of the left sacral ala that extends along the left greater sciatic buttress and across the quadrilateral surface with loosening of the acetabular cup. A: Preoperative view. B: Postoperative view after internal fixation and cup revision. |
some degree of hemorrhage is inevitable. The principal sites of
bleeding are outlined in Table 17.3 The
anticipated sites of major hemorrhage correlate with the region of the
pelvis fracture, the vector of the provocative blow, and the magnitude
of pelvic displacement. For example, a displaced fracture of the
posterior ilium that exits through the roof of the greater sciatic
notch is likely to be associated with an injury to the superior gluteal
vessels.
 |
|
Table 17.3. Principal Sites of Hemorrhage after a Pelvic Fracture
|
of one or both hemipelvi, in which the ligamentous floor of the pelvis
is disrupted, a large volume of blood may collect in the
retroperitoneal space (42) and extravasate into
the scrotum, the thighs, and superiorward. With pelvic instability,
possibly combined with a consumption coagulopathy in a hypothermic
patient, the volume of blood that may be extravasated around the pelvis
is truly enormous, and it may require 50 or more units of blood to be
transfused. These pathologic responses to major pelvic trauma are
minimized by prompt immobilization of the pelvis, correction of major
deformity, direct control of hemorrhage from large vessels or tamponade
of smaller ones, along with general resuscitation of the patient. In
the presence of a large open wound, packing is a critical measure to
provide tamponade (51).
The cutaneous disruptions as well as visceral injuries provide a source
of contamination for the bone and can lead to an infection of the
pelvis and hip joint (Table 17.5).
 |
|
Table 17.4. Cutaneous Manifestations of Pelvic Trauma
|
 |
|
Table 17.5. Potential Sources of Infection for the Injured Pelvis and Hip Joint
|
sites for neurologic impairment are listed in Table 17.6.
 |
|
Table 17.6. Potential Sites of Neurologic Impairment with Pelvic Trauma
|
laceration, and iatrogenic injury resulting from the surgery. Direct
compression of the nerve, caused, for example, by a displaced sacral
bone fragment in a foramen, may benefit from surgical decompression.
Other types of neurologic injury do not respond favorably to available
surgical measures.
Injury to the lower urinary tract is a common consequence of a
disruption of the anterior pelvic ring, and it can involve the ureter
or, more commonly, the bladder and urethra. Urologic injury occurs in
as many as 25% of all cases involving disruption of the pelvic ring,
and it is more common with bilateral disruptions of the pubic arch. In
the setting of a fractured pelvis, extraperitoneal bladder rupture at
the anterolateral aspect near the neck is more common than
intraperitoneal rupture along the bladder dome; the converse is true in
blunt pelvic trauma without ring disruption. The overall occurrence
rate of a pelvic fracture with a concomitant bladder injury is reported
to be as high as 15%. Urethral injury is common with a disruption of
the anterior pelvic ring, such as a pubic ramus fracture; 65% of these
injuries are complete disruptions. The incidence of concomitant bladder
neck injury is as high as 2.5%. There is an increased likelihood for
shearing of the significantly longer male urethra, with a reported
occurrence rate up to 11% for male versus 6% for female patients. In
the typical lateral compression type of pelvic fracture, displacement
of the pubic ramus relative to the ischial ramus results in a classic
urethral disruption just distal to the apex of the prostate gland. If
both rami are displaced as a combined unit, the injury is likely to
occur at the juncture of the membranous urethra and the bulbous
urethra. While no consistent association between a specific urologic
injury and a specific mechanism of pelvic fracture has been reported,
generally the severity of the pelvic injury pattern determines the
extent of the urologic injury.
 |
|
Table 17.7. Potential Sites of Visceral Injury with a Pelvic Fracture
|
include the presence of blood at or around the urethral meatus, local
swelling, the inability to void, gross hematuria, or a high-riding
prostate gland. If any of these signs are present, obtain a dynamic
retrograde urethrogram prior to the insertion of a urinary catheter to
rule out a significant urethral injury. After catheterization, do a
static cystogram to evaluate the bladder. In female patients, a
meticulous gynecologic examination is essential, particularly in the
presence of vaginal bleeding.
fracture in the presence of a concomitant urologic injury was a common
error, which occurred because of the fear of a postsurgical infection
secondary to urinary contamination of the surgical wound. In actuality,
these concerns have been exaggerated. Routt et al. (60)
reported a low rate of infection and a 30% incidence of late urologic
complications when definitive internal fixation of a pelvic fracture
was combined with indirect stabilization of a urethral disruption with
a temporary stent by the use of two magnetic catheters. Currently, with
the diverse methods that are available to stabilize a pelvic disruption
in the presence of urethral trauma, an alternative strategy for
fixation can be used that minimizes the risk of a wound infection.
Admittedly, most urologists favor a delayed urethral reconstruction,
which in their view lowers the risk of impotence, incontinence, and
urethral stricture. When the urologic injury is a rupture of the
bladder, an acute surgical repair may be indicated, while concomitant
fixation of the anterior pelvic arch with a plate is strongly
recommended. Likewise, concomitant injuries of the urethra and bladder
may warrant immediate surgical repair. In the presence of a stable
pelvic fracture and an extraperitoneal bladder injury, nonoperative
management of both problems may be undertaken. In the case of an
unstable pelvic ring disruption with a contaminated suprapubic
catheter, external fixation of anterior disruptions of the pelvis can
be used to supplement open or percutaneous treatment of the posterior
pelvic disruption.
considerable mortality, although more recent series have displayed
mortalities that are comparable to those for closed injuries (7,11,25,52,77). The principal sites for the open fracture and associated wounds are shown in Table 17.8. Irrespective of the mechanisms of injury (Table 17.9),
the presence of an open wound is usually due to tremendous energy
dissipation on the pelvic ring resulting in an unstable fracture
dislocation.
 |
|
Table 17.8. Principal Bony Sites and Associated Injuries in Open Pelvic Trauma
|
 |
|
Table 17.9. Causes for an Open Pelvic Fracture or Dislocation
|
open wound, a highly unstable pelvic fracture may be found for which
stabilization is indicated. In the past, internal fixation was felt to
be contraindicated in open fractures because of the increased risk of
infection. While external fixation remains the most common method of
stabilization, improved wound care and newer techniques have made
internal fixation an option. Several different strategies for
definitive pelvic fixation may be considered, depending on the type and
site of the wound, the magnitude of contamination, the fracture
pattern, and the resources of the surgical team (Table 17.10).
 |
|
Table 17.10. Alternative Methods of Pelvic Stabilization Based on Characteristics of the Open Fracture Wound
|
anal sphincter, or when it involves a viscus, a diversion colostomy is
necessary to divert feces away from the open wound. Other implications
of the impact of the open wound on global management are outlined in Table 17.11.
 |
|
Table 17.11. Impact of an Open Wound on Management Decisions
|
America and Europe is having a profound impact on pelvic fracture
management (29). This statistical trend is anticipated to be even more dramatic over the next half century (Table 17.12).
 |
|
Table 17.12. Anticipated Changes in the U.S. Over 65 Population
|
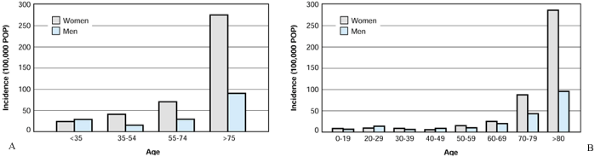
to violent trauma in young adults, currently more than 50% of pelvic
fractures occur in the over-60 population (Fig. 17.7), with a female predominance that reflects postmenopausal osteoporosis (46,55). The elderly experience higher mortality and late morbidity than younger patients with comparable injuries (Table 17.13) (29,34).
In the elderly person, the potential for intensive-care management and
the duration of hospitalization and rehabilitation are likely to be
much greater, resulting in increased costs.
 |
|
Table 17.13. Implications for Injury Patterns in the Elderly
|
 |
|
Figure 17.7. Incidence of pelvic fractures for the United States and Sweden with respect to sex and age. A:
Rochester, Minnesota, 1976–1985. (From Melton LJ, Sampson JM, Morrey BF, Ilstrup DM. Epidemiologic Features of Pelvic Fractures. Clin Orthop 1981;155:43.) B: Skaraborg County, Sweden, 1976–1985. (From Ragnarsson B, Jacobsson B. Epidemiology of Pelvic Fractures in a Swedish County. Acta Orthop Scand 1992;63:297.) |
are frequent cofactors that impact heavily on the management of the
pelvic fracture. Antecedent cardiac disease compromises the cardiac
reserve during the stressful early posttraumatic period and renders the
patient vulnerable to serious arrhythmia and myocardial infarction.
Posttraumatic atelectasis, possibly in association with multiple rib
fractures or a pneumothorax, is immeasurably aggravated by pretraumatic
pulmonary disease. Hepatic dysfunction in alcoholics may impair
coagulation and compromise the prognosis for retroperitoneal
hemorrhage. The trauma may dislodge a preexisting plaque in the common
or external iliac artery, resulting in a cold, pulseless limb that
requires medical or surgical intervention. With central neurologic
impairment, such as pretraumatic senility, intention tremor, or
generalized weakness, the rehabilitation after a pelvic fracture may be
greatly impeded. Not infrequently in the elderly, the
general
anesthetic required for initial care will be the only anesthetic that
is possible for the first 4–6 weeks of posttraumatic care because of
complications.
 |
|
Table 17.14. Geriatric Comorbidities That Frequently Impact on Pelvic Fracture Management
|
infirm have a higher incidence of intraoperative hemorrhage, wound
infection, and necrosis of flaps (22). External fixation pins tend to loosen quickly. Table 17.15 lists guidelines that can aid in the management of pelvic fractures in the elderly.
 |
|
Table 17.15. Fixation Strategies in the Elderly and Infirm
|
the force vectors and pelvic injury patterns also correlate with the
anticipated patterns of additional injuries to the abdomen, intrapelvic
contents, chest, and head, as well as with the potential for
significant hemorrhage. A single anteroposterior (AP) pelvic
radiograph, therefore, provides insight into the force vector and the
likelihood for co-injuries for which appropriate diagnostic tests can
be initiated promptly.
 |
|
Table 17.16. Principal Pelvic Fracture Patterns Based on the Vector of the Provocative Force
|
The crucial stabilizing ligaments extend from the sacrum, across the
sacroiliac (SI) joints and posterior; they transmit weight-bearing
forces either across the hip joints, into the lower extremities for
ambulation, or into the ischial tuberosities for sitting. The crucial
posterior SI ligaments stabilize the SI joints, along with the
iliolumbar,
sacrospinous,
and sacrotuberous ligaments. With its ring-like configuration, the
pelvis is intrinsically highly stable and resistant to deforming forces.
correlate with the vector and magnitude of the provocative blow and the
strength of the pelvic ring (Fig. 17.2). Subtle
changes in the force vector markedly alter the pattern of the
disruption. A direct lateral blow on the posterior ilium usually causes
a stable lateral compression injury with impaction of the sacral ala,
and accompanying unilateral or bilateral ramus fractures. A blow to the
anterior portion of the lateral ilium results in an internal rotational
moment that creates an unstable injury in which the ilium sustains a
vertical or crescent fracture with the sacral ala acting as a fulcrum (69).
With the rotational deformity of the ipsilateral hemipelvis, the sharp
edges of the ramus fractures can impale the bladder or occasionally the
bowel.
diastasis of the pubic symphysis and other manifestations of the injury
(Fig. 17.3). With a diastasis of 2 cm or less,
the symphyseal and diminutive anterior SI ligaments are disrupted while
the rest of the pelvic ligaments are spared. With a diastasis of 5 cm
or more, the sacrotuberous and sacrospinous ligaments are torn on one
or both sides and sagittal malrotation of the involved hemipelvis is
present. With a diastasis beyond 10 cm, the posterior SI ligaments are
torn so that the involved ipsilateral hemipelvis also displaces
posteriorly.
With vertical displacement of the pelvis, the ipsilateral lower lumbar
transverse processes are fractured. This is pathognomonic of this
injury.
Lumbosacral dissociation is caused by high energy falls, such as
endured by a suicidal jumper or a paratrooper who sustains a forceful
landing on his feet (62). In this insult,
usually a comminuted sacral fracture is greatly complicated by a
forward displacement of the first sacral body upon the fifth lumbar
vertebra.
 |
|
Table 17.17. Patterns of Sacral Fractures
|
 |
|
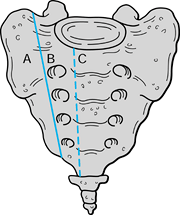
Figure 17.8. Pattern of sacral fractures. A: Transalar. B: Transforaminal. C: Central.
|
short-distance runners. The site of the involved fragment is highly
variable and includes the anterior superior and inferior spines and the
ischial tuberosity, caused by the sartorius, the rectus femoris, and
the hamstrings, respectively. While most of these heal uneventfully, a
few, especially the larger ones, progress to a symptomatic nonunion.
technetium bone scan or magnetic resonance imaging (MRI) may confirm
the diagnosis (18). The most common occult
fracture is a sacral injury for which overlying gas shadows may hamper
radiographic visualization. A computed tomography (CT) scan is helpful.
If the patient is hemodynamically unstable and has an unstable pelvis,
then early stabilization may be very important, as discussed below.
external bleeding, apply a pressure dressing. The pelvis can be quickly
and temporarily stabilized by wrapping a sheet tightly around it and
securing it with a clamp. On arrival in the emergency department, the
patient may be in a pneumatic antishock garment. Deflate it carefully
to avoid precipitous hypotension. If hypotension does occur, reinflate
the garment and transfer the patient to the operating room so that,
upon removal of the suit, immediate alternative surgical measures to
restore hemodynamic stability can be undertaken. The diagnosis of
intraabdominal hemorrhage can be made by ultrasound (45,54), peritoneal lavage, or minilaparotomy. Abdominal and pelvic CT scans are useful as well (3,63).
 |
|
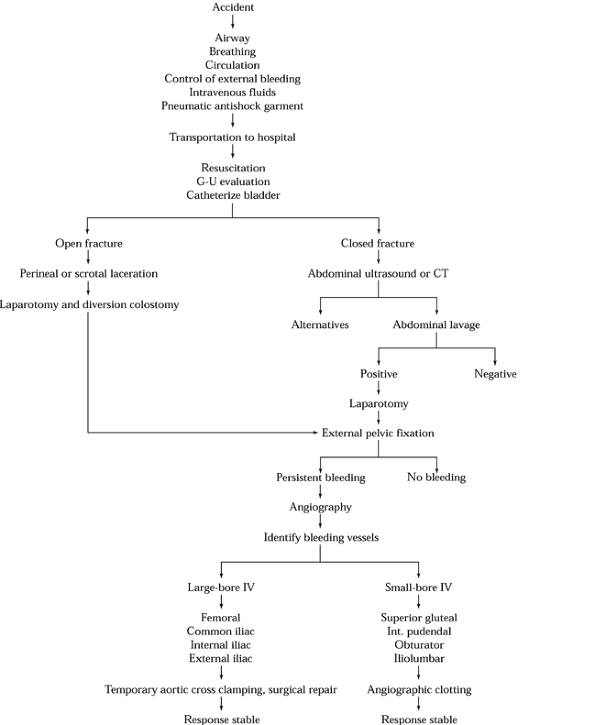
Figure 17.9. Algorithm that outlines the therapeutic measures for the control of hemorrhage in the patient with a pelvic fracture.
|
For initial emergent application, pelvic clamps have been devised that
permit rapid application and compression of the pelvis directly over
the lateral ilium at the level of the SI joints (16,17,51,52,76).
The clamp is intended for temporary application with subsequent
replacement by a suitable technique of internal fixation once
hemodynamic stability has been achieved. These devices have not been
popular because of potential complications from misapplication.
interstitial tissue pressure and provides a tamponade of the
retroperitoneal bleeding. It also markedly reduces the volume of the
true pelvis in which extravasated blood may accumulate. Reduction and
compression of the cancellous fracture surfaces reduces the rate of
bleeding. Nevertheless, contraindications to the use of external pelvic
fixation for the control of hemorrhage need to be clearly understood (Table 17.18).
With a bilaterally unstable posterior injury, the frame cannot control
the pelvic ring. Adding temporary longitudinal skeletal traction to the
affected side, combined with external fixation, may be of some help. In
the presence of iliac comminution and florid osteoporosis, the pins do
not achieve sufficient pelvic anchorage. In a small child, the
disproportionately small pelvis is not a realistic target for effective
anchorage of the pins.
 |
|
Table 17.18. Relative Contraindications to External Pelvic Fixation for Control of Acute Hemorrhage
|
use angiographic embolization of autologous blood clots or Gelfoam
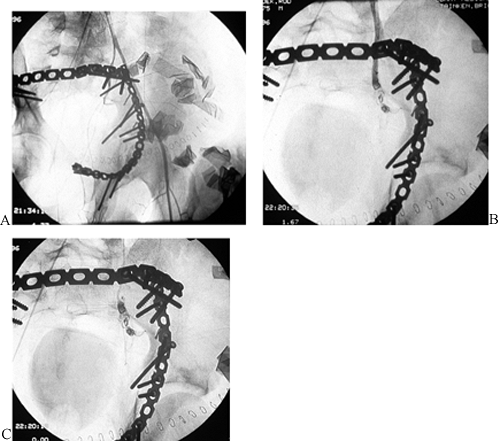
clots in combination with the insertion of a stent (1). An example is displayed in Figure 17.10.
Angiographic embolization can be done without violating the peritoneal
barrier and with minimal risk of infection. It is unsuitable, however,
for the management of venous bleeding, and it requires appropriate
radiographic resources. In dire circumstances, it can be used to
control bleeding of relatively large vessels, such as the internal
iliac artery. Such a procedure, however, possesses a considerable
morbidity, including necrosis of the gluteal muscles.
 |
|
Figure 17.10.
Arteriographic views of a 21-year-old man after a pelvic fracture with acute open reduction and internal fixation (ORIF) of sacral and left acetabular fractures. Persistent hemorrhage from the left hypogastric artery is identified arteriographically and secondarily controlled with a standard gel foam embolus. A: Arteriogram. B: During embolization. C: After embolization. |
greater than 5 mm in diameter, such as the femoral and common iliac
arteries, operative intervention is usually necessary. At least three
separate strategies, or various combinations of these, may be
appropriate (Table 17.19). A direct repair of a
major artery has a high likelihood for a successful result. Aortic
cross-clamping is a desperate measure that may provide a brief period
of tolerable ischemia while a source of uncontrolled hemorrhage is
identified and controlled. Acute retroperitoneal packing can also be
effective and may permit a concomitant or sequential open reduction and
internal fixation of the pelvis (66).
 |
|
Table 17.19. Methods of Intraoperative Vascular Control of Major Intrapelvic Hemorrhage
|
output as a crucial determinant of adequate volume resuscitation. In
the presence of a major pelvic fracture, urinary catheterization
requires special consideration (Table 17.20). The incidence of concomitant injury to the bladder or urethra is about 20% (52,53,60,67,73).
A Foley catheter can be placed directly into the bladder of a female
patient. In male patients, first obtain a urethrogram if there is any
suspicion of a urethral injury. Attempts to pass a catheter blindly
through a partially disrupted male urethra can aggravate a partial tear
and result in a stricture, incontinence, or impotence. If the
urethrogram indicates the passage of contrast medium into the bladder
without extravasation, advance the catheter into the bladder. Then perform a cystogram to exclude a rupture of the bladder.
 |
|
Table 17.20. Urologic Management for a Pelvic Fracture Victim
|
the kidneys and ureters. In the typical pelvic fracture victim, a large
pelvic hematoma usually distorts the image of the bladder. The hematoma
requires no specific treatment, although it may indicate a large blood
loss and the need for supplementary fluid or blood replacement. If a
urethral injury is identified, a suprapubic cystoscopy may be
indicated. This procedure can be technically difficult in the presence
of a large pelvic hematoma.
visible stents have been inserted primarily into the site of the
urethral disruption. Also, magnetic catheters have been devised, which
permit simultaneous urethral and suprapubic insertions (53,60). After the magnetic coupling of the catheters at the site of the rupture, advance the
urethral catheter into the bladder and remove the suprapubic catheter.
The newer methods that eliminate any suprapubic source of sepsis are
superior to the more traditional suprapubic catheter.
trauma are likely to undergo a lengthy period of nutritional depletion
following the acute resuscitation. Nutritional demands are
exceptionally great, and oral dietary intake is limited by ventilatory
support and multiple visits to the operating room. Certain patients,
notably those who are alcoholic and others with peculiar voluntary
dietary restrictions, may be nutritionally depleted at the time of
injury. Others, especially those with morbid obesity, are vulnerable to
a rapid onset of malnutrition following admission, which may not be
recognized by the therapeutic team. For these reasons, a sound scheme
of nutritional support is necessary for each patient (6).
Conventional radiographs may not reveal all pelvic injuries, especially
when the triradiate cartilage is involved. A three-dimensional (3D) CT
is especially useful in this scenario (30,44).
External fixation is greatly hampered by the small size of the
pediatric pelvis, but a double hip spica cast works well. Flexion of
the hips and knees aids indirect reduction of the pelvis by suitable
rotation of either hemipelvis. Occasionally, for an unstable and
displaced injury, especially one that involves the triradiate
cartilage, an open reduction and internal fixation is indicated.
 |
|
Table 17.21. Implications of a Pediatric Pelvic Fracture for Clinical and Radiographic Evaluation and Treatment
|
Because of the predilection of the elderly for diverse pretraumatic
comorbidities, the homeostatic compensatory mechanisms to cope with
multiple trauma may be heavily compromised. Similarly, the patient may
not possess the reserves to cope with reconstructive surgery. In the
presence of osteopenic bone, pelvic imaging may be hampered,
particularly in a case of an insufficiency or pathologic fracture. The
use of a CT scan, a technetium bone scan, or an MRI merits
consideration to confirm the site(s) and nature of the injury (18).
A prior history of pelvic irradiation therapy may provide a
predisposition for an insufficiency fracture. If prior heavy
irradiation of the pelvis is documented, then the role for extensive
open surgical reconstruction is markedly diminished, with greatly
increased surgical risk and potential for postoperative wound
dehiscence. Overall, in the elderly, the role for minimally invasive
reduction strategies and percutaneous fixation is increased, while the
place for full open reduction and external fixation is diminished.
 |
|
Table 17.22. Implications of a Geriatric Pelvic Fracture for Clinical and Radiographic Evaluation and Treatment
|
Try to determine the direction of the injuring force from the history,
and by inspecting for sites of contusion or ecchymosis. The history of
a high-energy motor-vehicle or industrial accident increases the
likelihood of a major unstable injury and concomitant visceral or
neurovascular insults. Examine the pelvic region for evidence of
asymmetry or instability, or the presence of an open wound. A
laceration
in the groin, scrotum, or perineal region or of the vagina and rectum
is highly suspicious of an open pelvic fracture. An apparent deformity
of the lower extremity in the absence of a fracture in the lower limb
may indicate a pelvic fracture.
 |
|
Table 17.23. Clinical and Radiographic Assessment of the Pelvic Fracture Victim
|
initial radiographic assessment of the pelvis to an AP view. Once
hemodynamic and other urgent considerations permit, obtain additional
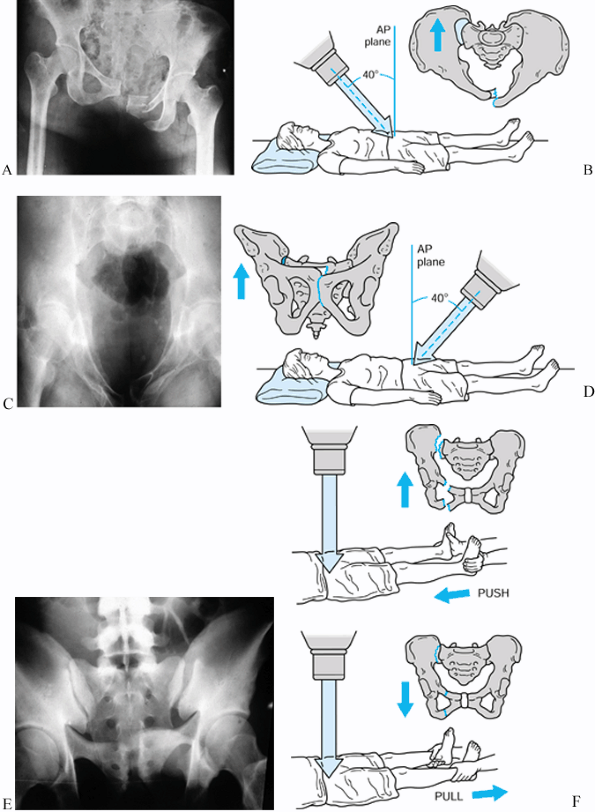
radiographic views so that the injury can be precisely characterized (Fig. 17.11).
At least three views are required: AP, inlet, and outlet. To obtain an
inlet view of the supine patient, direct the x-ray beam from the head
to the midpelvis at about 45° with respect to the radiographic table or
45° from the vertical reference axis. Such a projection is
perpendicular to the pelvic brim and illustrates the true pelvic inlet
as well as anteroposterior displacement of a pelvic disruption. To
obtain an outlet projection of a supine patient, direct the beam from
the foot to the pubic symphysis at 45° with respect to the radiographic
plate. The outlet projection discloses superior displacement of the
posterior half of the pelvis and either superior or inferior
displacement of the anterior portion of the rami. Apparent limb-length
discrepancy originating from elevation of the hip joint secondary to a
rotational displacement is highlighted, along with avulsion fractures
of the transverse processes of the lower lumbar vertebrae or ramus
fractures.
 |
|
Figure 17.11. Radiographic and schematic views of conventional pelvic images. A: AP radiographic view. B: Schematic method for inlet view. C: Radiographic inlet view. D: Schematic method for outlet view. E: Radiographic outlet view. F: Schematic assessment for pelvic instability.
|
disruption, obtain supplementary Judet or oblique obturator and iliac
views. Obtain these views by rolling the injured patient carefully from
one side to the other to provide 45° views. Occult pelvic instability
may be detected by AP radiographs before and after the application of
longitudinal traction on the relevant lower extremity (Fig. 17.11).
sites of pelvic disruption, displacement, and comminution. Its foremost
role is to clarify posterior disruption of a pelvic ring fracture. A
sacral fracture that is virtually invisible on plain radiographs is
readily seen on CT. The degree of separation and instability of a SI
joint or sacral fracture is evident. At a minimum, obtain five standard
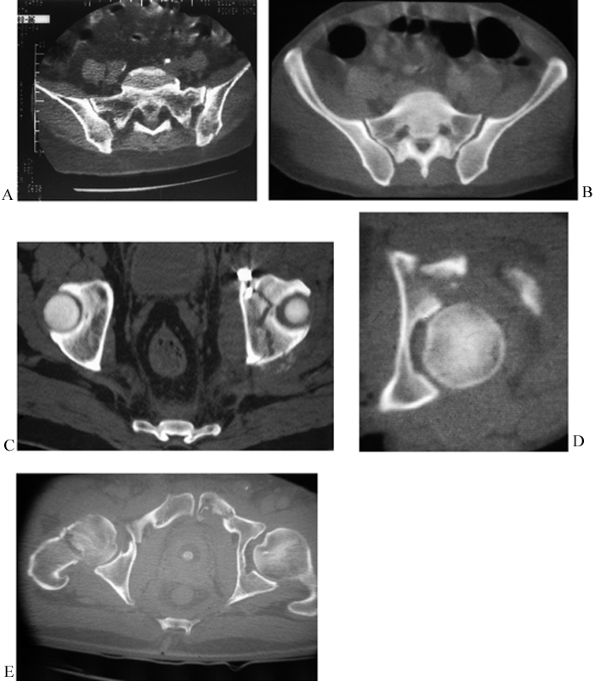
transaxial sections at 2 cm intervals (Fig. 17.12).
The most superior section demonstrates the iliac wings and the adjacent
SI joints. A second section displays the principal part of the sacrum
and the adjacent SI joints. The third section projects the dome of the
acetabulum with a circular cross section. The fourth section transects
the midacetabular region, where the femoral head opposes the anterior
and posterior columns. The most inferior section reveals the inferior
pubic ramus and the ischial tuberosity at the level of the greater
trochanter.
 |
|
Figure 17.12. Standard pelvic CT images at 2 cm intervals. A: SI joints with stable sacral alar lateral compression fracture. B: Sacral body and SI subluxation. C: Acetabular dome with transverse fracture. D: Mid-acetabular region with posterior wall fracture. E: Symphysis and rami.
|
of a comminuted, displaced pelvic fracture requires an accurate 3D
radiographic perspective to define the optimal method or surgical
approach, open reduction, and stabilization. When reviewing the
conventional two-dimensional (2D) radiographs and CT scans, even an
experienced surgeon has difficulty with the extensive mental
integration needed to realize a 3D image. Computer programs now can
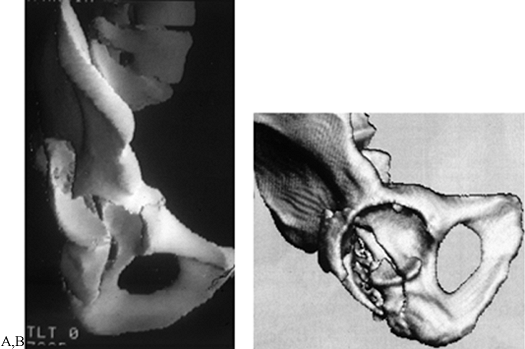
produce 3D surface reformations, or so-called 3D CT images (44), from sets of contiguous axial computed scans of the pelvis (Fig. 17.13).
While the images may be unavailable for truly emergent cases, they can
be obtained within 1–12 hours after the arrival of the patient in the
radiology suite. Document vascular injuries either by a conventional
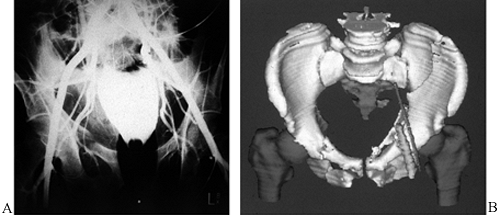
arteriogram (52) or a 3D CT with a prior insertion of radiopaque catheters into the relevant artery and vein (Fig. 17.14) (20).
 |
|
Figure 17.13. Three-dimensional CT scans of two hemipelves. A: A posterior T-type fracture. B: Disarticulated “dome” view of posterior column–posterior wall fracture.
|
 |
|
Figure 17.14. Arteriographic studies of pelvic fractures. A: Conventional arteriograph. B: 3D CT of a left both column acetabular fracture with radiopaque catheters in the left external iliac vessels.
|
and the degree of pelvic disruption (Table 17.24). Injuries may result from anteroposterior compression, lateral compression, vertical shear, or combinations of these forces (Fig. 17.2).
This classification provides insight into the nature of the injury, the
morbidity, the potential sites of disruption, and the degree of pelvic
instability; and it provides some therapeutic guidelines.
 |
|
Table 17.24. Pennal and Tile Classification of Pelvic Fractures
|
external rotation deformity, results from a blow that strikes the
posterior ilium or the anterior pelvis to disrupt the symphysis and the
anterior SI ligaments of one or both SI joints. Usually, the crucial
posterior SI complex is spared so that the injury is vertically stable,
although in a few of these cases the posterior SI ligaments are
violated to produce an unstable hemipelvis.
to the lateral ilium. A stable impacted fracture of the sacrum or,
rarely, the adjacent SI joint may result. In the presence of an
anterolateral or posterolateral force, however, the ipsilateral
hemipelvis rotates inwardly around the site of the posterior
disruption, involving the SI joint or the adjacent sacrum or ilium.
While they are characterized as “rotationally unstable,” in actuality
the injuries may be completely stable, rotationally unstable, or
unstable in a rotational and posterior direction. Usually, the
contralateral rami are fractured, or sometimes the ipsilateral pubic
rami or all four rami are disrupted.
unstable and have complete instability of the posterior ligament of one
or both SI joints. Most of the patients are victims of falls from a
great height or high-speed crashes. Usually, the associated anterior
ring disruption is a diastasis of the symphysis pubis. Supplementary
radiologic findings may be consistent with an unstable lateral
compression or a vertical shear injury with avulsion fractures of the
sacrotuberous and sacrospinous ligaments from the ischial spine of the
adjacent sacrum. Another possible associated injury is an avulsion of
the transverse processes of L-4 or L-5 at the site of the origin of the
iliolumbar ligaments. With the marked posterior displacement, which is
well documented in the inlet view or CT scan, the likelihood for
profuse hemorrhage secondary to injuries of the superior gluteal
vessels is high. An uncommon vertical injury has to be carefully
distinguished from the much more commonly encountered pattern with a
rotational deformity in a sagittal plane and possibly in a coronal
plane. Serious hemorrhage and neurologic complications are uncommon
with the latter type.
classified them by the site of injury as a combination of two or more
of five elementary patterns. This method provides an anatomic
description of the sites of involvement and eliminates the mechanism of
injury, which usually is poorly defined and generally irrelevant to the
treatment. The five sites of injury are listed in Table 17.25. Denis et al. (12) also classified sacral fractures by the region of involvement, using transalar, transforaminal, or central (Fig. 17.8).
 |
|
Table 17.25. Classification of Pelvic Fractures after LeTournel and Judet
|
classification scheme based on an anatomic assessment of
multiple-trauma victims in whom pelvic injuries were examined at
autopsy (Fig. 17.3; Table 17.26).
The clinical significance of the Bucholz scheme was a correlation with
therapeutic recommendations. While a group 1 injury responded well to
conservative management, a group 2 disruption usually required closed
reduction with external or internal fixation. Uniformly, a group 3
pattern required an open reduction with internal fixation of the
anterior and posterior injury sites.
 |
|
Table 17.26. Bucholz Classification of Anteroposterior Compression or External Rotational Injury Patterns
|
classification scheme has been devised by the collaborative efforts of
Arbeitsgemeinschaft für Osteosynthesefragen (AO), the Americn Society
of Internal Fixation (ASIF), the Orthopaedic Trauma Association (OTA),
and Societé Internationale
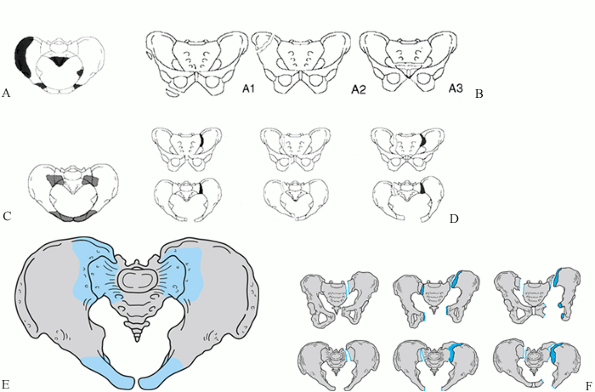
de Chirugie Orthopaedic et Traumatologie (SICOT) (71). The AO/ASIF–OTA–SICOT scheme has gained acceptance as the definitive method of classifying pelvic fractures (Fig. 17.15; Table 17.27), particularly for clinical research.
 |
|
Table 17.27. Comprehensive Classification Scheme of Pelvic Fractures
|
 |
|
Figure 17.15. Schematic views of the AO–OTA–SICOT alphanumeric classification of pelvic fractures. A: Sites of type 61-A stable injuries. B: Type 61-A subcategories by injury sites. C: Sites of type 61-B partially unstable injuries. D: Type 61-B subcategories by injury sites and displacement. E: Sites of type 61-C completely unstable injuries. F: Type 61-C subcategories by injuries sites. (From Tile M, Helfet DL, Kellam JF, et al.Comprehensive Classification of Fractures in the Pelvis and Acetabulum. Berne, Switzerland: Maurice E. Müller Foundation, 1995.)
|
diagnostic studies have been completed, outline a definitive management
protocol. The management priorities are established through discussions
between various surgical teams. If a hemodynamically stable patient has
an unstable pelvic fracture, definitive surgical stabilization can be
undertaken on the day of the acute injury. In the multiple-trauma
victim, however, a considerable delay may be necessary between the time
of the injury and the first realistic date for internal fixation. In
either event, the goals of management include anatomic and functional
objectives (Table 17.28). Also, pelvic
stabilization should minimize the likelihood of late problems such as a
painful nonunion or malunion of the pelvis.
 |
|
Table 17.28. Objectives of Definitive Pelvic Fracture Management
|
controversial, the authors’ experience has led to an algorithm that
facilitates the selection of the optimal stabilization technique for
almost any type of pelvic ring disruption (Fig. 17.16).
While the algorithm provides a number of viable therapeutic options,
the need for internal fixation has increased in the 1990s as internal
fixation provides much more stable fixation than external fixation.
With internal fixation, the frequency of postoperative follow-up is
decreased, whereas external fixation requires ongoing supervision,
including pin-track care. The placement of percutaneous lag screws
provides a stable method of internal fixation that requires even less
surgical exposure than the application of external fixation (37).
In fact, once a surgeon gains familiarity with techniques of
percutaneous surgical stabilization, the indications for external
fixation as a definitive method of pelvic fixation diminish rapidly.
 |
|
Figure 17.16. Algorithm for the optimal stabilization techniques for various patterns of pelvic ring disruption.
|
minimally displaced as a result of a low-velocity lateral compressive
force. Begin with bed rest and mobilize the patient
as soon as possible. Progress weight bearing as tolerated. Most patients bear full weight in 3–6 weeks.
injuries, there is a simple but displaced pelvic fracture, and the
posterior SI ligaments and the sacrum are intact; this scenario is
quite common. Treatment options include plate fixation, a simple
anterior external frame, or percutaneous fixation of the SI joint.
Perform surgical stabilization when the diastasis of the pubic
symphysis is greater than 2 cm. If an emergency laparotomy is done,
stabilize the pubic symphysis with a plate. Plate fixation also can be
used for unilateral or bilateral anterior ramus fractures using a
midline vertical incision (24). As an
alternative, external fixation can be used to manage an unstable
anterior injury with minimally displaced ramus fractures. Where the
rami are significantly displaced and impinging on skin or genitourinary
organs, perform an open reduction. This minimizes the risk of a
persistent nonunion or malunion that can lead to pain and dyspareunia.
Where a pubic diastasis is open, or a colostomy or a suprapubic
catheter is present, consider a closed reduction with percutaneous
fixation of the ipsilateral SI joint.
accompanied by superior migration of the hemipelvis, first apply
skeletal traction to correct the vertical displacement. Two alternative
strategies are available. An open reduction and plate fixation of the
symphysis or rami restores the alignment of the pelvic ring, so that
percutaneous fixation of the posterior injury is feasible. If
realignment of the anterior pelvic ring does not align the SI joint,
extend the incision to an ilioinguinal approach and openly reduce and
internally fix the SI joint.
perform a posterior approach and plate the fracture, avoiding
compression of the sacral nerve roots. If possible, add supplementary
percutaneous screw fixation into the S-1 sacral body to avoid the need
for anterior fixation (36). If percutaneous
fixation is impossible, anterior column fixation or external fixation
are alternative measures, although the former is preferred.
Most pelvic fractures with bilateral unstable SI dislocations and pubic
symphysis diastasis are usually caused by vertical shear injuries. For
these highly unstable injuries, use a bilateral ilioinguinal approach
to achieve open reduction of the symphysis and SI joints
simultaneously. In certain instances, where skeletal traction has
provided an accurate reduction of one or both SI joints, it is feasible
to undertake percutaneous fixation of the SI joints after fixation of
the symphysis. As an alternative, reposition the patient in a prone
fashion for a posterior approach to the SI joints and perform either
lag-screw fixation or transiliac plate fixation.
 |
|
Table 17.29. Patterns of Complex Pelvic Ring Disruptions
|
a pubic diastasis with an iliac “crescent” fracture (i.e., a lateral
compression injury), suitable immobilization may be achieved in one of
two ways. First, through an iliofemoral approach, perform an open
reduction of the lateral ilium with lag screws and plates. Then employ
an anterior vertical or Pfannenstiel approach for the anterior injury.
If these reductions are accurate, perform a percutaneous fixation of
the contralateral SI joint. Alternatively, provided that the imaging
technique is mastered, a retropubic screw can be inserted via a medial
entry site to stabilize both of the superior ramus fractures (61).
injury sites, a variety of options are available. In the presence of an
open posterior wound, an anterior approach or percutaneous
stabilization of an SI dislocation is indicated; in the presence of a
left lower quadrant colostomy, however, a posterior approach to an SI
dislocation may be preferred.
of reduction can be initiated at the anterior or the posterior injury.
The least invasive approach is to reduce the pelvic ring and plate it
anteriorly, followed by percutaneous posterior fixation. By 3 weeks
after the injury, reduction of the entire pelvic ring through a limited
approach is more difficult and may be impossible. In this situation, a
single or bilateral ilioinguinal exposure permits a simultaneous open
reduction of the anterior and posterior disruptions. Reduction of an SI
joint is easier from the anterior approach than from posteriorly. The
stability of anterior SI plating is comparable to that obtained with
lag screws across the SI joint. Simultaneous reduction of the anterior
and posterior disruptions through an ilioinguinal approach is more
efficient and avoids a major liability of sequential anterior and
posterior exposures: An imperfect anterior reduction may produce
posterior malalignment that cannot be corrected.
For the classic pubic symphysis diastasis with an SI dislocation,
definitive fixation of the two injuries is well accepted. If the
posterior injury is a simple displaced iliac fracture, isolated
fixation of the ilium suffices. If the accompanying anterior injury,
such as a symphysis diastasis, is not repaired, typically there is a
minor residual external rotation of the hemipelvis; usually this is not
of functional significance. If the iliac fracture is comminuted, then
anterior and posterior fixation is indicated.
multiple pubic rami fractures, may be sufficiently stabilized solely by
percutaneous cannulated screws across the sacrum. Within 3–4 weeks,
callus forms on the ramus fractures so that walking with crutches can
be started. This
strategy
avoids the application of an anterior external fixator or more
aggressive surgery. If the rami fractures are markedly displaced and
unstable, then plate them through a lower midline abdominal incision.
comminution or osteoporosis, more stable fixation is indicated. The
pelvic ring can be perceived as a system of three columns: the rami,
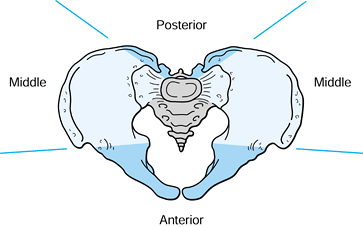
the sacral body, and the posterior sacral elements (36) (Fig. 17.17).
For a typical unstable injury, surgical fixation of two columns is
recommended. For the most unstable injury patterns or in patients with
osteoporosis, fixation of all three columns is advisable. Examples
include unstable fractures fixed late, osteoporotic sacral nonunions or
malunions, and displaced pathologic pelvic ring disruptions.
 |
|
Figure 17.17. Schematic view of the pelvic columns to aid in the fixation of unstable injuries.
|
or symphysis diastasis complicating an acetabular fracture, with or
without propagation into the ilium, is another consequence of a lateral
compression injury. First, undertake an open reduction and internal
fixation of the acetabulum. For an ipsilateral SI fracture, fix it
through the same exposure or do percutaneous fixation. Then stabilize
the rami with plates or retrograde screws, or external fixation.
including unilateral or bilateral acetabular fractures, bilateral SI
disruptions or other equivalent posterior injuries, and bilateral ramus
fractures, with or without a diastasis of the symphysis (75).
In most of these patients, virtually all of the principal ligamentous
supports of the pelvic ring are severely compromised. As a rule, each
site of pelvic disruption must be stabilized with internal fixation.
emergency treatment for hemorrhage and pain control is well
established, the optimal timing for delayed fixation of closed
fractures remains unclear. Whenever the patient is unstable or at risk
for excessive hemorrhage, defer surgery for 24–48 hours after the
injury. Further delay is likely to result in pulmonary, thromboembolic,
and urologic complications. When the patient’s general condition is
satisfactory, early surgical intervention permits the most rapid
recovery and minimizes the risk of complications.
systemic problems just discussed, highly contaminated open fractures,
concomitant infection, inadequate bone quality or bone stock, a
severely injured soft-tissue envelope, and a surgical team that is not
experienced or an operating room that is not equipped for this
difficult surgery.
It is completely radiolucent, and the absence of a central post
enhances imaging with the C-arm. Newer models provide longitudinal
and/or lateral skeletal traction.
 |
|
Figure 17.18. A Jackson radiolucent operating room table and modern C-arm image intensifier.
|
in. or 12 in. field size and high-resolution capabilities, are a major
technological advance for pelvic reconstruction. The integral software
package permits magnification of
focal
areas of interest. While the Siemens, Philips, and OEC-Toshiba models,
as well as others, are excellent for pelvic applications, the
OEC-Toshiba has a unique advantage. Its C-arm gantry possesses an extra
articulation that greatly simplifies the iliac and obturator oblique
views.
intensification is necessary. The radiographic technician needs to know
the AP, inlet, outlet, iliac, and obturator views, along with the
direct lateral view of the sacrum (37,61).
Once the patient is positioned on the operating table, and prior to
draping, rehearse the views to be sure that they can be obtained.
Correct any obstruction of the image by an ill-positioned upper
extremity, arm board, or other object.
bolsters. Drape with adhesive drapes and use skin staples to avoid the
use of towel clips.
Instrumentation and implants specifically designed for pelvic surgery
are essential. Currently, the Synthes instrumentation (Synthes, Inc.,
Wayne, PA) is the most complete. Tenaculum forceps are available in
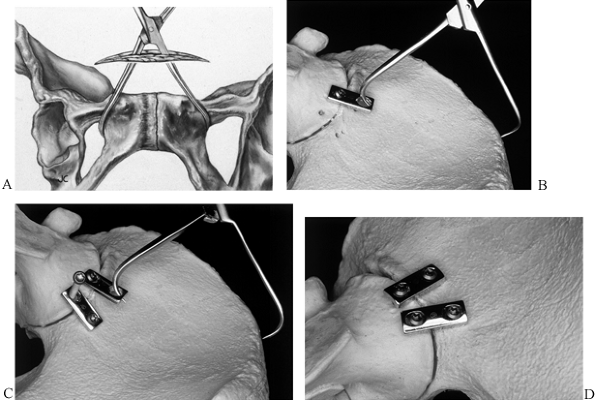
diverse sizes with straight and curved jaws (Fig. 17.19, Fig. 17.20 and Fig. 17.21).
The special pelvic forceps are exceptionally large with respect to the
length of the jaws and handles, and they provide a high degree of
mechanical advantage. The symmetrical King Tong and the asymmetrical
Prince Tong are the most useful models.
 |
|
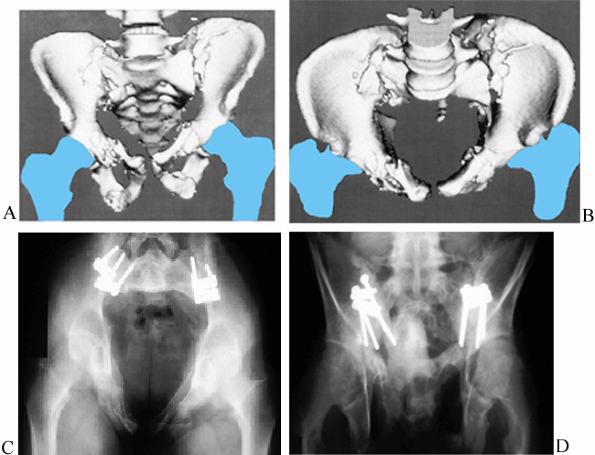
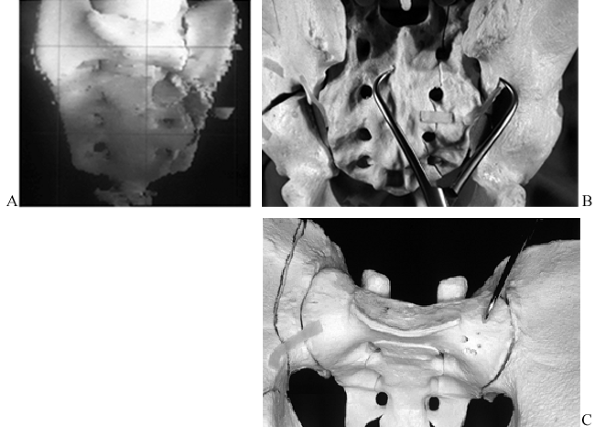
Figure 17.19. The use of tenaculum forceps for pelvic reductions. A: Symphysis pubis. B: Anterior SI joint in conjunction with a plate. C: SI joint after stabilization with a second plate. D: After plate fixation and removal of the forceps.
|
 |
|
Figure 17.20. Bilateral sacroiliac dislocation in an 18-year-old man in whom anterior SI open reductions were performed. A: Anterior 3D CT view. B: Inlet 3D CT view. C: Postoperative inlet view. D: Postoperative outlet view.
|
 |
|
Figure 17.21. Fixation of a sacral fracture with a tenaculum forceps. A: 3D CT with displaced alar fragment. B: Schematic view with reduction of the ala from a posterior approach. C: Posterior reduction of a transforaminal fracture.
|
 |
|
Table 17.30. General Strategies and Instrumentation for the Reduction of Pelvic Fractures
|
devices are available that provide unique attributes for stabilization
of the pelvis (Table 17.31). Of the
small-fragment screws, the 4.0 mm fully threaded cancellous design
affords excellent purchase in the pelvis. For the crucial
load-bearing
portions such as the SI joints, we prefer the larger core-diameter of
the 4.5 mm cortical or 6.5 mm cancellous screws. The strength of
fixation is exponentially related to the length of the thread in bone (42).
Wherever possible, we use longer screws. For example, where the
available bone stock is limited, such as in the superior pubic rami,
long screws of smaller diameter provide more effective fixation than
shorter ones of a larger diameter.
 |
|
Table 17.31. Principal Fixation Devices for Internal Pelvic Fixation
|
utilize corresponding guide wires with a range of diameters between 2.8
and 3.2 mm (37). The 2.8 mm wires are readily
inserted but bend easily, whereas 3.2 mm wires are significantly
stiffer, but because of greater frictional forces during their
insertion they can heat up and cause thermal damage. We prefer 2.8 mm
wires, although many pelvic surgeons prefer the larger wires.
threaded-tip guide wire or a drill bit. Threaded-tip guide wires
usually remain in position when the drill is removed, whereas drill
bits tend to back out. Threaded-tip guide wires provide relatively poor
feedback to the surgeon,
which
can lead to accidental penetration of the adjacent soft tissues,
threatening important structures. Drill bits provide better feedback,
although they are more likely to break during insertion.
self-tapping, as this eliminates the need for drilling and tapping. The
risk for injury to soft tissues is minimized by using an oscillating
power drill. In the dense pelvic bone of young adult men, partial
drilling and tapping may be necessary to avoid broken implants.
injuries such as an SI joint separation. For anterior SI fixation, use
two of the two-hole 3.5 mm or 4.5 mm plates. The narrower span between
the 3.5 mm holes is ideal for SI fixation and is consistent with the
use of 4.5 mm cortical screws (Fig. 17.19 and Fig. 17.20).
Typically, the displaced ilium is posterior and superior to the
adjacent sacrum. Anchor the plates to the sacrum with a screw. With the
superior displacement corrected, place a screw in the ilium through the
plate. Tightening the screw reduces the ilium to the sacrum. In
contrast, highly flexible reconstruction plates (Fig. 17.22)
are used where the objective is to contour the plate to the underlying
curved surface of the pelvis without displacement of the fracture, such
as along the iliac crest and true pelvic brim. The 4.5 mm and some
steel 3.5 mm reconstruction plates are too stiff for this application.
They must be contoured perfectly to the bone prior to insertion of the
screws. We prefer the more flexible, straight 3.5 mm reconstruction
plates, particularly those of titanium.
 |
|
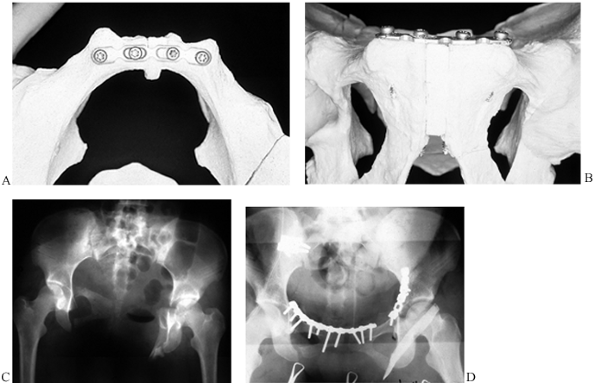
Figure 17.22. The use of a flexible reconstruction plate on the symphysis pubis and rami. A: Model with inlet view. B: Model with outlet view. C: AP radiograph with diastasis bilateral ramus fractures and right SI dislocation. D: Postoperative AP view with anterior plates and anterior SI fixation.
|
It can be contoured to fit directly on the irregularly curved surface
of the sacrum. In large men, this plate also can be used for fixation
of the pubic symphysis.
buttress thin and comminuted or osteopenic bone (19)
on the quadrilateral, posterior, and anterior surfaces of the
acetabulum and the posterior surface of the sacrum. The most useful
length is a three-hole, 3.5 mm one-third tubular plate. Fashion the
hooks at one end of the plate by cutting the end of the plate obliquely
through the middle of the screw hole and then bending the resulting
tines into hooks using a large needle holder. Bend a gentle convex
curve into the plate so that the hooks engage the bone and buttress the
fracture as the central screw is tightened. The hooks must not impale
important adjacent structures.
While conventional cable passers can be used, a useful tool for passing
along the inner pelvic wall is a Statinski vascular clamp. The
insertion of passing tools is hazardous, particularly in the presence
of a marked pelvic deformity or delayed surgery, where neurovascular
structures may be malpositioned and tethered to the bone, making them
more vulnerable to traction injury or a laceration. Cables also may be
threaded through the cannulation of screws as a way to augment screw
fixation in osteopenic bone (Fig. 17.24).
 |
|
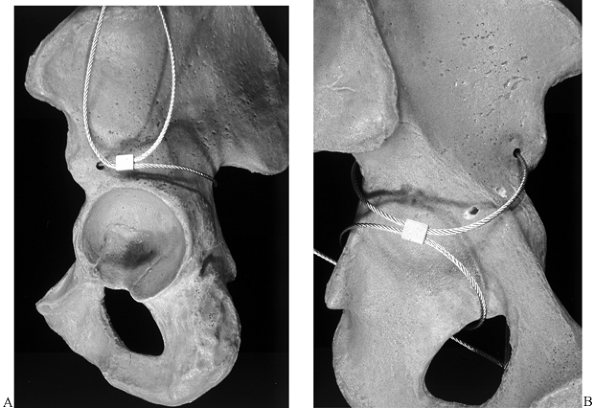
Figure 17.23. Cable fixation of the pelvis. A: Fixation from the greater sciatic notch to the anterior inferior iliac spine (AIIS). B: Buttressing the quadrilateral surface.
|
 |
|
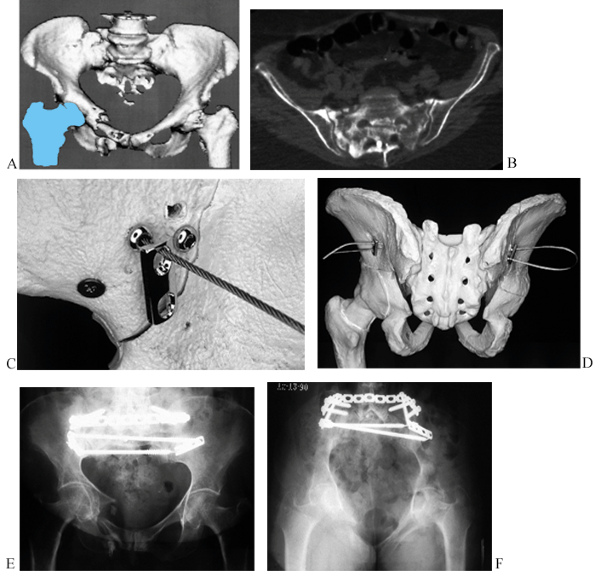
Figure 17.24. Multiple views of a bilateral sacral insufficiency fracture in a 78-year-old woman after irradiation therapy. A: 3D CT anterior view. B: CT scan of the sacrum. C: Screw heads of cannulated screws with a cable and a two-hole plate at the level of S-1. D: Posterior view after passage of the cable. E: Postoperative AP view. F: Postoperative inlet view.
|
While mechanically effective, the reduction must be anatomic. Rods are
usually symptomatic postoperatively and require removal. We prefer
transverse plate fixation rather than rods, as plates provide more
predictable fixation with a low morbidity, and they rarely need removal.
risk of iatrogenic neurologic injury, especially with respect to the
peroneal division of the sciatic nerve, lumbosacral
plexus,
and the sacral nerve roots. Formerly, somatosensory evoked potential
(SSEP) monitoring was popular, but continuous electromyographic (EMG)
motor monitoring has proven to be better (23,72).
Disadvantages of SSEPs include the need for highly trained personnel, a
significant incidence of spurious results, and a latent period of
potentially up to a few minutes between an intraoperative injury to a
sensory nerve and a significant alteration of the SSEP signal.
Continuous EMGs have the advantage of reflecting instantaneously any
compromise to a motor nerve, with a minimum of false negative results.
The method also is simpler than SSEPs.
or labial electrodes, but we prefer continuous EMGs for monitoring the
peroneal division of the sciatic nerve. While both techniques can be
used to monitor sciatic nerve function, it is not clear that the use of
both methods is better than the use of EMGs alone.
approaches to the pelvis. The following description focuses on specific
applications for the pelvic ring, including the authors’ preferences.
The available approaches are outlined in Table 17.32.
 |
|
Table 17.32. Pelvic Surgical Approaches
|
the symphysis, although a midline lower abdominal incision provides a
comparable view. For unilateral or bilateral ramus fractures, the
midline lower abdominal incision provides a minimally invasive and
rapid approach. This approach provides visualization of the pelvic brim
to the SI joint and the entire quadrilateral surface. It has a lesser
morbidity than the ilioinguinal approach and achieves a comparable
visualization. If the rami and lateral ilium need to be exposed, the
midline lower abdominal approach can be combined with a limited
incision along the iliac crest for what has been termed the
ilioanterior approach (Fig. 17.25) (24)
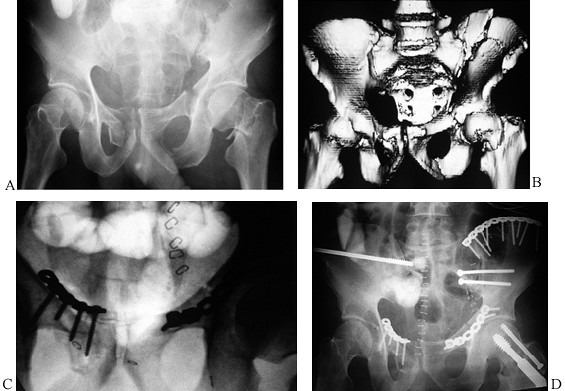 |
|
Figure 17.25.
Multiple views of bilateral ramus fractures, right SI disruption, and left SI fracture dislocation where an ilioanterior approach was used along with an iliofemoral exposure for the left-sided crescent fracture. A: Preoperative AP view. B: Preoperative anterior 3D CT view. C: Intraoperative outlet view. D: Postoperative AP view. |
straight longitudinal incision to a curvilinear incision. The
transverse presacral incision (Fig. 17.26 and Fig. 17.27) is useful when exposure of the sacrum and both SI joints is needed (38,39).
Cosmetically, it is superior to two parallel vertical incisions, as
plastic reconstruction of a wound slough from the latter is
complicated. Although a transverse incision is cosmetically superior to
a midline vertical approach, a proximal extension to the upper lumbar
spine requires a vertical incision (52).
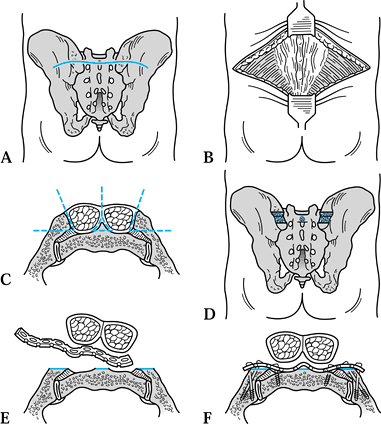 |
|
Figure 17.26. The transverse presacral approach. A: Cutaneous incision. B: Exposed posterior iliac crests, gluteus maximus, and paraspinous muscles. C: Bilateral elevation of the paraspinous muscles and preparation of the notches in the posterior superior spines. D: Subperiosteal tunnel across the back of the sacrum for plate insertion. E: Insertion of the inverted plate. F: Cross section of the screw fixation.
|
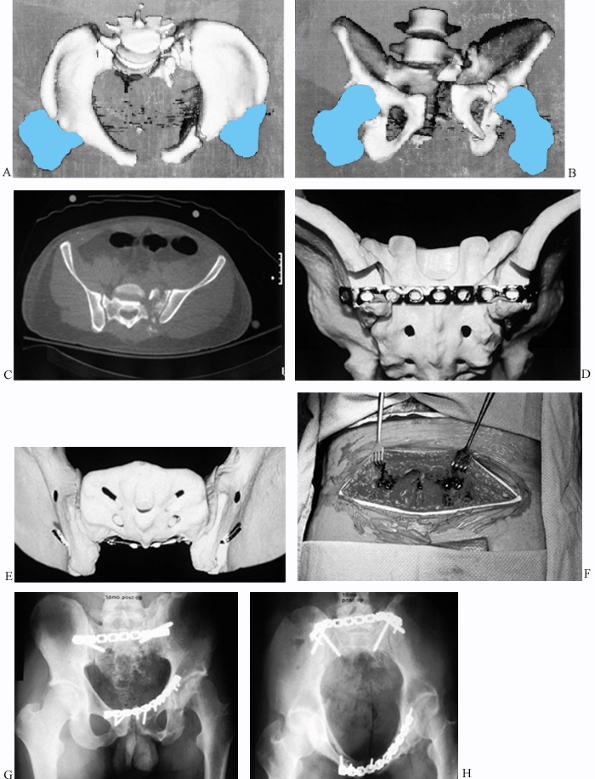 |
|
Figure 17.27.
Transiliac plate fixation of a comminuted vertical sacral fracture and plate fixation of a symphyseal diastasis and left anterior column fracture in a 34-year-old man. A: Preoperative inlet 3D CT. B: Preoperative outlet 3D CT. C: Preoperative CT scan. D: Model of an intraosseous tunnel created for a 4.5 mm reconstruction plate. E: Model of a contoured plate resting in the intraosseous tunnel. F: Intraoperative view of a plate resting beneath the paraspinous muscles. G: Postoperative AP view. H: Postoperative inlet view. |
be applied through the transverse presacral incision or through short
vertical incisions over the lateral aspects of the posterior superior
spines. Restore accurate alignment of the pelvis prior to the insertion
of the sacral rods. Advance the threaded bars through parallel rows of
drill holes that are prepared under image intensification, secure the
nuts to the rods, and compress the fracture. Overreduction is possible
in view of the mechanical advantage of the threaded system.
 |
|
Figure 17.28. Technique for transiliac rod fixation. A: Cutaneous incisions. B: Site for preparation of a drill hole in the posterior superior spine. C: Insertion of a threaded rod. D: Final view.
|
We reserve the extended iliofemoral and triradiate approaches for
complex injuries with associated acetabular fractures. Limited versions
of both exposures have been described that eliminate the need for an
osteotomy of the greater trochanter or a corresponding detachment of
the gluteal insertions (21,41).
This permits visualization from the base of the acetabulum across the
ischial tuberosity to the inferior aspect of the symphysis, which is
particularly useful for a nonunion.
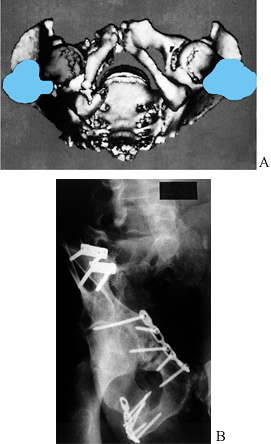 |
|
Figure 17.29. The use of an ischiopubic approach to stabilize the inferior pubic ramus in a 42-year-old man. A: Inferior 3D CT view. B: Postoperative obturator oblique view.
|
the Hoffmann-II system. Whichever system is used, a typical frame uses
5 mm threaded half-pins inserted into the anterior ilium. Simple frames
use one or two, and rarely three, 5 mm half-pins per hemipelvis,
inserted into the anterior iliac crest near the anterior superior iliac
spine (ASIS) or the anterior inferior iliac spine (AIIS) (42,52).
Where the frame is intended for temporary stabilization for a few days,
or when the injury pattern is rotationally unstable but vertically
stable, one pin per hemipelvis suffices. When the pelvis is unstable,
insert two pins per hemipelvis, spacing the pins widely to achieve
optimal stability. For a markedly obese patient with an unstable
pelvis, three pins per hemipelvis may be necessary. We prefer to place
the pins into the gluteal tubercle, ASIS, and AIIS.
-
Prior to the procedure, determine the
required number and length of pins. Organize the appropriate drill
sleeves, drill bits, pins, and frame constituents. -
Position the patient supine on a radiolucent table.
-
Unless speed is important, take AP,
inlet, and outlet views with the image intensifier to assess the
deformity and determine the strategy for reduction. If there is
superior migration of the hemipelvis, apply longitudinal skeletal
traction to restore leg lengths. Correct a diastasis of the symphysis
with a rotational deformity after the half-pins have been inserted,
using the pins as reduction tools. -
If reduction of the pelvis is not
necessary, place the pins through small stab wounds directly over the
entry point. If reduction is required, particularly of external
rotation, make bilateral oblique incisions in the skin where the
reduced pelvis will eventually lie. The incisions must not parallel the
iliac crest but be in line with the direction of pin movement. For
example, with 5 cm of pubic diastasis, place each incision 2–2.5 cm
medial to the crest.
the pelvis is only minimally displaced and where intraoperative image
intensification is available.
-
Identify the pin entrance sites on the superior aspect of the iliac crest at the gluteal tubercle and at the ASIS or AIIS.
-
Make a stab incision over the site and
cut down to the top of the iliac crest. At the AIIS, the ilium is
deeper than at the ASIS. Spread the tissues with a small periosteal
elevator. -
Slide a 2.0 mm Kirschner (K-) wire
subperiosteally down the inner table of the pelvis parallel to the axis
of the pin to be inserted to act as a guide pin. Some surgeons slide a
second K-wire down the outer table as well to help define the position
of the ilium. Use the C-arm for this and verify that the wires are
placed appropriately. -
Drill a 4.0 mm hole at the entry site for
the pins, through the cortex but not into the cancellous bone between
the tables. A short entry hole minimizes the risk of accidental
penetration of the cortex. -
Under fluoroscopic control, insert two
Schanz-type long threaded half pins into each ilium so that they
advance between the two cortices of the bone. Angulate the pins so that
they are divergent from each other in the AP plane. The deeper the
pins, the better the fixation, but avoid penetration of the opposite
cortex or the hip joint. Leave enough pin protruding to connect the
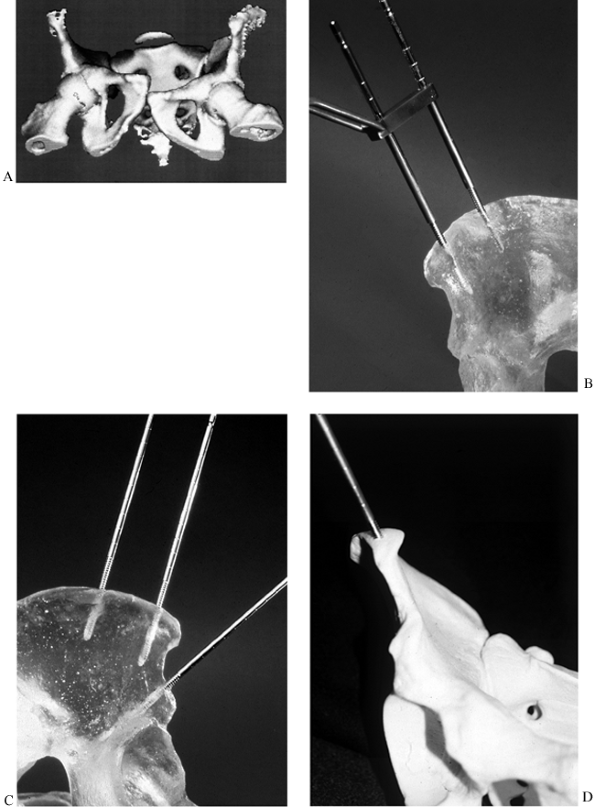
frame at an appropriate height (Fig. 17.30A, Fig. 17.30C).![]() Figure 17.30. Insertion of half pins for external pelvic fixation. A:
Figure 17.30. Insertion of half pins for external pelvic fixation. A:
Anterior outlet view to highlight appropriate target zone for pins
between the iliac tables. The view also displays errant pin tracks from
a closed insertion technique without image guidance. B: Optimal pin sites on the anterior iliac crest. C: Ancillary pin site on the anterior inferior spine. D: Outlet view with optimal alignment for a percutaneously inserted pin. -
Attach an 8 mm carbon fiber or other suitable bar of appropriate length to each pin with a pin-bar clamp.
P.566
Attach the two sets of bars to each other with a bar-to-bar clamp in
the midline. Be certain that the frame will not encroach on the abdomen
when the patient sits up. Adding cross bars between each set of bars
increases stability. -
Reduce the pelvis under fluoroscopic visualization.
-
Prior to the final assembly and
tightening of the frame, turn the patient into a lateral position and
manipulate the hemipelvis until the deformity is corrected. -
When the final position is secured, check
the skin about the pins and extend the wounds to relieve any tension
where skin has gathered on the pelvis. -
Then close the subcutaneous fat and skin snugly about the pins and dress the wounds.
-
Take a full series of plain radiographs before leaving the operating room to check the reduction and pin position.
-
Two different skin incisions can be made.
An incision parallel to the iliac crest is simplest, but skin tension
frequently requires that the incision be “T” ed. Separate 2.5-cm-long
incisions can be made for each pin. Place these in line with the skin
tension on the pins, roughly at right angles to the iliac crest. -
Expose the top of the crest and inner
table by subperiosteal dissection so that the pins can be placed under
direct vision. Some surgeons expose the outer table as well, but
usually this is unnecessary. -
Proceed as previously described for the remainder of the procedure.
invasive temporary pelvic stabilization. Currently, two designs of
pelvic clamp are available. The original Ganz design (16,17) (Fig. 17.31)
immobilizes the region of the SI joints for optimal mechanical
fixation. The liability of the design is the proximity of the greater
sciatic notches, with the
potential
for a significant neurovascular injury. The Browner design utilizes
anterior iliac pins that afford considerably greater safety, but the
frame provides less stability. Whichever method is used, the surgical
team needs to be familiar with the techniques for insertion of the pins
and erection of the frame. These clamps should be used only by surgeons
who have had experience with them, as routine frames are safer. While a
clamp can be applied in an emergency room, sterile technique must be
maintained.
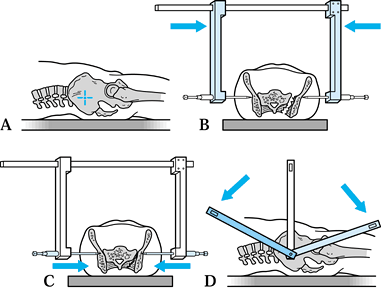 |
|
Figure 17.31. Schematic views of the Ganz type of external pelvic clamp (17). A: Site for pin insertion. B: Maneuver for pelvic reduction. C: Postreduction with tightening of the locking nuts. D: Potential displacement of the frame.
|
write out a surgical strategy that defines the injuries, positioning,
and intraoperative tactics and alternatives (Table 17.33).
Where sequential reductions are planned through separate surgical
incisions, as a rule the first one is the most accurate. If the first
one is imperfect, the magnitude of the error is progressively magnified
from the second to the third or fourth stage. Once a first stage is
completed and the patient is repositioned for the second one, any error
in the first stage cannot be eliminated without reopening the first
wound.
 |
|
Table 17.33. Preoperative Planning for an Internal Fixation
|
for one of two tactics. In the first strategy, the priorities for
accuracy of reduction are (a) acetabulum, (b) sacrum and SI joints, and
(c) rami and symphysis. Certain associated injuries, however, are
linked. For example, about 20% of transverse acetabular fractures are
associated with an ipsilateral SI subluxation as an external rotation
deformity. Unless the SI joint is reduced initially, the acetabulum
cannot be accurately realigned. In this instance, start the reduction
with the SI joint and follow with the acetabulum. The second strategy
is simultaneous reductions of multiple displaced injuries using more
extensile surgical approaches (24).
 |
|
Table 17.34. Alternative Approaches for Pelvic and Femoral Fractures
|
malrotation or 2 cm of linear displacement in any plane merits
consideration for open reduction and internal fixation. For example, an
external rotation injury can be characterized by the amount of widening
of the symphysis or the degree of malrotation of the hemipelvis. If a
symphysis diastasis is less than 2.5 cm, nonoperative treatment is
usually possible. Wider diastasis usually has sagittal malrotation,
reflected in the AP radiograph by the involved ramus being higher than
the uninvolved side. This indicates that the ipsilateral sacrospinous
and sacrotuberous ligaments are compromised. If the diastasis is wider
then 5 cm, the posterior injury is bilateral, or it is a highly
unstable unilateral disruption in the form of SI dislocation or a
sacral fracture. If deformity is evident in the sagittal or coronal
planes, the injury pattern is even less stable than if the deformity is
only in the transverse plane. Both of the latter usually require
internal fixation to prevent nonunion and malunion of the pelvic ring.
In type C injuries, the hemipelvis is usually posteriorly displaced,
whereas true vertical displacement is uncommon. Vertical displacement
is easily confused with sagittal malrotation of the hemipelvis. On the
AP radiograph, vertical displacement is consistent with a hemipelvis
that is high riding but otherwise a replica of the contralateral
hemipelvis. In sagittal deformity, the ischial tuberosities are at
different levels, although the tops of the iliac crests are symmetrical
and the involved hemipelvis shows a change in the shape of the pelvic
brim.
Specialized plates are available, but they have not been proven to be
superior despite increased costs. Both 3.5 mm and 4.5 mm reconstruction
plates are commonly used. We prefer the 3.5 mm plates with longer
screws. In our experience, two screws per ramus (Fig. 17.22)
is adequate. Two plates mounted at right angles to one another provide
the strongest construct, but one plate is sufficient and decreases the
soft-tissue stripping.
-
Expose the pubic symphysis and adjacent pubic rami through a Pfannenstiel or lower midline abdominal incision (Chapter 2).
The distal portions of the rectus abdominus muscle insertions usually
can be left intact, while the more proximal portions of the muscle are
reflected in a lateral direction from the anterior and superior
portions of the superior rami. -
Place the tines of a tenaculum forceps into the obturator foramen and reduce the symphysis
-
Contour a four- to five-hole 3.5 mm or 4.5 mm reconstruction plate to fit on the superior surface of the rami and symphysis.
-
Palpate or visualize the obturator foramen as a guide to the acetabulum.
-
Do not insert a screw that is directed toward the center of the acetabulum.
-
If a screw in the area of the acetabulum is essential, insert the screw more posteriorly and along the pelvic brim.
-
If a retrograde pubic screw is used, for image intensification use inlet, outlet, and iliac and obturator oblique views.
-
Insert the first two screws adjacent to
the symphysis, parallel to the joint and the posterior surface of the
rami. Protect the bladder and determine screw direction by palpating or
visualizing the posterior aspect of the symphysis. Be certain that the
screws are bicortical. Insert the other two screws. -
Close the wound in a layered fashion over a suction drain.
-
Use a vertical midline abdominal incision.
-
Look carefully for a corona mortis and ligate it.
-
Contour four- or five-hole 3.5 mm plates to fit along the superior posterior aspects of the pelvic brim.
-
Insert screws using radiographic control to ensure that the screws miss the hip joint.
-
Alternatively, use the retrograde medullary superior ramis screw fixation technique, as described previously.
-
Close in layers over a suction drain.
common: In most cases, it is done for a late painful nonunion.
Stabilization may require a bone graft for a defect (Fig. 17.29).
-
Position the patient in the lateral
decubitus position on a radiolucent table with maximum flexion of the
ipsilateral hip and knee, which are uppermost. -
Prep and drape carefully to exclude the rectum and genitalia.
-
Make an ischiopubic exposure (Chapter 2).
-
Reduce the fracture with a small tenaculum or lion-jaw forceps.
-
Contour and apply a 3.5 mm reconstruction plate.
-
Use the obturator oblique view to ensure that screws do not penetrate the acetabulum and hip joint.
-
Close in layers over a suction drain.
-
For a late presentation with a fixed
deformity, a structural bone graft may be needed to reestablish the
continuity of the ischium. -
Where the fracture is higher in the
ischium and transverse, use lag screws inserted directly in the
prominent portion of the ischial tuberosity. (See Fig. 17.39 on p. 578.)
-
Position the patient supine on a radiolucent table.
-
Expose the inner ilium, anterior SI joint, and lateral 2.5 cm of sacrum through an ilioinguinal approach (Chapter 2).
-
If fixation of the symphysis is required, expose it simultaneously.
-
Clear the SI joint of debris and soft tissues.
-
Attach a three-hole 3.5 mm plate to the
sacral ala in a convenient location, leaving room for a second plate.
Use a 4.5 mm cortical screw. -
Place a large tenaculum forceps with one
point in the lateralmost hole of the plate and the other on the iliac
crest, and reduce the SI joint. -
Contour a second, similar plate and fix it to the sacrum with one screw and to the ilium with two screws.
-
Remove the forceps and insert the remaining screws in the first plate.
-
Verify the reduction and screw placement with the C-arm using inlet and outlet views.
for screw insertion is used, the irregularity of the posterior margins
of the SI joint impedes assessment of the reduction. The undersurface
or superior surface of the SI joint can be palpated, but intraoperative
imaging is essential. Accurate reduction is necessary to provide a safe
and predictable site for the fixation screws. For assessment of the
target zone in the sacrum, the direct lateral image is the optimal
view. When the AP, inlet, and outlet views are used without a lateral
image, the most common error is a screw that appears to be
satisfactorily positioned but that, on the lateral view, is in fact
superior to the S-1 body.
-
During the exposure, use bone wax on the intraosseous veins at the base of the internal iliac fossa to control bleeding.
-
If the articular cartilage of the SI joint is damaged, thoroughly remove it to achieve a fusion.
-
Direct the screws parallel to the SI joint.
-
Diverge the plate position to increase stability.
-
Tightening of the first iliac screw should reduce the posterior displacement of the ilium.
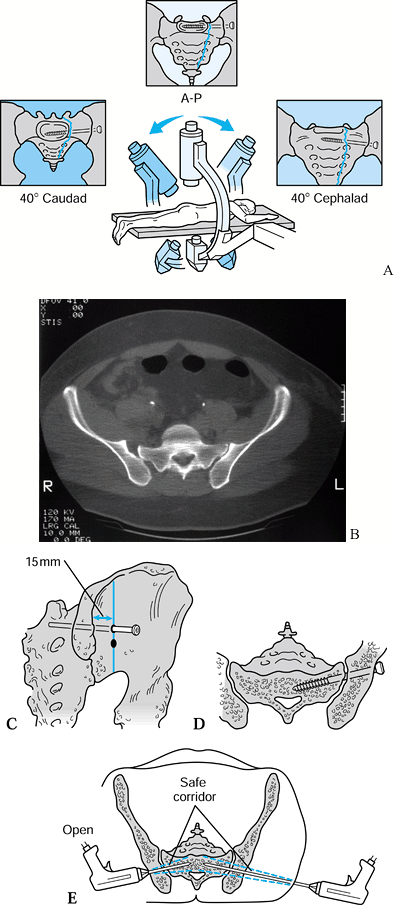 |
|
Figure 17.32. The use of intraoperative image intensification for iliosacral screw insertion. A: General method for the prone patient. B: CT of SI malreduction where iliosacral screw insertion is not technically feasible. C: Landmarks for screw insertion. D: SI cross section with ideal screw placement. E: Target zones for iliosacral screws with an open or closed technique.
|
-
Position the patient halfway between prone and lateral so that the involved ilium is parallel to the table.
-
Make a vertical incision at the posterior superior spine.
-
Palpate the inferior margin of the SI joint through the greater sciatic notch.
-
Reduce the SI joint with a tenaculum
forceps and check the reduction radiographically (AP, inlet, outlet,
and obturator oblique views). -
Identify the insertion site for lag
screws on the lateral ilium, halfway between the posterior superior
spine and the anterior aspect of the roof of the greater sciatic notch. -
Insert two 6.5-mm-long threaded cancellous screws or corresponding 7.3 mm cannulated screws.
-
Assess final imaging using AP, direct lateral, inlet, and outlet views.
perhaps the least complex technically, with a relatively low risk for
injury to neurovascular bundles or viscera, apart from the superior
gluteal vessels. The fixation is outstanding, so an accompanying pubic
symphysis diastasis or ramus fracture does not have to be immobilized.
Since the posterior SI ligaments are intact, the SI joint is stabilized
upon fixation of the fracture.
-
With the posterolateral position, gravity facilitates the reduction.
-
Palpation over the superior aspect of the SI joint of the alar region is helpful in the reduction.
-
While drilling, feel the drill bit
advance across three cortical layers: the superficial iliac table and
the iliac and sacral subchondral bone at the SI joint. Do not penetrate
a fourth dense layer of bone, which represents the S-1 foramen or the
anterior aspect of the sacrum. -
Measure 50–60 mm of penetration of the
drill and select a corresponding screw. Usually, the S-1 foramen is 55
to 65 mm deep from the lateral ilium and thereby is avoided. -
Use a direct lateral sacral view to ensure that the screws are anchored in the S-1 body.
-
Carefully protect the superior gluteal vessels.
-
Direct the screws through the iliac crest between the tables.
-
Insert the inferior screws into the posterior inferior spine.
-
Use the iliac and obturator oblique views to guide the fixation.
-
Limit the use of SI screws to comminuted fractures or for the rare occasion when the posterior SI ligaments are completely torn.
-
Position the patient halfway between prone and lateral.
-
Drape the ipsilateral lower extremity free.
-
Make a limited iliofemoral approach and reflect the hip abductors to expose the fracture.
-
Reduce the fracture with tenaculum forceps and positioning of the leg.
-
Apply a 3.5 mm reconstruction plate along the iliac crest.
-
Where the inferior half of the fracture
is a simple configuration, insert two 4.5 mm cortical or 6.5 mm
cancellous screws using lag technique. -
If the inferior half of the fracture is comminuted, apply a 3.5 mm plate on the lateral ilium.
relatively low morbidity and the stability is outstanding. We usually
insert supplementary iliosacral screws for two-column pelvic fixation.
An alternative is to fix the anterior column where iliosacral screws
are not appropriate. If vertical displacement of a sacral fracture is
present, use preoperative skeletal traction to reduce it. Maintain
traction during the procedure.
-
With the patient prone, make a transverse presacral exposure at the level of S-1.
-
Expose the fracture and remove any displaced foraminal fragments.
-
Reduce the fracture with a large tenaculum and King Tong forceps.
-
Make notches in the posterior superior spines.
-
Contour and apply a 4.5 mm reconstruction plate with seven to nine holes.
-
If a large anterior sacral alar fragment
is present, reduce it by inserting a tenaculum forceps superior to the
SI joint, under image control. -
As an alternative, the transverse plate can be applied superficial to the S-2 segment.
-
Direct the sacral alar screws parallel to
the SI joint on divergent pathways. Do not fill the central presacral
screw holes that are over the spinal canal and foramina.
-
Verify position with AP, inlet, and outlet images.
-
If a supplementary iliosacral screw is used, insert it before applying the plate to optimize imaging.
-
If a supplementary transverse fracture line is present, apply an additional hook plate near the lateral margin of the bone.
-
As an alternative to the lengthy
transverse plate, short plates that solely bridge the fracture can be
used. The exceedingly limited safe target zones for the screws need to
be clearly understood (52).
While the method is technically elegant, the sites for safe and
effective screw placement are small and anatomically variable and
therefore hazardous to the sacral nerves. Prior to performing this
procedure, observe the technique in the hands of an experienced surgeon.
utilizes two rods that span from one posterior superior iliac spine to
the other. It is suitable for a single site of posterior injury, such
as a vertical sacral fracture. For a bilateral SI dislocation, it
affords no intrinsic pelvic stability unless supplementary iliosacral
screws are used. Perhaps the most frequent shortcoming of the method is
failure to obtain an accurate or acceptable reduction, particularly
when using minimally invasive techniques, where malunion can occur. The
method also depends on the presence of good quality bone. With
osteopenia, premature loss of fixation is likely.
-
Use the same approach as described for
the insertion of posterior lag-screw fixation of the SI joint, with the
addition of a smaller exposure on the contralateral side to expose the
lateral aspect of the iliac wing anterior to the posterior spine. -
Reduce the fracture.
-
Place drill holes through the posterior tubercles of the ilium just dorsal to the sacral lamina.
-
Place two rods posterior to the sacrum and 2–4 cm apart, between the two intact posterior tubercles of the iliac wings.
-
Place the first rod just proximal to the S-1 foramen and the second rod just distal to the S-2 foramen.
-
Insert the rods proximally to avoid entering the sacrum and endangering the nerve roots within the sacral canal.
-
Alternatively, use cannulated rods and imaging to help guide the rod placement.
-
Insert nuts and washers to compress the system.
-
Based on its intrinsic biomechanical
limitations, limit the use of this technique to the fixation of a
unilateral unstable sacral fracture, or use it in combination with
posterior screw fixation to stabilize an SI joint dislocation or a
bilateral sacral disruption. -
Avoid overzealous compression of the
fracture site, particularly in the presence of foraminal fractures, to
minimize the risk of neurologic compromise.
The anterior displacement of the sacrum is aggravated by prone
positioning of the patient, so the reduction is challenging and
potentially hazardous. A pointed reduction forceps or hook must be
cautiously inserted around the lateral margins of the sacrum to the
edges of the anterior surface to achieve the reduction. With the
proximity of the rectum, the maneuver must be done with care. A variety
of plates, lag screws, and cables have been used to anchor the sacrum.
Supplementary compression rods with pedicular screws may be needed to
immobilize the lower lumbar spine to the sacrum (16).
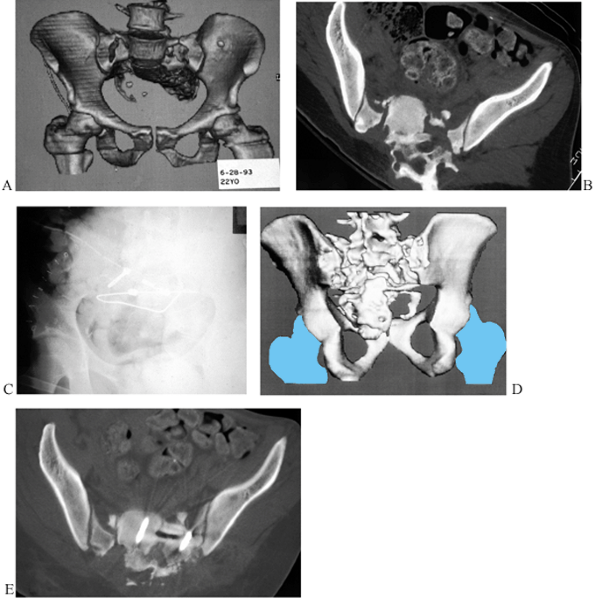 |
|
Figure 17.33. A lumbosacral dissociation in a man of 29 years, in whom a combination of cable fixation and lag screws was used. A: Preoperative anterior 3D CT view. B: CT scan. C: Postoperative AP view. D: Postoperative 3D CT posterior view. E: Postoperative CT scan.
|
-
Make a suitable midline longitudinal exposure of the sacrum and relevant portions of the lumbar spine.
-
Apply a transverse plate across the S-1 and S-2 segments, possibly with associated hook plates.
-
Immobilize the sacrum to the lumbar
spine. Distraction or compression rods can be used to anchor the
relevant lumbar spinal segment to the two transverse plates by the use
of hooks placed over the superior or inferior margins of the transverse
sacral plates. Alternatively, pedicular screws can be employed (16). -
Three major technical problems must be
overcome: sacral realignment, and fixation and immobilization of the
sacrum to the lumbar spine.
principal structural portions of the pelvic ring, notably for
iliosacral fixation, and 4.5 mm screws for periacetabular fixation. We
use drill sleeves to protect the adjacent soft tissues.
table with a wholly radiolucent draping technique that eliminates towel
clips. A modern image intensifier with a large field size of 9–12
inches is strongly recommended. The appropriate radiographic views for
effective guidance of the relevant screws need to be clearly understood
and technically feasible. Prior to draping, rehearse the views to
ensure their feasibility (Fig. 17.32). Table 17.35 lists the indications and contraindications of the procedure.
 |
|
Table 17.35. Indications and Contraindications for Percutaneous Internal Pelvic Fixation
|
In more complex disruptions, open reduction and fixation of an anterior
ring disruption such as a diastasis of the symphysis may realign the
posterior ring so that percutaneous stabilization is feasible.
supine, prone, or lateral position, although imaging is slightly more
difficult in the lateral position (33,52,58). When open anterior ring fixation immediately precedes the iliosacral fixation, we prefer the supine position (Fig. 17.34).
When the patient is positioned prone for an accompanying procedure,
maintain that position. In the presence of external fixation, the
lateral position greatly facilitates reduction of an SI dislocation.
Irrespective of the position, the method for screw insertion is similar.
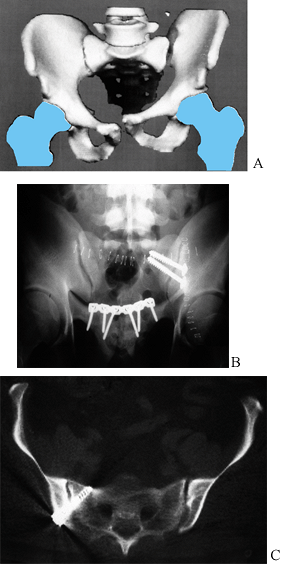 |
|
Figure 17.34. An example of iliosacral screw fixation in a man of 33 years. A: Anterior 3D CT scan. B: Postoperative outlet view. C: Postoperative CT scan.
|
-
Make a 2 cm transverse incision on the
lateral flank, along the line that is halfway between the greater
trochanter and the posterior surface of the back. -
Spread the underlying soft tissues to bone.
-
Insert the guide wire and drill sleeve to the bone and confirm the position in the AP view.
-
Using power, advance the guide wire just across the SI joint (use AP, inlet, outlet, and direct lateral views).
-
If the first wire is not where you want
it, leave it in place and insert a second wire, using the first as a
supplementary guide. -
Advance the guide wire to the level of the S-1 foramen.
-
Insert a second guide wire into the S-1 body.
-
Measure for screw length with a depth gauge and insert the appropriate long-threaded screws of 45–60 mm in length.
-
For the ideal inlet view, adjust the gantry until the anterior margins of S-1 and S-2 coincide.
-
Use the AP view for transverse
orientation, the inlet for AP positioning, the outlet for the S-1
foramen and superior S-1 endplate, and the direct lateral of S-1 for
location of the screw tips. -
If the screw is 45 mm, use the obturator
oblique view to confirm that the screw threads are not across the joint
so it can be lagged together. -
Do not use screws longer than 60 mm.
-
For bilateral screw insertion, insert all of the guide wires before any screws to optimize imaging.
iliosacral screws is similar to that described for an SI joint, there
are some additional considerations (59). Once
the screw passes beyond the S-1 foramen, the potential for neurologic
injury increases. The target zone for the iliosacral screw markedly
diminishes, while the corresponding demands imposed on the
intraoperative imaging increase. Anatomic variations of the L5–S1
articulation can further compromise the target zone available in the
sacrum (Fig. 17.35).
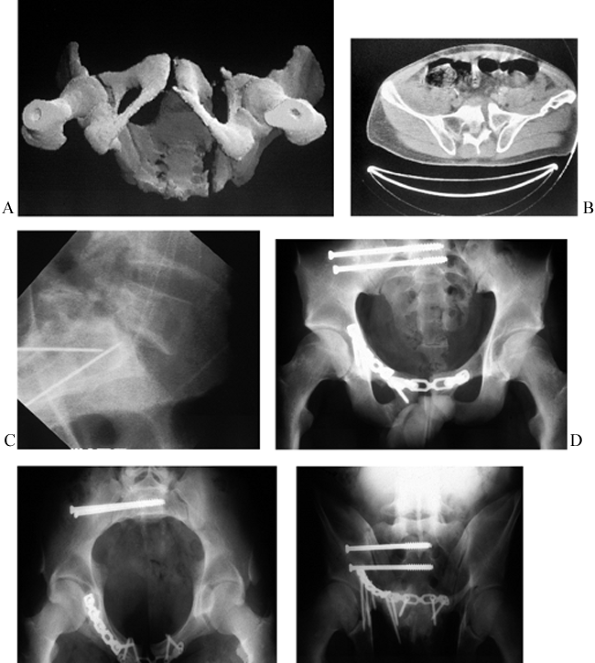 |
|
Figure 17.35.
Percutaneous iliosacral screw fixation of a transforaminal sacral fracture with an initial open reduction of the right superior pubic ramus in a 31-year-old man. A: Inferior 3D CT scan. B: CT scan. C: Intraoperative direct lateral sacral view. D: Postoperative AP view. E: Postoperative inlet view. F: Postoperative outlet view. |
-
Using the supine or prone position, draw a reference line as described previously.
-
As described under iliosacral screw
insertion for the SI joint, make a lateral incision and insert a guide
wire into the S-1 body. Place it mid axially from anterior to
posterior, and centralized between the superior endplate and the S-1
foramen (use AP, lateral, inlet, and outlet views). -
Advance the guide wire to the level of the contralateral S-1 foramen.
-
For an unstable fracture, insert a second
guide wire for another iliosacral screw that is anterior and inferior
to the first but still in the S-1 body. -
Measure for depth and insert two long threaded screws of appropriate length.
-
Obtain final imaging.
although the target zone is much smaller than S-1. To account for the
variable degree of lumbar lordosis, select the inlet view where the
anterior margins of S-1 and S-2 are superimposed. The direct lateral
view is crucial to determine appropriate insertion of the guide wire.
comminuted and highly unstable. If unstable, plate fixation is
recommended to augment iliosacral screws. For bilateral transforaminal
sacral fractures without additional comminution, either of two
strategies for iliosacral screw fixation can be used. Insert either
ipsilateral screws for each injury site (Fig. 17.36)
or insert long screws that transfix both sides of the sacrum. The
latter method necessitates an anatomic reduction, the absence of
congenital sacral anomalies, and highly accurate placement of the guide
wires. Where bilateral transfixing screws are inserted, advance the
guide wires with a perfectly transverse orientation and mid axial from
anterior to posterior in S-1 (Fig. 17.24). The screws are usually 120–150 mm long.
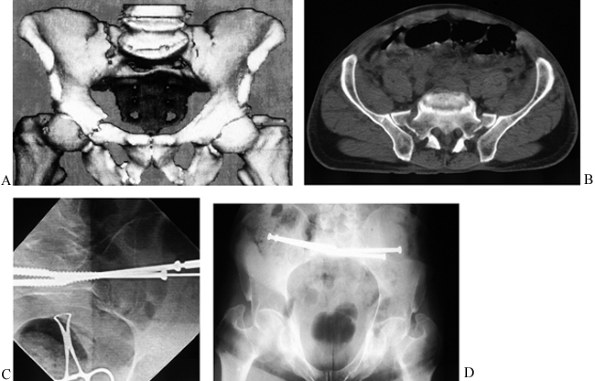 |
|
Figure 17.36. Bilateral sacral alar fixation in a man of 41 years. A: Preoperative anterior 3D CT view. B: CT scan. C: Intraoperative AP view. D: Postoperative inlet view.
|
displacement and extending from the iliac crest to the greater sciatic
notch are appropriate for percutaneous screw fixation (52).
For the more posterior injuries that are at or posterior to the
anterior border of the SI joint, use the prone position. If the iliac
fracture is further anterior, either a prone or a supine position is
feasible. In the prone position,
the insertional site is the posterior superior spine (Fig. 17.37).
In the supine position, use the anterior inferior spine. Insert two
parallel screws into either site with a spacing of about 15 mm. Keep in
mind the divergent pathway of the ilia, from posterior to anterior
throughout the target zone (52).
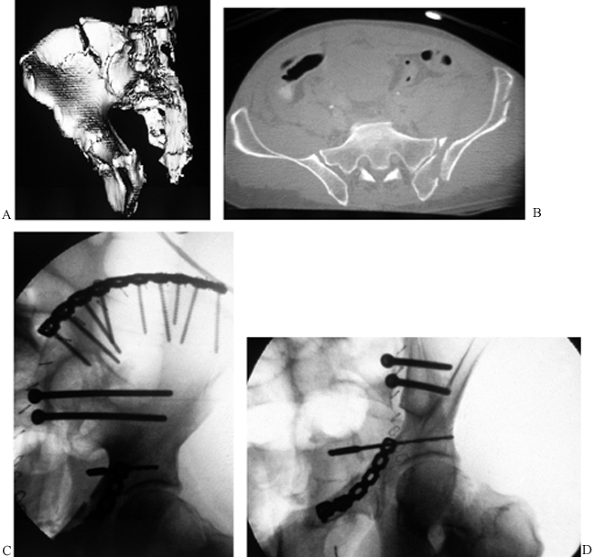 |
|
Figure 17.37. The method for percutaneous insertion of a transiliac screw to immobilize the posterior ilium. A: Iliac oblique 3D CT view. B: CT view. C: Intraoperative iliac oblique view. D: Intraoperative obturator oblique view.
|
-
Place the patient prone.
-
Make a 10 mm stab wound that is about 20 mm superior to the posterior superior spine.
with the anterior inferior spine as the initial target for the guide
wire. Direct the guide wire toward the posterior superior spine.
-
Insert a guide wire into the wound and
down to bone. Direct the guide wire on the iliac oblique view toward
the anterior inferior spine and on the obturator oblique view between
the inner and the outer iliac tables. -
Advance the guide wire until at least 35 mm is into bone beyond the fracture.
-
Insert a second parallel guide wire.
-
Measure for and drive the two screws.
screw inserted antegrade from laterally, or alternatively retrograde
from medially. The former is difficult and hazardous. The antegrade
screw is not recommended for routine use, as the obliquity of the
screw, the limited target zone of bone, and the juxtaposition of the
external iliac vessels makes the technique hazardous. When inserted
retrograde, the shorter length of the screw limits the technical
complexities, although a virtually anatomically reduced fracture is necessary (52,61) (Fig. 17.38).
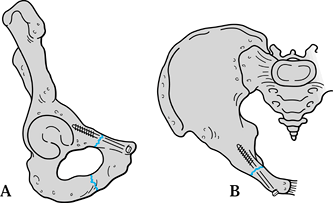 |
|
Figure 17.38. Schematic views of the insertion of a percutaneous retrograde ramus screw. A: Obturator oblique view. B: Iliac oblique view.
|
-
With the patient in the supine position, make a 10 mm incision anterior, medial, and slightly inferior to the pubic tubercle.
-
Perform a closed reduction of the injured hemipelvis.
-
Advance a guide wire to bone. On an
oblique outlet fluoroscope view, direct the guide wire into and up the
superior pubic ramus. On the inlet view, direct the guide wire parallel
to the inner border of the true pelvic brim. -
Advance the guide wire beyond the fracture.
-
Measure for screw length and insert an appropriate 3.5 mm or 4.5 mm cortical screw and tighten.
posterior column of the acetabulum can be fixed using percutaneous
screws. The entry site for the screw is the ischial tuberosity with
vertical orientation of the screw along the posterior column (Fig. 17.39).
 |
|
Figure 17.39. Schematic views of the insertion of a percutaneous posterior column screw via the ischial tuberosity. A: Obturator oblique view. B: Iliac oblique view.
|
-
Place the patient supine on a radiolucent
table with the hip fully flexed and the knee at 45° to 80°. Abduct the
hip and place the leg in stirrups, or on a Mayo stand. -
After suitable draping, palpate the ischial tuberosity and make a 10 mm incision.
-
Insert the guide wire to bone and image (AP, internal oblique, and outlet views).
-
Direct the guide wire vertically toward
the gluteal tubercle. While imaging on the internal oblique view,
direct the guide wire parallel to the posterior border of the greater
sciatic notch, halfway between the notch and the posterior acetabular
rim. On the oblique outlet view, direct the guide wire so that it
passes halfway between the posterior acetabular rim and the most
posterior margin of the obturator foramen. At a more superior level,
the guide wire is halfway between the posterior acetabular rim and the
center of the anterior inferior spine. -
Insert a 4.5 mm screw of appropriate length.
-
Prior to skin closure, move the hip to be certain that the screw is not in the joint.
be used for supplementary reduction and fixation. The ringlike
configuration of the pelvis makes cables and wires particularly useful (43).
the pelvis as the principal site for intended use. When a cable is
tightened, it shortens its pathway. To control the pathway and the
vector of displacement of the bone, we usually pass the cable through a
small drill hole in the pelvis. In this way, the displacement of the
tightening cable and its effect on bony alignment can be controlled.
Many cable systems possess a tightening mechanism at one end of the
cable that blocks its insertion into a drill hole. In some
applications, both ends of the cable have to be passed through drill
holes. In this situation, the use of a “double-ended” cable or wire is
essential.
semicircle designed for the femoral shaft. Such a design does not work
well on the pelvis. For example, at the greater sciatic notch, the
semicircular design can injure the sciatic nerve. As an alternative,
use a Statinski vascular clamp, which has angled jaws and a blunt tip
and is available in various sizes that work well on the pelvis.
many sizes. They vary from excessively bulky to low-profile
configurations. Some tighteners can be removed while a cable is
provisionally held tight. We prefer to use double-ended cables with a
low-profile tightener. Cable fixation is useful for a wide variety of
unusual problems, such as sacral dissociation (Fig. 17.33).
-
Whether a medial or lateral exposure is used, pass the cable through the greater sciatic notch.
-
Anteriorly pass it around the base of or
through a drill hole in the anterior inferior spine. Align the lateral
ilium and tighten and secure the cable (Fig. 17.23).
-
At the base of the posterior column, pass
the cable through the lesser sciatic notch. Then pass it through a
drill hole at the base of the anterior inferior spine. -
After reduction of the fracture, tighten and secure the cable.
quadrilateral surface fragment that is difficult to fix. The opposing
articular surface of the hip joint eliminates most of the bone from
consideration for screw fixation. Buttress plates are highly vulnerable
to late displacement. A cable or heavy wire can be used to supplement a plate on the pelvic brim (Fig. 17.23).
-
Reduce the iliac and superior pubic ramus portions of the fracture and apply a plate along the pelvic brim.
-
Drill 2.5 mm holes through two the central holes in the plate that exit on the quadrilateral surface.
-
Prepare a 4×5 cm structural bone graft from the inner iliac table and make two corresponding drill holes in it.
-
Pass the cable or wire down through the
pelvic brim, out through the quadrilateral table, and through the holes
in the structural graft. Then retrograde the cable through the
quadrilateral surface and pelvic brim. -
Tighten and secure the cable.
-
Under image intensification, inset two
iliosacral screws that just traverse the entire posterior width of the
pelvis (120–150 mm length). -
Through a transverse posterior exposure, identify the sites of the screw heads and screw tips.
-
Pass a cable through the cannula of one screw.
-
Apply a two-hole 3.5 mm plate on either
end of the cable and feed each end of the cable through the second hole
in the appropriate plate. -
Retrograde one end of the wire through the second cannulated screw.
-
Tighten and secure the cable.
patient varies considerably, depending on the specific nature of the
injury and the presence of a wide variety of associated injuries to the
appendicular skeleton or spine. Nevertheless, certain general
principles provide helpful guidelines. The fundamental goal is to
mobilize the patient as rapidly as possible without an excessive
likelihood for a loss of the pelvic reduction.
The morale of patients with these severe injuries is low because of the
physical impact of the injury, the vocational and financial
implications, and sometimes the injuries to other family members or
friends in the same accident. Rapid mobilization is a great boost to
morale. Outpatient and home-based rehabilitation is preferred to
institutionalization. If inpatient therapy is needed, arrange for it to
be near the patient’s home so that friends and family members can visit
easily.
whenever the general condition of the patient permits, begin
bed-to-chair transfers, possibly with a sliding board the day of
surgery. This facilitates pulmonary toilet and heightens morale.
Encourage the patient, as soon as practical, to begin touchdown
walking. About 6 weeks later, advance the patient to a partial
weight-bearing gait, and progress to full weight bearing over the next
6–8 weeks. Prescribe progressive resistance exercises to the muscle
groups around the pelvis and in lower extremities, with emphasis on the
hip abductors and the quadriceps muscles.
internal fixation is used, the activity level depends on the degree of
pelvic instability. For a vertically stable injury, early touchdown
gait training may be feasible. For an equivocally stable situation,
encourage bed-to-chair transfers. Defer gait training for 6 weeks until
the external frame is removed. At home, use a wheelchair and a bedside
commode.
specialized needs for persistent physical disability, and vocational
planning. A generalized weakened state follows most pelvic fractures,
especially in multiple trauma, because of malnutrition and prolonged
disuse. Nutritional advice is needed after discharge.
pelvic muscles, followed in a month with a generalized strengthening
program. Include water therapy with a pool, ambulation, and swimming.
At 6 weeks after the injury, initiate an aerobic exercise program with,
for example, a stationary bicycle or a rowing machine. Full
rehabilitation often requires 1–2 years of enthusiastic participation
in a program. Return to a laboring job is likely to require at least
6–12 months.
resting ankle–foot orthosis along with a custom-molded shoe insert.
Initiate passive stretching exercises of the heel cord, and muscle
stimulation. As recovery progresses, institute active and active
assistive exercises.
patient-controlled anesthesia (PCA) with narcotics is effective in
alert patients (15). While patient satisfaction
is usually good, nausea, urinary retention, drowsiness, confusion, and
overdosage can occur. Within a day or so, oral narcotics
usually replace the PCA. A muscle relaxant may be necessary to manage muscle cramps.
effective pain control, but an indwelling Foley catheter is usually
required. Resultant motor weakness may interfere with mobilization.
look for persistent pelvic instability; wound hematoma; infection;
dysesthetic pain from a nerve root, plexus, or a peripheral nerve
injury; and other causes.
from lumbosacral plexus injury, scarring, or reflex sympathetic
dystrophy. Referral to a pain management specialist clinic may be
necessary to identify the cause. Reassure the patient that substantial
or complete recovery is likely over a period of many months (42,51,52,70).
(DVT) and pulmonary embolus (PE) in pelvic trauma and reconstruction,
routine prophylaxis is indicated (47,48 and 49).
The role for differing regimens is controversial. We use early
mobilization from bed, as well as either low-molecular-weight heparin
or coumadin for a postoperative period of 2–3 weeks. When long-term
prophylaxis is indicated we use coumadin.
In patients transferred late to us, and in other high-risk patients, we
do a duplex ultrasound evaluation. If clots are found in the thigh
veins, we insert an IVC filter prior to surgery. Another option is
transient heparinization, with a temporary reversal for the pelvic
surgery followed by reanticoagulation with heparin, coumadin, or
low-molecular-weight heparin (see Chapter 5 for more details).
insertion. Examine the patient and obtain radiographs at 6 and 12 weeks
after surgical management or acute trauma. Most pelvic injuries heal by
12 weeks.
to work, at least in some capacity, a few salient observations may be
helpful. For sedentary workers with a stable pelvis, part-time return
to work may be feasible within 2–3 weeks after injury. Typically, a
patient can tolerate 2–4 hours per day provided that suitable
transportation is feasible. Within 12 weeks, the sedentary worker may
resume full-time work in an incremental fashion.
require a disability period of 6–12 months. About half of the
individuals complain of long-term posterior pelvic or low back pain
that impairs their recovery and may necessitate industrial retraining
or work modifications.
spontaneous loosening of the clamps. Provide patients with wrenches so
that they can tighten all the nuts weekly until the frame is removed.
Typically, the frame is removed under a short-acting general anesthetic
as an outpatient procedure.
immobilize the pelvis. When the horizontal bars that connect the
opposing hemipelves are removed, the residual portions of the frame can
be grasped and manipulated to document the degree of pelvic stability.
When persistent instability is evident, which is unusual, reassemble
the frame. After pin removal, irrigate with weak betadine solution and
apply dry dressings. Allow a partial weight-bearing gait with crutches,
and in the next month progress to full weight bearing. In more severe
pelvic disruption, progress weight bearing over an 8-week period. When
a stable union is evident radiographically and clinically, resume full
activity.
unstable pelvic injury. Therefore, it is important to be able to
recognize the various clinical presentations of inadequate fixation,
and to mount an appropriate response.
-
Once the pelvic instability is recognized, use supplementary skeletal traction for vertical instability.
-
If the general condition improves, add or replace with internal fixation.
presumed stable injury. The patient may complain of persistent
posterior pelvic pain that hampers sitting, transfers, or gait; or
radiographic loss of reduction is noted when the patient is mobilized.
-
Obtain additional radiographs or use CT to confirm an occult unstable posterior injury.
-
Perform secondary posterior internal fixation with possible replacement of the frame by anterior internal fixation.
isolated external fixation in an ill, elderly individual. In view of
the comorbidities, there is usually no possibility of later internal
fixation.
-
After 6 weeks of conservative treatment, remove the frame and associated skeletal traction.
-
Mobilize the patient to tolerance.
-
Once the general condition and nutrition
of the patient have stabilized, evaluate the residual pelvic problem
clinically and radiographically.
-
Lack of an integrated acute resuscitative
protocol. Prior to the emergency management of these patients, the
trauma team must have a cohesive resuscitative and diagnostic protocol. -
Needless deferral of definitive pelvic
reconstruction. Deferral of definitive surgical treatment of the
disrupted pelvis beyond the optimal period of a few days after injury
leads to enforced recumbency with its complications: increased
technical difficulties with open reduction, increased pain, and other
systemic problems. -
Inadequate preoperative imaging. A
thorough understanding of the injury through a complete radiographic
assessment, including CT and potentially 3D CT, is necessary to
formulate an appropriate operative strategy. -
Inadequate preoperative planning and lack
of appropriate resources for effective pelvic reconstruction. Pelvic
fractures and disruptions are complex. Meticulous preoperative planning
and often consultation with a more experienced surgeon, even in another
location, is necessary to devise appropriate fixation and avoid
complications. Early transfer of the patient to a regional center that
possesses specialized resources and experience may be prudent. -
Excessive use of external fixation.
External fixation is useful for open pelvic fractures, unstable pelvic
injuries with acute hemodynamic instability, and external rotation
injuries with a symphysis diastasis, but it is not a particularly
effective method for many unstable disruptions. External fixation
compromises the cutaneous envelope and increases the risk of infection.
External fixation tends to be overused on stable but perhaps slightly
deformed internal rotation injuries, where immediate weight bearing to
tolerance would be a better choice.
associated major injuries can result in fatal hemorrhage. Prolonged
extrication at the accident site, delays in transfer of the patient to
an appropriate treatment facility, hypothermia, and antecedent
comorbidities are contributing factors. The risk of hemorrhage can be
minimized with standing protocols for resuscitation and an integrated
team approach to management.
immediate laparotomy in the operating room and early reduction and
stabilization of pelvic disruptions. See Chapter 5 and Chapter 14 for more details.
of pin loosening or infection, and inadequacies of the reduction and
fixation. Improper insertion and location of fixation pins in the ilium
usually results in early loss of fixation. This leads to pain, loss of
reduction, and pin-track infection. Open insertion of the pins with
exposure of both tables of the ilium minimizes the risk in the hands of
less experienced surgeons. A snug fit of the skin around the pins to
avoid excessive motion at the skin–pin interface or skin tension at the
pin site minimizes the risk of wound problems.
unstable pelvic injuries merit serious consideration for supplementary
internal fixation or replacement with internal fixation.
fixation of the pelvis are caused by a lack of appreciation for the
complexity of the deformity, particularly rotary displacement, which
results in inadequate fixation leading to persistent instability,
nonunion, and malunion. Referral of the patient to a center that
specializes in treating pelvic injuries is advisable in many cases. A
thorough understanding of the mechanism of injury, the 3D pathologic
anatomy, complete imaging, and careful preoperative planning will
minimize these risks. The risk of postoperative infection of pelvic
surgery is significant, as the coexistence of other injuries
compromises the patient’s immunologic status and provides additional
sites for bacterial contamination and infection, such as intravenous
lines and suprapubic catheters. For that reason, minimally invasive,
percutaneous techniques are advantageous. The soft-tissue envelope
around the pelvis may also be compromised, requiring alternative
approaches or delay in the surgery. See Chapter 18 for more details.
morbidity after a pelvic ring disruption, particularly a posterior ring
disruption. The incidence of neurologic injury varies between 2% and
50% (23,51,56,72), depending on the mechanism and severity of the injury. Typically, the injury involves the lumbosacral
trunk and/or the superior gluteal nerve. The lumbosacral trunk is
injured as a result of traction from significant external rotation and
posterosuperior displacement of the hemipelvis. The superior gluteal
nerve usually is injured directly by fracture fragments near the SI
joint. Neurologic assessment may be hampered by the presence of a
concomitant head injury, intubation, and the influence of medications
used for sedation or anesthesia. Carefully assess bowel and bladder
function to rule out sacral plexus injury. Urologic consultation may be
helpful. Myelography and EMG studies can help in the diagnosis after 3
weeks.
Most nerve injuries are traction induced and require early conservative
treatment. Some advocate prompt exploration and decompression of sacral
nerve roots, although results have not demonstrated a clear benefit (52).
The results of surgical repair of the sciatic nerve are poor, so
surgical exploration is limited to situations where impalement of the
nerve on a bone spike or in a fracture merits removal of the offending
bone fragment.
immediately subcutaneous. The unusual problems are a painful plate on
the anterior iliac crest that is aggravated by a belt, or a plate on
the superior pubic rami and symphysis in a lean individual that causes
dyspareunia. Be aware that pain near hardware may represent an occult
nonunion, for which a CT scan, tomograms, or a technetium bone scan
merit consideration. Rather than removal, revision of the fixation and
bone graft may be necessary. Transverse posterior rods are nearly
always symptomatic and merit removal.
subsequent fatal PE is a common complication of a pelvic fracture for
which the risk can be minimized by the use of appropriate prophylactic
measures. The availability of enhanced DVT screening techniques such as
magnetic resonance venography has led to earlier and more frequent
detection of proximal and distal clots in the venous system (2,47,48 and 49,74).
While a distal thrombus is not as likely to embolize, a proximal
thrombus is at high risk for dislodgement and fatal PE. In a pelvic
fracture victim, the incidence of DVT is 35% to 60%, and the incidence
of a proximal clot is 25% to 35%. The incidence of a subsequent PE is
2% to 12%, with a 0.5% to 10% risk of a fatal PE. These rates may
become much higher if the patient has sustained polytrauma with the
involvement of multiple organ systems. A high injury severity score,
increased age, a direct venous injury, multiple blood transfusions, a
concomitant lower extremity or spinal cord injury, and a past history
of venous stasis disease increase the risk. Prophylaxis and treatment
for thromboembolic disease are discussed in Chapter 5.
Other sites of late discomfort are less common. Frequently, the
specific site of the pain is difficult to identify. More than half of
patients with pelvic fractures have also sustained injuries to the
lumbosacral spine. Examples include fractures of the transverse and
spinous processes, a crack in a vertebral body, a herniated nucleus
pulposus, and a traumatic spondylosis or spondylolisthesis. Associated
neurologic injury is common as well. The SI joint may be a source of
pain because of instability or posttraumatic arthritis. All of these
possibilities must be considered.
velocities and an MRI or myelography may be useful for evaluating
neurologic pain. Evaluate the SI joint with plain radiographs and CT as
well as a CT-guided injection of local anesthetic and a technetium bone
scan. Before considering fusion of a posttraumatic arthritis seen on a
CT scan, it is important to check for increased activity on a bone scan
and temporary relief of pain after a CT-guided injection of xylocaine.
If these tests are negative, an SI fusion is unlikely to result in
relief of pain.
is limited. Late sequelae may include incontinence, urinary
dysfunction, dyspareunia, dysmenorrhea, and difficulty with vaginal
delivery and overall sexual function. Overall, serious problems appear
to be relatively uncommon. Even after surgical fixation of the
symphysis and both SI joints, a vaginal delivery is uneventful in many
women. Evaluation for potential cephalopelvic disproportion, however,
is necessary. In male patients, impotence and urinary dysfunction are
the most common long-term genitourinary problems after a pelvic
fracture. In 145 male and female patients, Tscherne et al. (51)
reported only a 6% incidence of long-term urologic disturbance and less
than a 1% incidence of impotence and dyspareunia. In contrast, other
studies have reported rates of complete impotence as high as 50% (60,67,73). Further study is clearly needed.
reliance on external fixation remains a problem. As more orthopaedic
surgeons become familiar with the alternative techniques of internal
fixation, this problem should resolve.
and CT is crucial for preoperative planning to avoid inadequate
fixation. Because of the highly diverse fracture patterns, the presence
of concomitant injuries, and other variables of age and pretraumatic
health of the patients, the surgeon must be familiar with a wide range
of surgical methods and choose those best suited to the circumstances.
With the rapidly aging population and increasing incidence of
insufficiency fractures, improved fixation strategies for osteopenic
bone are needed. Because of the obvious advantages of minimally
invasive techniques, such as cannulated screw fixation under image
guidance, these techniques are being expanded and refined. Closed
reduction of these injuries remains a challenge.
computer-aided technologies are progressing rapidly. With the limited
size of bony targets and irregular columns of bone suitable for screw
fixation, computer-aided navigation should be an enormous technical
advance for pelvic surgery.
available new as a therapeutic aid. Between the period of acute
resuscitation and the definitive pelvic reconstruction, the use of
erythropoietin may help to lessen transfusion needs (40).
Late pelvic pain remains a perplexing problem that needs further
investigation to identify etiologies and design therapeutic
interventions.
scheme: *, classic article; #, review article; !, basic research
article; and +, clinical results/outcome study.
SF, Shah K, Jaffe J, et al. Arterial Embolization Is a Rapid and
Effective Technique for Controlling Pelvic Fracture Hemorrhage. J Trauma 1997;43:395.
CP, Marjolein CH, Goodman SB. Biomechanical Comparison of Posterior
Internal Fixation Techniques for Unstable Pelvic Fractures. J Orthop Trauma 1996;10:517.
CE, Bosse MJ, McCarthy ML, et al. Effect of Trauma and Pelvic Fracture
on Female Genitourinary, Sexual and Reproductive Function. J Orthop Trauma 1997;11:73.
SA, Burgess AR, Siegel JH, et al. Pelvic Fractures in Multiple Trauma:
Classification by Mechanism Is Key to Pattern of Organ Injury,
Resuscitation Requirements, and Outcome. J Trauma 1989;29:981.
FH, Hossenbaccus M, Duparc F, et al. Long-Term Functional Prognosis of
Posterior Injuries in High-Energy Pelvic Disruption. J Orthop Trauma 1998;12:145.
JA, Mason DL, Roubeau JP, Goldstein SA. Comminuted Posterior Wall
Acetabular Fractures: A Biomechanical Evaluation of Fixation Methods. J Bone Joint Surg [Am] 1994;76:1457.
DS, Mears DC, Cooperstein LA. Three-Dimensional Angio-Computed
Tomography: New Technique for Imaging the Acetabulum and Adjacent
Vessels in a Patient with Acetabular Protrusio. J Arthroplasty 1989;4:353.
D, Fishman EK, Ney DR, et al. Acetabular and Pelvic Fractures in the
Pediatric Patient: Value of Two- and Three-Dimensional Imaging. J Pediatr Orthop 1992;12:621.
DC, Durbhakula SM, Miller B. Developments in Blood Management: The
Potential Role for Epoetin Alfa in Orthopedic Trauma.
1999;22(Suppl.1):S151.
DC, Ward AJ, Wright MS. The Radiological Assessment of Pelvic and
Acetabular Fractures Using Three-Dimensional Computed Tomography. Int J Orthop Trauma 1992;2:196.
KD, Potter HG, Helfet DL. The Detection and Management of Proximal Deep
Venous Thrombosis in Patients with Acute Acetabular Fractures: A
Follow-Up Report. J Orthop Trauma 1997;11:330.
JR, Takayama TK, Defalco AJ. Traumatic Posterior Urethral Injury and
Early Realignment Using Magnetic Urethral Catheters. J Urol 1997;158:425.
BL, Butterfield SL, Diamond DL, et al. Acute Mortality Associated with
Injuries to the Pelvic Ring: The Role of Early Patient Mobilization and
External Fixation. J Trauma 1993;35:671.
MLC, Kregor PJ, Simonian PT, Mayo KA. Early Results of Percutaneous
Iliosacral Screws Placed with the Patient in the Supine Position. J Orthop Trauma 1995;9:207.
ML, Simonian PT, Defalco A. Internal Fixation in Pelvic Fractures and
Primary Repairs of Associated Genitourinary Disruptions—A Team
Approach. J Trauma 1996;40:784.
MLC, Simonian PT, Grujic L. The Retrograde Medullary Superior Pubic
Ramus Screw for the Treatment of Anterior Pelvic Ring Disruptions. J Orthop Trauma 1995;9:35.
A, Regel G, Tscherne H. Rehabilitation and Reintegration of Multiply
Injured Patients: An Outcome Study with Special Reference to Multiple
Lower Limb Fractures. Injury 1996;27:133.
JA, Mino DE, Werner FW, Murray DG. Posterior Stabilization of Pelvic
Fractures by Use of Threaded Compression Rods. Case Reports and
Mechanical Testing. Clin Orthop 1985;192:240.
M, Gordon RG, Mears DC, et al. Intraoperative Somato-Sensory Evoked
Potential Monitoring of Pelvic and Acetabular Fractures. J Orthop Trauma 1992;6:50.
LX, Rush PT, Fuller SB. Greenfield Filter Prophylaxis of Pulmonary
Embolism in Patients Undergoing Surgery of Acetabular Fracture. J Orthop Trauma 1992;6:139.
MG Jr, Zwally HJ II, Burgess AR. Innominosacral Dissociation: Mechanism
of Injury as a Predictor of Resuscitation Requirements, Morbidity, and
Mortality. J Orthop Trauma 1997;11:82.
P, Heini P, Ganz R. Pelvic Clamps for Controlling Shock in Posterior
Pelvic Ring Injuries. Applications, Biomechanical Aspects, and Initial
Clinical Results (German). Orthopade 1992;21:393.